��Ŀ����
��1������������N2H4Ϊȼ��,NO2Ϊ������,���߷�Ӧ����N2��ˮ����, ��֪
N2(g)+2O2(g)=2NO2(g) H1 ���� 67.7kJ/mol
N2H4(g)+O2(g)= N2(g)+2H2O(g) H2���� 534kJ/mol
��д��N2H4�� NO2��Ӧ���Ȼ�ѧ����ʽ ____________
(2)��������������Ӧ����1molˮ��������241.8KJ��д���÷�Ӧ���Ȼ�ѧ����ʽ_______________________ __________ ��
��1gˮ����ת����Һ̬ˮʱ����2.444KJ����ӦH2(g) + 1/2O2(g) == H2O(l)��
��H=______________ kJ?![]() ��
��
2N2H4(g)+2NO2(g)=3N2(g)+4H2O(g) ��H��1135.7 kJ?mol��1
N2H4(g)+2F2(g)=N2(g)+4HF(g) ��H��1126kJ?mol��1

��ϰ��ϵ�д�
 �ʰ�Ӣ��ͬ����ϰ��ϵ�д�
�ʰ�Ӣ��ͬ����ϰ��ϵ�д� ѧϰʵ����ϵ�д�
ѧϰʵ����ϵ�д�
�����Ŀ
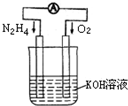 ���û�ѧ��Ӧԭ���о��������ȵ��ʼ��仯����ķ�Ӧ����Ҫ���壮
���û�ѧ��Ӧԭ���о��������ȵ��ʼ��仯����ķ�Ӧ����Ҫ���壮