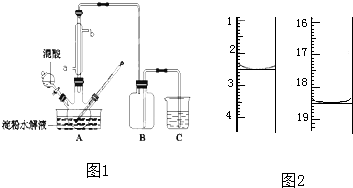
��Ŀ����
��A��B��C��D��E���ֳ���������������������е�һ�ֻ����γɵģ��������У�SO42-��NO3-��HCO3-��OH-���������У�Na+��K+��Cu2+��Al3+��
Ϊ�˼�������������ֱ��������ʵ�飬�����ǣ�
�ٷֱ���������ˮ��DΪ��ɫ��Һ��������Ϊ��ɫ��Һ��
�ڽ�E��Һ���뵽C��Һ�г��ְ�ɫ�����������μӣ������ܽ⣻
�۽�����ɫ��Ӧ��ֻ��B��CΪ��ɫ������ɫ�ܲ�������
���ڸ���Һ�м������ᱵ��Һ���ټӹ���ϡ���ᣬA�зų���ɫ���壬C��D�в�����ɫ������
�ݽ�B��D����Һ��ϣ�δ���������������ɣ�
��������ʵ����գ�
��1��д��B��D�Ļ�ѧʽ��B ��D ��
��2������1mol A����Һ�뺬1mol E����Һ��Ӧ�����ɣ����õ�һ�ֻ�����û�����Ļ�ѧʽΪ ��
��3����A��Һ�м�����������ʯ��ˮ�������ӷ���ʽΪ ��
Ϊ�˼�������������ֱ��������ʵ�飬�����ǣ�
�ٷֱ���������ˮ��DΪ��ɫ��Һ��������Ϊ��ɫ��Һ��
�ڽ�E��Һ���뵽C��Һ�г��ְ�ɫ�����������μӣ������ܽ⣻
�۽�����ɫ��Ӧ��ֻ��B��CΪ��ɫ������ɫ�ܲ�������
���ڸ���Һ�м������ᱵ��Һ���ټӹ���ϡ���ᣬA�зų���ɫ���壬C��D�в�����ɫ������
�ݽ�B��D����Һ��ϣ�δ���������������ɣ�
��������ʵ����գ�
��1��д��B��D�Ļ�ѧʽ��B
��2������1mol A����Һ�뺬1mol E����Һ��Ӧ�����ɣ����õ�һ�ֻ�����û�����Ļ�ѧʽΪ
��3����A��Һ�м�����������ʯ��ˮ�������ӷ���ʽΪ
���㣺���������ӵļ���,���������ӵļ���
ר�⣺
��������1��������ˮ��DΪ��ɫ��Һ��������Ϊ��ɫ��Һ����D����Ϊͭ���ӣ�
�ڽ�E��Һ���뵽C��Һ�г��ְ�ɫ�����������μӣ������ܽ⣬��֪E�к������������ӣ�C�к��������ӣ�
�۽�����ɫ��Ӧ��ֻ��B��CΪ��ɫ��˵��B��C�к��м����ӣ�
���ڸ���Һ�м������ᱵ��Һ���ټӹ���ϡ���ᣬA�зų���ɫ���壬A�к���̼��������ӣ�AΪ̼�����ƣ�C��D�в�����ɫ������˵��C��D�к�����������ӣ�CΪKAl��SO4��2��DΪCuSO4��
�ݽ�B��D����Һ��ϣ�δ���������������ɣ�DΪ����ͭ����Bֻ�ܺ�����������ӣ�Ϊ����أ�
��2��AΪ̼�����ƣ�EΪ�������ƣ����߷�Ӧ����̼���ƣ�
��3��̼�������������������Ʒ�Ӧ����̼��ơ�̼���ƺ�ˮ��
�ڽ�E��Һ���뵽C��Һ�г��ְ�ɫ�����������μӣ������ܽ⣬��֪E�к������������ӣ�C�к��������ӣ�
�۽�����ɫ��Ӧ��ֻ��B��CΪ��ɫ��˵��B��C�к��м����ӣ�
���ڸ���Һ�м������ᱵ��Һ���ټӹ���ϡ���ᣬA�зų���ɫ���壬A�к���̼��������ӣ�AΪ̼�����ƣ�C��D�в�����ɫ������˵��C��D�к�����������ӣ�CΪKAl��SO4��2��DΪCuSO4��
�ݽ�B��D����Һ��ϣ�δ���������������ɣ�DΪ����ͭ����Bֻ�ܺ�����������ӣ�Ϊ����أ�
��2��AΪ̼�����ƣ�EΪ�������ƣ����߷�Ӧ����̼���ƣ�
��3��̼�������������������Ʒ�Ӧ����̼��ơ�̼���ƺ�ˮ��
���
�⣺��1��������ˮ��DΪ��ɫ��Һ��������Ϊ��ɫ��Һ����D����Ϊͭ���ӣ�
�ڽ�E��Һ���뵽C��Һ�г��ְ�ɫ�����������μӣ������ܽ⣬��������ǿ�Ӧ����������������������������ƫ��������ӣ���֪E�к������������ӣ�C�к��������ӣ�
�۽�����ɫ��Ӧ��ֻ��B��CΪ��ɫ����Ԫ�����ճ�����ɫ������ɫ�ܲ�����������B��C�к��м����ӣ�
�ڸ���Һ�м������ᱵ��Һ���ټӹ���ϡ���ᣬA�зų���ɫ���壬�ܹ����ᷴӦ��������ĸ���������ֻ��̼������ˣ�����A�к���̼��������ӣ�AΪ̼�����ƣ�C��D�в�����ɫ���������������������Ӧ�������ᱵ��ɫ������˵��C��D�к�����������ӣ���CΪKAl��SO4��2��DΪCuSO4��
B��D����Һ��Ϻ�δ���������������ɣ���Bֻ�ܺ���NO3-������ΪKNO3��
��AΪNaHCO3��BΪKNO3��CΪKAl��SO4��2��DΪCuSO4��EΪNaOH��
�ʴ�Ϊ��KNO3��CuSO4��
��2��1mol̼��������1mol��������ǡ�÷�Ӧ����̼���ƣ��������ɺ�õ��Ĺ���ΪNa2CO3���ʴ�Ϊ��Na2CO3��
��3��̼�������������������Ʒ�Ӧ����̼��ơ�̼���ƺ�ˮ����Ӧ�����ӷ���ʽΪ��2HCO3-+Ca2++2OH-�TCaCO3��+CO32-+2H2O��
�ʴ�Ϊ��2HCO3-+Ca2++2OH-�TCaCO3��+CO32-+2H2O��
�ڽ�E��Һ���뵽C��Һ�г��ְ�ɫ�����������μӣ������ܽ⣬��������ǿ�Ӧ����������������������������ƫ��������ӣ���֪E�к������������ӣ�C�к��������ӣ�
�۽�����ɫ��Ӧ��ֻ��B��CΪ��ɫ����Ԫ�����ճ�����ɫ������ɫ�ܲ�����������B��C�к��м����ӣ�
�ڸ���Һ�м������ᱵ��Һ���ټӹ���ϡ���ᣬA�зų���ɫ���壬�ܹ����ᷴӦ��������ĸ���������ֻ��̼������ˣ�����A�к���̼��������ӣ�AΪ̼�����ƣ�C��D�в�����ɫ���������������������Ӧ�������ᱵ��ɫ������˵��C��D�к�����������ӣ���CΪKAl��SO4��2��DΪCuSO4��
B��D����Һ��Ϻ�δ���������������ɣ���Bֻ�ܺ���NO3-������ΪKNO3��
��AΪNaHCO3��BΪKNO3��CΪKAl��SO4��2��DΪCuSO4��EΪNaOH��
�ʴ�Ϊ��KNO3��CuSO4��
��2��1mol̼��������1mol��������ǡ�÷�Ӧ����̼���ƣ��������ɺ�õ��Ĺ���ΪNa2CO3���ʴ�Ϊ��Na2CO3��
��3��̼�������������������Ʒ�Ӧ����̼��ơ�̼���ƺ�ˮ����Ӧ�����ӷ���ʽΪ��2HCO3-+Ca2++2OH-�TCaCO3��+CO32-+2H2O��
�ʴ�Ϊ��2HCO3-+Ca2++2OH-�TCaCO3��+CO32-+2H2O��
���������⿼�������ʵ��ƶϣ����ճ������Ӿ��е�������Ӧ�ͼ��鷽���ǽ���ؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
��֪X��YԪ��ͬ���ڣ��ҵ�һ������X��Y������˵������ȷ���ǣ�������
| A���縺��YС��X |
| B��X��Y�γɻ�����ʱ��X�Ը��ۣ�Y������ |
| C����ۺ���������ԣ�X��Ӧ����������Y��Ӧ������ |
| D����̬�⻯����ȶ��ԣ�HmYС��HnX |
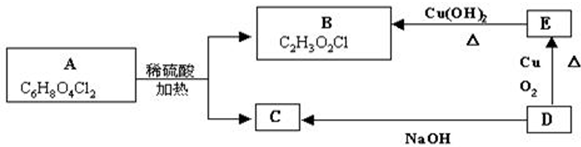
PHB������һ�ֿ������������½���Ļ��������ϣ���ṹ��ʽΪ��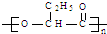 �������й�PHB˵���У�����ȷ���ǣ�������
�������й�PHB˵���У�����ȷ���ǣ�������
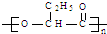 �������й�PHB˵���У�����ȷ���ǣ�������
�������й�PHB˵���У�����ȷ���ǣ�������| A��PHB��һ�־��� |
| B���ϳ�PHB�ĵ�����CH3CH2CH��OH��COOH |
| C��PHB�Ľ�����������CO2��H2O |
| D��PHBͨ���Ӿ۷�Ӧ�Ƶ� |