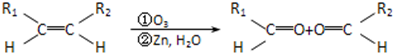
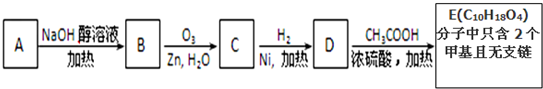
��Ŀ����
�л���AΪ����������ͼ��������Է�������Ϊ70������ط�Ӧ��ͼ��ʾ������B��D��E�Ľṹ�о�����2��-CH3�����ǵĺ˴Ź��������о�����4���壮��ش�

��1��D�ķ���ʽΪ ��
��2��E�����������ŵ�����Ϊ ��
��3����ķ�Ӧ����Ϊ ������ĸ��ţ���
a����ԭ��Ӧ b���ӳɷ�Ӧ c��������Ӧ d����ȥ��Ӧ
��4��C��E����һ�������·�Ӧ����F��FΪ����ζ���л�������÷�Ӧ�Ļ�ѧ����ʽΪ ��
��5��E�ж���ͬ���칹�壬����һ��ͬ���칹���ܷ���������Ӧ�����������������������������ܷ�����ȥ��Ӧ����ṹ��ʽΪ ��

��1��D�ķ���ʽΪ
��2��E�����������ŵ�����Ϊ
��3����ķ�Ӧ����Ϊ
a����ԭ��Ӧ b���ӳɷ�Ӧ c��������Ӧ d����ȥ��Ӧ
��4��C��E����һ�������·�Ӧ����F��FΪ����ζ���л�������÷�Ӧ�Ļ�ѧ����ʽΪ
��5��E�ж���ͬ���칹�壬����һ��ͬ���칹���ܷ���������Ӧ�����������������������������ܷ�����ȥ��Ӧ����ṹ��ʽΪ
���㣺�л�����ƶ�
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
������B����NaOH�Ҵ���Һ���ȵ������·�Ӧ����A����A�к���C=C���л���AΪ����������ͼ��������Է�������Ϊ70���������෨��
=5����A�ķ���ʽΪC5H10��B�ķ���ʽΪC5H11Br��D����������Һ��Ӧ��˵��D�к���-CHO��E�к���-COOH��B��D��E�Ľṹ�о�����2��-CH3�����ǵĺ˴Ź��������о�����4���壬˵������4�ֲ�ͬ��Hԭ�ӣ�
DӦΪ CH3-CH��CH3��-CH2-CHO��EӦΪ CH3-CH��CH3��-CH2-COOH��CΪ CH3-CH��CH3��-CH2-CH2OH��BΪCH3-CH��CH3��-CH2-CH2Br����AΪCH3-CH��CH3��-CH=CH2������л���Ľṹ�����ʽ����⣮
��1��DӦΪ CH3-CH��CH3��-CH2-CHO���ݴ���д����ʽ��
��2��EӦΪ CH3-CH��CH3��-CH2-COOH�����ݽṹ��ʽ���
��3���л������ķ�ӦΪ��ԭ��Ӧ��
��4��CΪ CH3-CH��CH3��-CH2-CH2OH��EӦΪ CH3-CH��CH3��-CH2-COOH�������ǻ������⣬����������Ӧ��
��5���ܷ���������Ӧ��˵������ȩ�������������������������������ܷ�����ȥ��Ӧ��˵������-OH�������ǻ�������̼��û����ԭ�ӣ�
| 70 |
| 14 |
DӦΪ CH3-CH��CH3��-CH2-CHO��EӦΪ CH3-CH��CH3��-CH2-COOH��CΪ CH3-CH��CH3��-CH2-CH2OH��BΪCH3-CH��CH3��-CH2-CH2Br����AΪCH3-CH��CH3��-CH=CH2������л���Ľṹ�����ʽ����⣮
��1��DӦΪ CH3-CH��CH3��-CH2-CHO���ݴ���д����ʽ��
��2��EӦΪ CH3-CH��CH3��-CH2-COOH�����ݽṹ��ʽ���
��3���л������ķ�ӦΪ��ԭ��Ӧ��
��4��CΪ CH3-CH��CH3��-CH2-CH2OH��EӦΪ CH3-CH��CH3��-CH2-COOH�������ǻ������⣬����������Ӧ��
��5���ܷ���������Ӧ��˵������ȩ�������������������������������ܷ�����ȥ��Ӧ��˵������-OH�������ǻ�������̼��û����ԭ�ӣ�
���
�⣺B����NaOH�Ҵ���Һ���ȵ������·�Ӧ����A����A�к���C=C���л���AΪ����������ͼ��������Է�������Ϊ70���������෨��
=5����A�ķ���ʽΪC5H10��B�ķ���ʽΪC5H11Br��D����������Һ��Ӧ��˵��D�к���-CHO��E�к���-COOH��B��D��E�Ľṹ�о�����2��-CH3�����ǵĺ˴Ź��������о�����4���壬˵������4�ֲ�ͬ��Hԭ�ӣ�
DӦΪ CH3-CH��CH3��-CH2-CHO��EӦΪ CH3-CH��CH3��-CH2-COOH��CΪ CH3-CH��CH3��-CH2-CH2OH��BΪCH3-CH��CH3��-CH2-CH2Br����AΪCH3-CH��CH3��-CH=CH2��
��1�������Ϸ�����֪DΪCH3-CH��CH3��-CH2-CHO������ʽΪC5H10O��
�ʴ�Ϊ��C5H10O��
��2��EΪ CH3-CH��CH3��-CH2-COOH������������-COOH������Ϊ�Ȼ���
�ʴ�Ϊ���Ȼ���
��3����Ӧ����D���ʣ�CH3��2CHCH2CHO�����������ӳɷ�Ӧ����C���ʣ�CH3��2CHCH2CH2OH��Ҳ���ڻ�ԭ��Ӧ��
�ʴ�Ϊ��ab��
��4��C��E����һ�������·�Ӧ����F��FΪ����ζ���л����������������Ӧ�������ǻ������⣬���Ը÷�Ӧ�Ļ�ѧ����ʽΪ��
�ʴ�Ϊ�� ��
��
��5����CH3��2CHCH2COOH����һ��ͬ���칹���ܷ���������Ӧ������������������������������-CHO��-OH���Ҳ��ܷ�����ȥ��Ӧ����ṹ��ʽΪ�� ��
��
�ʴ�Ϊ�� ��
��
| 70 |
| 14 |
DӦΪ CH3-CH��CH3��-CH2-CHO��EӦΪ CH3-CH��CH3��-CH2-COOH��CΪ CH3-CH��CH3��-CH2-CH2OH��BΪCH3-CH��CH3��-CH2-CH2Br����AΪCH3-CH��CH3��-CH=CH2��
��1�������Ϸ�����֪DΪCH3-CH��CH3��-CH2-CHO������ʽΪC5H10O��
�ʴ�Ϊ��C5H10O��
��2��EΪ CH3-CH��CH3��-CH2-COOH������������-COOH������Ϊ�Ȼ���
�ʴ�Ϊ���Ȼ���
��3����Ӧ����D���ʣ�CH3��2CHCH2CHO�����������ӳɷ�Ӧ����C���ʣ�CH3��2CHCH2CH2OH��Ҳ���ڻ�ԭ��Ӧ��
�ʴ�Ϊ��ab��
��4��C��E����һ�������·�Ӧ����F��FΪ����ζ���л����������������Ӧ�������ǻ������⣬���Ը÷�Ӧ�Ļ�ѧ����ʽΪ��

�ʴ�Ϊ��
 ��
����5����CH3��2CHCH2COOH����һ��ͬ���칹���ܷ���������Ӧ������������������������������-CHO��-OH���Ҳ��ܷ�����ȥ��Ӧ����ṹ��ʽΪ��
 ��
���ʴ�Ϊ��
 ��
��
���������⿼���л�����ƶϣ��漰±����������ȩ�������������ת��������ȷ��A�ķ���ʽ�ǹؼ���ע���������չ����ŵ�������ת������Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ��ְٷְټ���ϵ�д�
��ְٷְټ���ϵ�д� �����ƻ���ĩ��̶�100��ϵ�д�
�����ƻ���ĩ��̶�100��ϵ�д� �ܿ���ȫ��100��ϵ�д�
�ܿ���ȫ��100��ϵ�д�
�����Ŀ
��֪X��Y��Z��W��T�Ƕ�������ԭ���������������5������Ԫ�أ�����Y��X����Ԫ�ؿ������1��1��1��2��1��3��1��4�ȶ��ֻ���� Zԭ�������������Ǵ�����������3����W��Xͬ���壬W��T�γɵĻ�����WT�dz�������ĵ�ζƷ������˵����ȷ���ǣ�������
| A��ԭ�Ӱ뾶��T��W��Z��Y |
| B��W��T�ĵ��ʿ�ͨ����ⱥ�͵�WTˮ��Һ��� |
| C��Z��W�γɵ����ֻ������л�ѧ��������ȫ��ͬ |
| D��Ԫ��X��Z��T���γɶ����� |
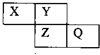 X��Y��Z��Q��Ϊ������Ԫ�أ����������ڱ������λ����ͼ��ʾ����Xԭ�ӵ������������ڲ��������2��������˵���У���ȷ���ǣ�������
X��Y��Z��Q��Ϊ������Ԫ�أ����������ڱ������λ����ͼ��ʾ����Xԭ�ӵ������������ڲ��������2��������˵���У���ȷ���ǣ�������| A��X��Q�Ļ������в����й��ۼ� |
| B������������Ӧˮ��������ԣ�Q��Zǿ |
| C���⻯���ȶ��ԣ�Y��Zǿ |
| D��Q��Fe��Ӧ���ɵĻ������У���Ԫ����+3�� |
���������в���ȷ���ǣ�������
| A���嵥����ǿ�����ԣ��ж������Կ���������Ѭ������ɱ���� |
| B��Һ��Ӧ������ĥ�ڲ�������ɫϸ���Լ�ƿ�У���������ˮ����ˮ�� |
| C���������ھƾ����������þƾ��ӵ�ˮ�н�����ȡ���� |
| D���ü��ȵķ����ɽ����л��е�����ɳ�ӳ�ȥ |