��Ŀ����
3��ij����ѧϰС�����۱�������˵����������ȷ˵������ĿΪ���������ٹ�ҵ�ϳɰ����˹��̵�����Ҫ��������ҵ�������r��ˮ���Ҫ�õ�����ʯ��ʯ
��AlCl3��Һ��Al��OH��3����ı������������Ƿ��ж����ЧӦ
�����涨0.024kg12C���е�ԭ����Ϊ�����ӵ����������״����44gCO2�����ԼΪ44.8L����������Ĵ�С��Ҫȡ���ڷ�����Ŀ�ͷ��Ӽ��ƽ������
��ͬ��ͬѹ�£��������Ķ�����������Ͷ�������̼�����ܶȱ�Ϊ16��11�������Ϊ11��16
�ް�0.05mol��������������뵽100mL1.0mol/L�������У���Һ�����Ի�������
�߹�ҵ����ȡ���ý����ء��ơ��ơ�þ�������ɵ���Ӧ�������������Ƶ�
��ͬ�������壺C60��12C�����ʯ��ʯī��
| A�� | 3 | B�� | 4 | C�� | 5 | D�� | 6 |
���� ���˹��̵�����Ϊ�������½���Ԫ�صĵ���ת��Ϊ������Ĺ��̣����ݹ�ҵ����������ˮ�ࡢƯ�ۼ�������ʯұ����ԭ�Ϸ������
����Һ�ͽ��屾�������Ƿ�ɢ����ֱ����С��
�۰���٤��������Ϊԭ����2����Ħ������Ҳ��仯Ϊԭ����2��������Ħ�����Ҳ�仯Ϊԭ����2����
���¶�ѹǿһ������ķ��Ӽ������ȣ������Ӽ����Զ���ڷ��ӵĴ�С����������������С������Ϊ������Ŀ�Ķ��٣�
��ͬ��ͬѹ�£��ܶ�֮�ȵ�����Է�������֮�ȣ����������Ϊ1g������n=$\frac{m}{M}$����������ʵ�����ͬ��ͬѹ�£����֮�ȵ������ʵ���֮�ȣ�
����Һ�ĵ������������ӵ�Ũ���йأ������ʵ�ǿ���أ�Ũ��Խ������Խǿ��Ũ��ԽС��������Խ�����Դ������
�߹�ҵ����ȡ���ý����ء��ơ��ơ�þ���ɵ���Ӧ�������Ȼ����Ƶã�
��ͬ����������ͬ��Ԫ����ɵIJ�ͬ���ʣ�
��� �⣺�ٹ�ҵ�ϳɰ���N2��H2��һ�������·�Ӧ����NH3�������˹��̵�������ͨ������ԭ�ϣ�ʯӢɰ��ʯ��ʯ������ȣ��ƹ�����ˮ���ԭ�ϣ�ʯ��ʯ�������ұ��������ԭ�ϣ�����ʯ����̿��ʯ��ʯ������Ҫ��ʯ��ʯΪԭ�ϣ��ʢ���ȷ��
����Һ�ͽ��屾�������Ƿ�ɢ����ֱ����С�����������ֻ��������Һ�ͽ����һ�����ʣ��ʢڴ���
�۰���٤��������Ϊԭ����2����Ħ������Ҳ��仯Ϊԭ����2��������Ħ�����Ҳ�仯Ϊԭ����2����44g������̼���ʵ���Ϊ0.5mol������Ħ�����Ϊ44.8L/mol�������涨0.024kg12C���е�ԭ����Ϊ�����ӵ����������״����44gCO2�����ԼΪ22.4L���ʢ۴���
��Ӱ����������������ӵĴ�С�����Ӽ�ľ����Լ���Ŀ���٣�һ���¶�ѹǿ������ķ��Ӽ������ȣ������Ӽ����Զ���ڷ��ӵĴ�С����������������С������Ϊ������Ŀ�Ķ��٣��¶�ѹǿ��֪��������Ĵ�С��Ҫȡ���ڷ�����Ŀ�ͷ��Ӽ��ƽ�����룬�ʢ���ȷ��
��ͬ��ͬѹ�£��ܶ�֮�ȵ�����Է�������֮�ȣ���������Ͷ�����̼������ܶ�֮��Ϊ64��44=16��11��ͬ��ͬѹ�£����֮�ȵ������ʵ���֮�ȣ��������Ķ�������Ͷ�����̼��������֮��Ϊ$\frac{1}{64}$mol��$\frac{1}{44}$mol=11��16���ʢ���ȷ��
����������Һ�м���0.05molBa��OH��2���壬���������������Ӧ�����Ȼ�����ˮ��������Ȼ���ת��Ϊ�Ȼ���������ǿ����ʣ���Һ����Ũ�ȱ仯����ֻ�����ӵ�ת����������Һ���������仯���ʢ���ȷ��
�߹�ҵ����ȡ���ý����ء��ơ��ơ�þ���ɵ���Ӧ�������Ȼ����Ƶã����ǵ�������������Ʊ����ʢߴ���
��̼Ԫ�ص�ͬ�������壺C60�����ʯ��ʯī��12C��̼Ԫ�ص�һ��ԭ�Ӳ��ǵ��ʣ��ʢ����
��ѡB��
���� ���⿼�������ʷ��࣬�������ʣ�����Ħ���������ʵ�����⣬�����ᴿ������������ԭ��Ӧʵ�ʵȻ�ѧ֪ʶ������Ӧ�ã����ջ�����ϸ�·����ǽ���ؼ�����Ŀ�Ѷ��еȣ�

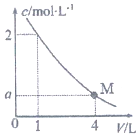 ��ͼ��BaC12��Һ��ϡ�����У�c��Ba2+������Һ����ı仯����ͼ����M��ʱ����Һ��c��Cl-��Ϊ��������
��ͼ��BaC12��Һ��ϡ�����У�c��Ba2+������Һ����ı仯����ͼ����M��ʱ����Һ��c��Cl-��Ϊ��������| A�� | 0.25mol/L | B�� | 0.5mol/L | C�� | 1 mol/L | D�� | 2mol/L |
 ��ʹ������к͵ζ����ⶨ���۰״���������g•100mL-1����
��ʹ������к͵ζ����ⶨ���۰״���������g•100mL-1������ʵ�鲽��
��1������100mL����״���Һ������ʽ�ζ��ܣ����������ƣ���ȡ10.00mLʳ�ð״ף����ձ�����ˮϡ�ͺ�ת�Ƶ�100ml����ƿ�����������ƣ��ж��ݣ�ҡ�ȼ��ô���״���Һ��
��2������ʽ�ζ���ȡ����״���Һ20.00mL����ƿ�У������еμ�2�η�̪��ָʾ����
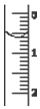
��3����ȡʢװ0.1000mol•L-1 NaOH ��Һ�ļ�ʽ�ζ��ܵij�ʼ���������Һ��λ����ͼ��ʾ�����ʱ�Ķ���Ϊ0.60mL��
��4���ζ�������Һ����ɫǡ�ñ�Ϊ��ɫ�����ڰ�����ڲ���ɫʱ��ֹͣ�ζ�������¼NaOH��Һ���ն������ظ��ζ�3�Σ�
��ʵ���¼
| �ζ�����ʵ�����ݣ�mL�� | 1 | 2 | 3 | 4 |
| V����Ʒ�� | 20.00 | 20.00 | 20.00 | 20.00 |
| V��NaOH�������ģ� | 15.95 | 15.00 | 15.05 | 14.95 |
��1����ʵ���������ݣ��ɵ�c�����۰״ף�=0.75mol•L-1�����۰״�������=4.5g•100mL-1��
��2���ڱ�ʵ��ĵζ������У����в�����ʹʵ����ƫ�����ab��д��ţ���
a����ʽ�ζ����ڵζ�ʱδ�ñ�NaOH��Һ��ϴ
b����ʽ�ζ��ܵļ����ڵζ�ǰ�����ݣ��ζ���������ʧ
c����ƿ�м������״���Һ���ټ�����ˮ
d����ƿ�ڵζ�ʱ����ҡ����������Һ�彦����
| A�� | Ag+��Cu2+��Sb3+ | B�� | Cu2+��Ag+��Sb3+ | C�� | Sb3+��Ag+��Cu2+ | D�� | Ag+��Sb3+��Cu2+ |
| A�� | ${\;}_{1}^{2}$Hԭ���е��������������� | |
| B�� | ${\;}_{18}^{36}$Arԭ���е��������������� | |
| C�� | ${\;}_{4}^{8}$Be2+�е��������͵����� | |
| D�� | ${\;}_{12}^{26}$Mg2+�е��������͵����� |
| A�� | ��״���£�224 mLˮ�к��еĵ�����Ϊ0.1NA | |
| B�� | 44 g��N2O��CO2��ɵĻ�������к��е�ԭ����Ϊ3NA | |
| C�� | ��20 mL 0.5 mol•L-1 FeCl3��Һ��У����ý�����ĿΪ0.01NA | |
| D�� | �ڷ�Ӧ��KClO3+6HCl�TKCl+3Cl2+3H2O�У�ÿ����3mol Cl2��ת�Ƶĵ�����Ϊ6NA |