��Ŀ����
14�����ռ�������ˮ�����ξ��ƣ���һ�ξ�����Ҫ���ó�������ȥ����ˮ��Ca2+��Mg2+��Fe3+��SO42-�����ӣ��������£��������ˮ�м������BaCl2��Һ�����ˣ�
����������Һ�м������Na2CO3��Һ�����ˣ�
����Һ���������pH�����һ�ξ�����ˮ��
��1�����̢��ȥ��������SO42-����Ӧ�����ӷ���ʽ��Ba2++SO42-�TBaSO4����
��2�����̢����ɵIJ��ֳ��������ܽ�ȣ�20��/g�������
| CaSO4 | Mg2��OH��2CO3 | CaCO3 | BaSO4 | BaCO3 |
| 2.6��10-2 | 2.5��10-4 | 7.8��10-4 | 2.4��10-4 | 1.7��10-3 |
�ڳ�ȥMg2+�����ӷ���ʽ��2Mg2++2CO32-+H2O�TMg2��OH��2CO3��+CO2��
��3���ڶ��ξ���Ҫ��ȥ����I-��IO3-��NH4+��Ca2+��Mg2+������ʾ����ͼ��
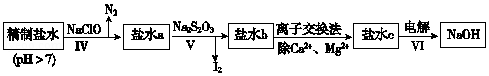
���̢���ȥ��������NH4+��I-��
���� ��1�������Ȼ����ܹ�����������ӷ�Ӧ�������ᱵ���н��
��2���ٸ��ݱ������ݷ���BaSO4��CaSO4���ܽ�ȴ�С���ܽ��ԽС������Խ��ȫ��
��þ�����ܹ���̼������ӷ�Ӧ����Mg2��OH��2CO3�������Ӷ���ȥþ�������ʣ��ݴ�д����Ӧ�����ӷ���ʽ��
��3������������Ӿ���ǿ�����ԣ��ܹ�����笠����Ӻ͵����ӣ�
��� �⣺��1�������ˮ�м������BaCl2��Һ��������������Ȼ�����Һ��Ӧ�������ᱵ������ͨ�����˳�ȥ���ᱵ���Ӷ���ȥ����SO42-�����ӷ�Ӧ����ʽΪ��Ba2++SO42-�TBaSO4����
�ʴ�Ϊ��SO42-��Ba2++SO42-�TBaSO4����
��2�������ᱵ���ܶȻ�Ϊ2.4��10-4������Ƶ��ܶȻ�Ϊ2.6��10-2����Ȼ����Ƶ��ܶȻ��������ᱵ����BaSO4���ܽ�ȱ�CaSO4��С���ɽ�SO42-�������꣬
�ʴ�Ϊ��BaSO4���ܽ�ȱ�CaSO4��С���ɽ�SO42-�������ꣻ
��þ�����ܹ���̼������ӷ�Ӧ����������Mg2��OH��2CO3����Ӧ�����ӷ���ʽΪ��2Mg2++2CO32-+H2O�TMg2��OH��2CO3��+CO2����
�ʴ�Ϊ��2Mg2++2CO32-+H2O�TMg2��OH��2CO3��+CO2����
��3�����̢�����������ƣ�������������ܹ�����NH4+��I-���Ӷ���ȥ����NH4+��I-���ʴ�Ϊ��NH4+��I-��
���� ��Ŀ�����˴��ε��ᴿ�����ܵ���ʵij���ƽ�⼰ת�������ԭ�������ӷ���ʽ����д��֪ʶ����Ŀ�Ѷ��еȣ������漰�������ϴ�֪ʶ��϶࣬ע�����մ��ε��ᴿ���������ʵķ������ᴿԭ���ܹ���ȷ��д���ӷ���ʽ��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ��Al��OH��3����θ����� | B�� | ����ʯ���к����������� | ||
| C�� | �ý������������ȷ����� | D�� | ��ˮ�����Ʊ��轺��ľ�ķ���� |
| A�� | �����Ǻ��� | B�� | ���ۺ���ά�� | ||
| C�� | Ӳ֬�����֬�� | D�� | �ʰ������������ |
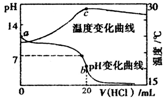
| A�� | a����ˮ�������c��H+����1.0��10-14mol•L-1 | |
| B�� | b��ʱ����c��NH4+��+c��NH3•H2O��=c��C1-�� | |
| C�� | c���Ժ���Һ�¶��½���ԭ��������ij������� | |
| D�� | �����������п��ܻ���ֵ�����Ũ�ȹ�ϵ��c��NH4+��=c��C1-����c��H+��=c��OH-�� |
��1���÷�Ӧ�У�Fe3+���������ԣ�
��2�������ܷ�Ӧ��ԭ����ͼ1��ʾ�������ĵ缫��Ӧʽ��CuFeS2-4e-�TFe2++2S+Cu2+��
��3��һ���¶��£����ƽ�ȡ��pH=1��ȡ������ͬ������ͭ���ĩ�ֱ��������ʵ�飺
| ʵ�� | ���� | 2Сʱ��Cu2+������/% |
| I | ��������0.10mol•L-1 Fe2��SO4��3��Һ | 78.2 |
| II | ��������0.10mol•L-1 Fe2��SO4��3��Һ��ͨ����� | 90.8 |
| III | ��������0.10mol•L-1 Fe2��SO4��3��Һ���ټ�������0.0005mol•L-1 Ag2SO4��Һ | 98.0 |
����ʵ�� III�Ʋ⣬�ڽ�ȡCu2+������Ag+����������ԭ���ǣ�
����CuFeS2+4Ag+�TFe2++Cu2++2Ag2S
����Ag2S+2Fe3+�T2Ag++2Fe2++S
Ϊ֤���ô�ԭ������������ʵ�飺
a��ȡ������ͭ���ĩ����������0.0005mol•L-1 Ag2SO4��Һ����ֻ�Ϻ��ã�ȡ�ϲ���Һ������ϡ���ᣬ�۲쵽��Һ������������֤��������Ӧ i��
b��ȡ����Ag2S��ĩ������pH=1��0.10mol•L-1Fe2��SO4��3��Һ��Һ����ֻ�Ϻ��ã�ȡ�ϲ���Һ������ϡ���ᣬ�а�ɫ������֤��������Ӧ ii��
��4����ʵ�� II�Ľ�ȡҺ�����ȡͭ��ԭ����ͼ2��ʾ��
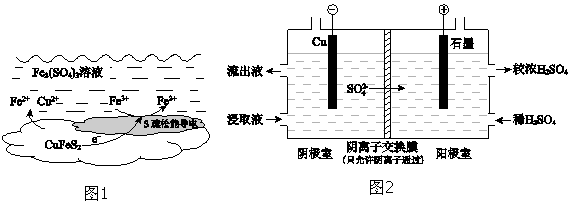
�ٵ����ڣ�����û��ͭ�������õ缫��Ӧʽ����ԭ����Fe3++e-�TFe2+��
�ڽ������ҵ�����Һ���������ң���ʹ��ȡ��������������ԭ����Fe2+������ʧ��������Fe3+��Fe2+-e-�TFe3+��SO42-ͨ�������ӽ���Ĥ���������ң�Fe2��SO4��3��Һ������
| A�� | �� 12C ���������� | B�� | �� 12C ��ѧ�������� | ||
| C�� | �� 12C ��Ϊͬ�������� | D�� | ����������������� |
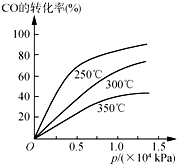 ��ҵ��һ�����ܱ������в������з�Ӧ�ϳɼ״���
��ҵ��һ�����ܱ������в������з�Ӧ�ϳɼ״���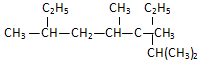 ����2��3��4��6-�ļ�-3-�һ����飮
����2��3��4��6-�ļ�-3-�һ����飮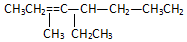 ���ƣ�2-��-3-�һ�-1-��ϩ��
���ƣ�2-��-3-�һ�-1-��ϩ��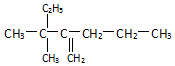 ���ƣ�3��3-����-2-����-1-��ϩ��
���ƣ�3��3-����-2-����-1-��ϩ��