��Ŀ����
��1��������������ʵ���Ũ�ȵĴ��������������Һ��Ϻ���Һ�� ������ԡ��������ԡ����ԡ�������Һ��c��Na+�� c��CH3COO-�����������=����������
��2��pH=3�Ĵ����pH=11������������Һ�������Ϻ���Һ�� ������ԡ��������ԡ����ԡ�����Һ��c��Na+�� c��CH3COO-�� ���������=����������
��3�����ʵ���Ũ����ͬ�Ĵ��������������Һ��Ϻ���Һ��CH3COO-��Na+Ũ����ȣ����Ϻ���Һ�� ������ԡ��������ԡ����ԡ������������ ����������Һ������������=����������
��2��pH=3�Ĵ����pH=11������������Һ�������Ϻ���Һ��
��3�����ʵ���Ũ����ͬ�Ĵ��������������Һ��Ϻ���Һ��CH3COO-��Na+Ũ����ȣ����Ϻ���Һ��
���㣺�����ʱ�Ķ����жϼ��й�ph�ļ���
ר�⣺����ƽ������Һ��pHר��
��������1������������ʵ���Ũ�ȵĴ��������������Һ��ϣ�ǡ�����ɴ����ƣ����������ˮ���Լ��ԣ�
��2��pH=3�Ĵ��ᣬ��Ũ�ȴ���0.001mol/L��pH=11���������ƣ���Ũ�ȵ���0.001mol/L���������Ϻ��������
��3�����ݵ���غ��֪��c��Na+��+c��H+��=c��OH-��+c��CH3COO-�����������Ũ�Ȼ��ʱ�Լ��ԣ�������ʱӦʹ���������
��2��pH=3�Ĵ��ᣬ��Ũ�ȴ���0.001mol/L��pH=11���������ƣ���Ũ�ȵ���0.001mol/L���������Ϻ��������
��3�����ݵ���غ��֪��c��Na+��+c��H+��=c��OH-��+c��CH3COO-�����������Ũ�Ȼ��ʱ�Լ��ԣ�������ʱӦʹ���������
���
�⣺��1������������ʵ���Ũ�ȵĴ��������������Һ��ϣ�ǡ�����ɴ����ƣ����ڴ�������Ӳ���ˮ�⣬��Һ�Լ��ԣ���c��H+����c��OH-�������ݵ���غ�c��Na+��+c��H+��=c��OH-��+c��CH3COO-����֪��c��Na+����c��CH3COO-����
�ʴ�Ϊ�����ԣ�����
��2������Ϊ������ʣ�pH=3�Ĵ�����Һ��������Ũ��Ϊ0.001mol/L�������Ũ�ȴ���0.001mol/L����pH=11������������Һ��Ũ��Ϊ0.001mol/L������Һ�������Ϻ�����������Һ�����ԣ���c��H+����c��OH-������Һ�д��ڵ���غ㣺c��CH3COO-��+c��OH-��=c��Na+��+c��H+������c��Na+����c��CH3COO-����
�ʴ�Ϊ�����ԣ�����
��3�����ݵ���غ��֪��c��Na+��+c��H+��=c��OH-��+c��CH3COO-������Һ��CH3COO-��Na+Ũ����ȣ���c��H+��=c��OH-������ҺΪ���ԣ�
�����������Ũ�ȵĴ���������������Һ���ʱ�����Һ�Լ��ԣ���Ϊ���ԣ�Ӧʹ������������������������������Һ�������
�ʴ�Ϊ�����ԣ�����
�ʴ�Ϊ�����ԣ�����
��2������Ϊ������ʣ�pH=3�Ĵ�����Һ��������Ũ��Ϊ0.001mol/L�������Ũ�ȴ���0.001mol/L����pH=11������������Һ��Ũ��Ϊ0.001mol/L������Һ�������Ϻ�����������Һ�����ԣ���c��H+����c��OH-������Һ�д��ڵ���غ㣺c��CH3COO-��+c��OH-��=c��Na+��+c��H+������c��Na+����c��CH3COO-����
�ʴ�Ϊ�����ԣ�����
��3�����ݵ���غ��֪��c��Na+��+c��H+��=c��OH-��+c��CH3COO-������Һ��CH3COO-��Na+Ũ����ȣ���c��H+��=c��OH-������ҺΪ���ԣ�
�����������Ũ�ȵĴ���������������Һ���ʱ�����Һ�Լ��ԣ���Ϊ���ԣ�Ӧʹ������������������������������Һ�������
�ʴ�Ϊ�����ԣ�����
���������⿼��������Լ���ҺpH�Ĵ�С�Ƚϣ�Ϊ�߿��������ͣ���Ŀ�Ѷ��еȣ�����ע�����Һ�����ԵĽǶȱȽ���Һ����Ũ�ȵĴ�С��ϵ�������������ˮ���ԭ������Ӧ�÷�����

��ϰ��ϵ�д�
 ǧ�������������ĩ�����Ծ�����ϵ�д�
ǧ�������������ĩ�����Ծ�����ϵ�д�
�����Ŀ
�йص�ⱥ��ʳ��ˮ��˵����ȷ���ǣ�������
| A���������������� |
| B����������Na+Ũ�Ȳ��� |
| C��ˮ�Ȳ���������Ҳ���ǻ�ԭ�� |
| D����Ӧ�����ǰѻ�ѧ��ת���ɵ��� |
������γ���Ҫ�����ڣ�������
| A��ɭ����ҿ��ҷ����ƻ�����̬���� |
| B�������ų�����β�� |
| C�������ж�����̼�ĺ������� |
| D������ʯȼ�ϵĴ���ȼ�� |
���ʯ��ʯī����̼Ԫ����ɵ����ֽṹ��ͬ�ĵ��ʣ�ͬ�������壩����100kPaʱ��1 molʯīת��Ϊ���ʯ��Ҫ����1.895kJ�����ܣ��ݴˣ����ж���100kPaѹǿ�£����н�����ȷ���ǣ�������
| A��ʯī�Ƚ��ʯ�ȶ� |
| B�����ʯ��ʯī�ȶ� |
| C��1 molʯī��1 mol ���ʯ���������� |
| D��1 mol���ʯ��1 mol ʯī���������� |
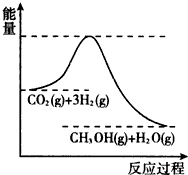 ��������ЧӦ����Դ��ȱ�����⣬��ν��ʹ����е�CO2���������Կ������ã������˸������ձ����ӣ�Ŀǰ��ҵ����һ�ַ�������CO2����ȼ�ϼ״���һ�������·�����Ӧ��CO2��g��+3H2��g��?CH3OH��g��+H2O��g�����÷�Ӧ�������仯��ͼ��ʾ��
��������ЧӦ����Դ��ȱ�����⣬��ν��ʹ����е�CO2���������Կ������ã������˸������ձ����ӣ�Ŀǰ��ҵ����һ�ַ�������CO2����ȼ�ϼ״���һ�������·�����Ӧ��CO2��g��+3H2��g��?CH3OH��g��+H2O��g�����÷�Ӧ�������仯��ͼ��ʾ��