��Ŀ����
4�����ڽ���п�����ļ�ֵ���ߣ����ҹ���ҵп���ϵĻ��������ʱȽϵͣ�ij�������о����ú�����ͭ�����Ĵ�п�Ʊ�����п��������ʵ���Դ�ۺ����ã��乤������ͼ��ͼ�м�������ʾ�Ϊ���������й��������£�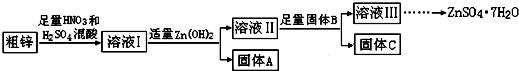
| ���� | Fe��OH��3 | Cu��OH��2 | Zn��OH��2 | CuS | ZnS |
| Ksp | 4.0��10-38 | 5.0��10-20 | 2.0��10-16 | 8.5��10-45 | 1.2��10-23 |
��1������A����Ҫ�ɷ���Fe��OH��3���������B����Ҫ�����dz�ȥ��Һ���е�Cu2+��
��2����п�е�ͭ��ϡ������Һ��Ӧ�����ӷ���ʽΪ3Cu+8H++2NO3-=3Cu2++2NO��+4H2O��
��3������ҺII��c��Cu2+��Ϊ0.05mol•L-1������ҺII��pH��5��
��4����B��Zn��ȡ8.320gC��ȫ�ܽ���500mL 1mol•L-1ϡ�����У����ռ���2240mL���壬����������Һ�м���NaOH��Һ���պ����ɳ�����࣬��ʱ���ó���������m����ȡֵ��Χ��13.42����B����һ�����ʣ�ȡ����C���Թ��У����������������г�����ζ���壬��÷�Ӧ�����ӷ���ʽΪZnS+2H+=H2S��+Zn2+��
��5����ҺIII�������루NH4��2S��Һ��Ӧ�Ʊ�ZnS��ʵ��������ѡ�ã�NH4��2S��Һ������Na2S��Һ��Ϊ��Ӧ�����Ϊ�����Ƶõ�ZnS�лẬ�н϶��Zn��OH��2���ʣ�
���� ���ú�ͭ�����Ĵ�п�Ʊ�����п����ͭ��ͭ���������ᡢ���ᷴӦ������ͭ���ӡ������ӣ�����Ksp��֪��������ͭ��������п�����ܣ�����Һ�м���Zn��OH��2���õ�����AΪFe��OH��3��������������B����B��ͬ����C�ɷֲ�ͬ������Һ��Ϊ����п��Һ���Ʊ���ˮ������п����ֹ�ᾧˮʧˮ������ȴ�ᾧ�����ˡ�ϴ�ӡ�������õ��ϴ���������п���壬
��1���������Ϸ�������ɷ�����֪Ҫ����ͭ���ӣ�
��2��Cu�����ᷴӦ��������ͭ��NO��ˮ��
��3�������ܶȻ�����������Һ�е�����������Ũ�ȣ��ټ���pH��
��4����B��Zn�������CΪZn��Cu�Ļ����ɹ�ϵʽZn��Cu��2e-��Zn2+��Cu2+��2OH-��Cu��OH��2��Zn��OH��2��֪���������������Ϊ���������ӵ�������
��5��Na2S����Һ��ˮ���Լ��ԣ�
��� �⣺���ú�ͭ�����Ĵ�п�Ʊ�����п����ͭ��ͭ���������ᡢ���ᷴӦ������ͭ���ӡ������ӣ�����Ksp��֪��������ͭ��������п�����ܣ�����Һ�м���Zn��OH��2���õ�����AΪFe��OH��3��������������B����B��ͬ����C�ɷֲ�ͬ������Һ��Ϊ����п��Һ���Ʊ���ˮ������п����ֹ�ᾧˮʧˮ������ȴ�ᾧ�����ˡ�ϴ�ӡ�������õ��ϴ���������п���壬
��1�������Ϸ�����֪����A����Ҫ�ɷ���Fe��OH��3������B��Ϊ������ͭ������ͭ����п��п�ȣ����ڳ�ȥ��Һ���е�Cu2+��
�ʴ�Ϊ��Fe��OH��3����ȥ��Һ���е�Cu2+��
��2����п�е�ͭ��ϡ������Һ��Ӧ�����ӷ���ʽΪ3Cu+8H++2NO3-=3Cu2++2NO��+4H2O���ʴ�Ϊ��3Cu+8H++2NO3-=3Cu2++2NO��+4H2O��
��3������Һ����c��Cu2+��Ϊ0.05mol•L-1����Ksp=5.0��10-20��֪��c��OH-��=$\sqrt{\frac{5��1{0}^{-20}}{0.05}}$mol/L=1.0��10-9mol/L����c��H+��=1.0��10-5mol/L��
����pH��5��
�ʴ�Ϊ��5��
��4����B��Zn�������CΪZn��Cu�Ļ���ȡ8.320gC��ȫ�ܽ���500mL1mol•L-1ϡ�����У����ռ���2240mL���壬��n��NO��=$\frac{2.24L}{22.4L/mol}$=0.1mol��ת��0.3mol���ӣ��ɹ�ϵʽZn��Cu��2e-��Zn2+��Cu2+��2OH-��Cu��OH��2��Zn��OH��2��֪��������Ϊ8.320g+0.3mol��17g/mol=13.42g��������B����һ�����ʣ�ȡ���ֹ���C���Թ��У��������������ᣬ�г�������ζ���������������Ϊ���⣬��ʵ��Ϊ�Ʊ�����п����BӦΪZnS������C�к���ZnS��CuS�������ᷴӦ�������������Ϊ��п����ӦΪ��ZnS+2H+=H2S��+Zn2+��
�ʴ�Ϊ��13.42��ZnS+2H+=H2S��+Zn2+��
��5��Na2S����Һ��ˮ���Լ��ԣ������Ƶõ�ZnS�лẬ�н϶��Zn��OH��2��
�ʴ�Ϊ��Zn��OH��2��
���� ���⿼�������ú�ͭ�����Ĵ�п�Ʊ�����п�����̷�����������ѧ���ķ�����ʵ��ͼ��������Ŀ��飬�漰���Ļ���������ܽ�ƽ��Ӧ�á�������ԭ��֪ʶ����������ܽ�ƽ����йؼ����ǽⱾ��ؼ�����Ŀ�Ѷ��еȣ�

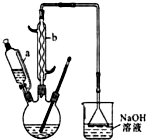 ������
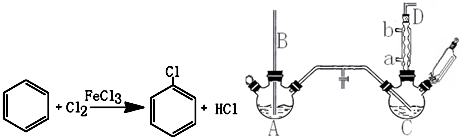
������ 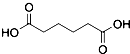 ��һ�ֹ�ҵ�Ͼ�����Ҫ������л���Ԫ�ᣬ�ڻ����������л��ϳɹ�ҵ��ҽҩ��������ȷ��涼����Ҫ���ã��ܹ��������η�Ӧ��������Ӧ�ȣ��������Ԫ�����۳ɸ߷��Ӿۺ���ȣ���������������ж�Ԫ�����еĵڶ�λ��ʵ���Һϳɼ�����ķ�Ӧԭ����ʵ��װ��ʾ��ͼ��
��һ�ֹ�ҵ�Ͼ�����Ҫ������л���Ԫ�ᣬ�ڻ����������л��ϳɹ�ҵ��ҽҩ��������ȷ��涼����Ҫ���ã��ܹ��������η�Ӧ��������Ӧ�ȣ��������Ԫ�����۳ɸ߷��Ӿۺ���ȣ���������������ж�Ԫ�����еĵڶ�λ��ʵ���Һϳɼ�����ķ�Ӧԭ����ʵ��װ��ʾ��ͼ��3
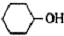 +8HNO3��3
+8HNO3��3 +8NO��+7H2O
+8NO��+7H2O�����õ����й����������
| ���� | �ܶȣ�20�棩 | �۵� | �е� | �ܽ��� | ��Է������� |
| ������ | 0.962g/cm3 | 25.9�� | 160.8�� | 20��ʱˮ���ܽ��3.6g���ɻ������Ҵ����� | 100 |
| ������ | 1.36g/cm3 | 152�� | 337.5�� | ��ˮ�е��ܽ�ȣ�15��ʱ1.44g��25��ʱ2.3g���������Ҵ��������ڱ��� | 146 |
I����������ƿ�м���16mL 50%�����ᣨ�ܶ�Ϊ1.31g/cm3�����ټ���1��2����ʯ����Һ©����ʢ����5.4mL��������
II��ˮԡ����������ƿ��50�����ң���ȥˮԡ�������μ�5��6�λ�������ҡ��������ƿ���۲쵽�к���ɫ����ų�ʱ�������μ�ʣ�µĻ�������ά�ַ�Ӧ�¶���60�桫65��֮�䣮
III����������ȫ��������������80�桫90��ˮԡ����Լ10min��ע������¶ȣ���ֱ������ɫ��������Ϊֹ��
IV�����Ƚ���ӦҺ�����ձ��У������ˮԡ����ȴ�������������ˡ�ϴ�ӵôֲ�Ʒ��
V���ֲ�Ʒ���ᴿ�����Ϊ5.7g��
��ش��������⣺
��1������b������Ϊ���������ܣ��������ܣ���
��2����������ƿ�еμӻ�����ʱ��Ҫ���ƺû������ĵ������ʣ���ֹ��Ӧ���ھ��ҵ����¶�Ѹ����������������ɽ����صĺ�������о�һ�����ܲ����ĺ����
��ӦҺ���г�������ܣ����ȹ����������ը��������NO2��������������Һ���ն����ݵ������У�
��3����֪��NaOH��Һ����β��ʱ��������ط�Ӧ����ʽΪ��
2NO2+2NaOH=NaNO2+NaNO3+H2O NO+NO2+2NaOH=2NaNO2+H2O��������ô�����Һ����β��ʱҲ�ܷ������Ʒ�Ӧ������ط�Ӧ����ʽΪ��
2NO2+Na2CO3=NaNO2+NaNO3+CO2��NO+NO2+Na2CO3=2NaNO2+CO2��
��4��Ϊ�˳�ȥ���ܵ����ʺͼ��ٲ�Ʒ��ʧ���ɷֱ��ñ�ˮ�ͱ�ϴ�Ӿ��壮
��5���ֲ�Ʒ�����ؽᾧ ���ᴿ����ʵ��������ƣ�����ʵ�����õ��ļ��������Ϊ75%��
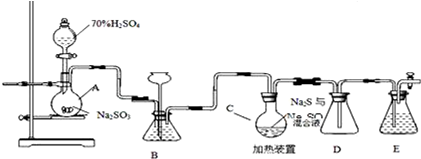
��֪���ȱ�Ϊ��ɫҺ�壬�е�132.2�棬�����²�������������Һ��Ӧ��
�ش��������⣺
��1��A��Ӧ��������ʵ���ҷ���ȡ�������пղ�����B��������ƽ����ѹ������������ˮӦ��a���a����b������ͨ�룮
��2���Ѹ��������ͨ��װ�и��ﱽ�ķ�Ӧ��C�У������൱�ڱ���1%����м��������������ά�ַ�Ӧ�¶���40��60��Ϊ�ˣ��¶ȹ������ɶ��ȱ���
�ٶ�C���ȵķ�����c������ţ���
a���ƾ��Ƽ��� b���ƾ���Ƽ��� c��ˮԡ����
��D���ڵ�����ɷ���HCl����������������
��3��C��Ӧ����Ӧ��ɺ�ҵ��Ҫ����ˮϴ����ϴ��ʳ�θ����������ϴ֮ǰҪˮϴ��Ŀ����ϴȥ�������ͬʱ���ټ����������Լ�ɱ���д����10%�������Ƽ�ϴʱ���ܷ����Ļ�ѧ��Ӧ����ʽ��FeCl3+3NaOH=Fe��OH��3��+3NaCl��HCl+NaOH=NaCl+H2O��д�������ɣ���
��4������װ��ͼ��A��C��Ӧ��֮�䣬��Ҫ����һ��U�ιܣ������������������������Ȼ��ƣ�
��5����ҵ�����б�����ʧ������£�
| ��Ŀ | ���ȱ� | β�� | ��ȷ������ | ��ʧ���� |
| ����ʧ����kg/t�� | 13 | 24.9 | 51.3 | 89.2 |
 ��ȩ��518��ʱ��;�����;����ֽ�Ϊ�����һ����̼����ͼ��ʾ����;���������仯����ʾ��ͼ�����������У�������ǣ�������
��ȩ��518��ʱ��;�����;����ֽ�Ϊ�����һ����̼����ͼ��ʾ����;���������仯����ʾ��ͼ�����������У�������ǣ�������| A�� | ��������������ʱ��;������;��������ȩ��ת������ͬ | |
| B�� | ;�����Ǽ������ʱ�����仯���ߣ��Ҵ�����I2 | |
| C�� | ;������;������ʱ䣨��H����ͬ | |
| D�� | �����������������Ӧ���ʣ������淴Ӧ���� |
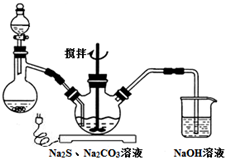 NaCNΪ�綾���ij��ȤС������ϵ�֪��ʵ�������NaCN��Һ��ʹ��Na2S2O3��Һ���нⶾ���٣����ǿ�չ����������ʵ�飬�����Ҫ��ش����⣺
NaCNΪ�綾���ij��ȤС������ϵ�֪��ʵ�������NaCN��Һ��ʹ��Na2S2O3��Һ���нⶾ���٣����ǿ�չ����������ʵ�飬�����Ҫ��ش����⣺