��Ŀ����
13��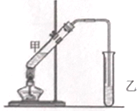 ���ڴ������������������ζ��������������ʵ��������Ҳ����������ͼ��ʾ��װ����ȡ�����������ش��������⣺
���ڴ������������������ζ��������������ʵ��������Ҳ����������ͼ��ʾ��װ����ȡ�����������ش��������⣺��1��д����ȡ���������Ļ�ѧ��Ӧ����ʽ��CH3COOH+C2H5OH$?_{��}^{ŨH_{2}SO_{4}}$CH3COOC2H5+H2O��
��2���ڴ��Թ�������һ���������Ҵ��������Ũ����Ļ��Һ�ķ����ǣ������Թ��м���һ�������Ҵ���Ȼ��ӱ����Թܽ�Ũ�������������Թܣ�����ټ������ᣮ
��3��Ũ����������ǣ��ٴ���������ˮ����
��4������ʵ���б���̼������Һ��������BD������ĸ����
A���к�������Ҵ�
B���к����Ტ���ղ����Ҵ�
C�������������ɣ���������
D�����������ڱ���̼������Һ�е��ܽ�ȱ���ˮ�и�С�������ڷֲ�����
��5����Ҫ���Ƶõ������������������Ӧ���õ�ʵ������Ƿ�Һ��
���� ��1��������Ҵ�����������Ӧ��������������ˮ��
��2���ȼ��Ҵ����ټ�Ũ���ᣬ�������ᣬ�������������ʣ�
��3��������ӦΪ���淴Ӧ����Ũ���������ˮ�ԣ�
��4������̼������Һ�������Ҵ�����ȥ���ᡢ���������������ܽ�ȣ�
��5������̼������Һ�����������ֲ㣬�������������ܶȱ�ˮ���ܶ�С���ֲ�������������ϲ㣮
��� �⣺��1��������Ҵ�����������Ӧ��������������ˮ���Ʊ���ӦΪCH3COOH+C2H5OH$?_{��}^{ŨH_{2}SO_{4}}$CH3COOC2H5+H2O��
�ʴ�Ϊ��CH3COOH+C2H5OH$?_{��}^{ŨH_{2}SO_{4}}$CH3COOC2H5+H2O��
��2���ڴ��Թ�������һ���������Ҵ��������Ũ����Ļ��Һ�ķ����������Թ��м���һ�������Ҵ���Ȼ��ӱ����Թܽ�Ũ�������������Թܣ�����ټ������ᣬ�������������ʣ�
�ʴ�Ϊ�������Թ��м���һ�������Ҵ���Ȼ��ӱ����Թܽ�Ũ�������������Թܣ�����ټ������
��3��������ӦΪ���淴Ӧ����Ũ���������ˮ�ԣ���Ũ���������Ϊ��������ˮ����
�ʴ�Ϊ����������ˮ����
��4������̼������Һ�������Ҵ�����ȥ���ᡢ���������������ܽ�ȣ�ֻ��BD���ϣ��ʴ�Ϊ��BD��
��5������̼������Һ�����������ֲ㣬��Ҫ���Ƶõ������������������Ӧ���õ�ʵ������Ƿ�Һ���ʴ�Ϊ����Һ��
���� ���⿼���л�����Ʊ�ʵ�飬Ϊ��Ƶ���㣬�����л���Ľṹ�����ʡ��л���ӦΪ���Ĺؼ������ط�����ʵ�������Ŀ��飬ע����������Ӧ�ã���Ŀ�ѶȲ���

| A�� | FeC13��MnO2���ɼӿ�H2O2�ֽ⣬ͬ�������¶��߶�H2O2�ֽ����ʵĸı���ͬ | |
| B�� | �����������䣬��ϡH2SO4�����Zn�Ļ�����м�����ͭƬ����������H2�������ʣ����ܼ�С����H2�����ʵ��� | |
| C�� | �����������䣬���Ȼ��ѹ����ʹH2��g��+I2��g��?2HI��g����v����v��ͬʱ���� | |
| D�� | v=$\frac{��c}{��t}$�ǻ�ѧ�ϱ�ʾ��ѧ��Ӧ���ʵ�Ψһ���� |
 ��
��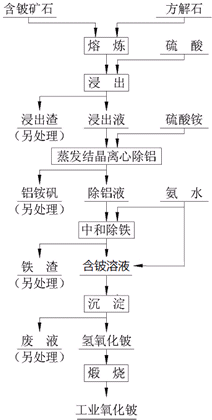 ���ᷨ���ִ���������������������й㷺Ӧ�õķ���֮һ����ԭ��������Ԥ�����ƻ���������ʯ-3BeO•Al2O3•6SiO2������FeO�ȣ��Ľṹ�뾧�ͣ��ٲ���������⺬����ʹ�롢�������������Խ���������Һ�࣬������ʯ����������룬Ȼ������Һ���о��������ӣ����յõ��ϸ�������루 ���������룩 ��Ʒ���乤��������ͼ��
���ᷨ���ִ���������������������й㷺Ӧ�õķ���֮һ����ԭ��������Ԥ�����ƻ���������ʯ-3BeO•Al2O3•6SiO2������FeO�ȣ��Ľṹ�뾧�ͣ��ٲ���������⺬����ʹ�롢�������������Խ���������Һ�࣬������ʯ����������룬Ȼ������Һ���о��������ӣ����յõ��ϸ�������루 ���������룩 ��Ʒ���乤��������ͼ��
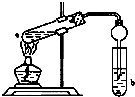 ��ͼ�����Թ�a���ȼ���3mL���Ҵ�����ҡ��������2mLŨ���ᣬ�ټ���2mL��ˮ���ᣬ�ò�������ֽ�����Թ̶ܹ�������̨�ϣ����Թ�b�м�����������̼������Һ�����Ӻ�װ�ã��þƾ��ƶ��Թܼ��ȣ����۲쵽�Թ�b������������ʱֹͣʵ�飮
��ͼ�����Թ�a���ȼ���3mL���Ҵ�����ҡ��������2mLŨ���ᣬ�ټ���2mL��ˮ���ᣬ�ò�������ֽ�����Թ̶ܹ�������̨�ϣ����Թ�b�м�����������̼������Һ�����Ӻ�װ�ã��þƾ��ƶ��Թܼ��ȣ����۲쵽�Թ�b������������ʱֹͣʵ�飮