��Ŀ����
11���״�����Ϊ21���͵�����ȼ�ϣ���ҵ����CH4��H2OΪԭ�ϣ�ͨ�����з�ӦI��II���Ʊ��״�����CH4��g��+H2O��g��?CO��g��+3H2��g����H1
��CO��g��+2H2��g��?CH3OH��g����H2��0
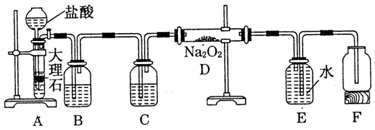
��1����1.0mol CH4��2.0mol H2O��g��ͨ�뷴Ӧ�ң��ݻ�Ϊ100L������һ�������·�����ӦI�����CH4��ת�������¶ȡ�ѹǿ�Ĺ�ϵ��ͼ1��
�ټ���100��ʱ�ﵽƽ�������ʱ��Ϊ5min������H2��ʾ��ƽ����Ӧ����Ϊ0.03 mol•L-1•min-1��
�ڷ�ӦI�ġ�H1����0 ������ڡ�����С�ڡ����ڡ�����100��ʱ��ӦI��ƽ�ⳣ��Ϊ2.25��10-2��
��2����ѹǿΪ0.1MPa���¶�Ϊ300�������£���a mol CO��3a mol H2�Ļ�������ڴ��������·�����ӦII���ɼ״�ƽ����������ݻ�ѹ����ԭ����$\frac{1}{2}$�������������䣬��ƽ����ϵ������Ӱ����CD ������ĸ��ţ���
A��c��H2������
B������Ӧ���ʼӿ죬�淴Ӧ���ʼ���
C������ƽ���CH3OH �����ʵ�������
D������ƽ���$\frac{c��{H}_{2}��}{c��C{H}_{3}OH��}$��С
��3����֪�ڳ��³�ѹ�£�
��2CH3OH ��l��+3O2��g��=2CO2��g��+4H2O��g����H=-1272.8kJ•mol-1
��2CO��g��+O2��g��=2CO2��g����H=-552.6kJ•mol-1
��H2O��g��=H2O��l����H=-44.0kJ•mol-1
д���״�����ȫȼ������һ����̼��Һ̬ˮ���Ȼ�ѧ����ʽCH3OH��l��+O2��g��=CO��g��+2H2O��l����H=-448.1kJ•mol-1��
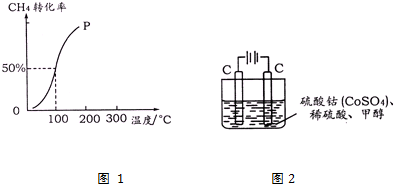
��4���״���ˮ�ʻ����һ������Ⱦ����һ�ֵ绯ѧ��������������Ⱦ����ԭ���ǣ�ͨ���Co2+ת����Co3+��Ȼ����Co3+����������ˮ�еļ״�������CO2��������
ʵ������ͼ2װ��ģ���������̣�
��д��������ӦʽCo2+-e-=Co3+��
����Co3+�����״��ķ�Ӧ�У����ɣ�����ġ������ɡ���H+��
���� ��1��100��ʱ�ﵽƽ��ʱ������ת����Ϊ50%����ϻ�ѧƽ������ʽ��ʽ����ƽ��ʱ����ֵ����ʵ�����
�ٸ���c=$\frac{\frac{��n}{V}}{��t}$����v��H2����
�ڸ���ƽ��״̬���¶ȱ仯ʱ��ƽ���ƶ��ķ����жϡ�H1������ƽ��״̬ʱ�����ʵ�Ũ�ȣ�����ƽ�ⳣ������ʽ���㣻
��1�������������䣬ƽ����������ݻ�ѹ����ԭ����$\frac{1}{2}$��ѹǿ���������淴Ӧ���ʶ���������Ӧ����������࣬ƽ��������Ӧ�����ƶ����������Ũ�����������¶Ȳ��䣬��ƽ�ⳣ�����䣬���ƽ�ⳣ����֪��ƽ��ʱ��Ӧ�����ֵ�Ũ�ȶ����ݴ˷������
��3�������Ȼ�ѧ����ʽ���ø�˹���ɼ��㷴Ӧ�Ȳ���д�Ȼ�ѧ����ʽ��
��4����ͨ���Co2+������Co3+������������ʧ���ӷ���������Ӧ���缫��ӦΪCo2+-e-=Co3+��
����Co3+����������ˮ�еļ״�������CO2����������������ԭΪCo2+��ԭ���غ������غ��֪����ԭ����H+����ƽ��дΪ��6Co3++CH3OH+H2O=CO2��+6Co2++6H+��
��� �⣺��1����1.0mol CH4��2.0mol H2O �� g ��ͨ���ݻ��̶�Ϊ10L�ķ�Ӧ�ң���һ�������·�����ӦI����ͼ���֪100�����ת����Ϊ50%���ʲμӷ�Ӧ�ļ���Ϊ1mol��50%=0.5mol����
CH4 ��g��+H2O ��g��=CO ��g��+3H2 ��g��
��ʼ����mol����1.0 2.0 0 0
�仯����mol����0.5 0.5 0.5 1.5
ƽ������mol����0.5 1.5 0.5 1.5
�ټ���100��ʱ�ﵽƽ�������ʱ��Ϊ5min������H2��ʾ�÷�Ӧ��ƽ����Ӧ����=$\frac{\frac{1.5mol}{10L}}{5min}$=0.03 mol•L-1•min-1��
�ʴ�Ϊ��0.03 mol•L-1•min-1��
���ɼ����ת�������¶ȱ仯�����֪���ﵽƽ��������¶ȣ������ת��������֪ƽ�������ƶ�������H1����0��100��ʱ��ӦI��ƽ��Ũ��Ϊc��CH4��=0.05mol/L��c��H2O��=0.15mol/L��c��CO��=0.05mol/L��c��H2��=0.15mol/L��
ƽ�ⳣ��K=$\frac{0.05��0.1{5}^{3}}{0.05��0.15}$=2.25��10-2��
�ʴ�Ϊ�����ڣ�2.25��10-2��
��2��A��ƽ����������ݻ�ѹ����ԭ����$\frac{1}{2}$��ѹǿ����ƽ��������Ӧ�����ƶ����������Ũ����������ƽ�ⳣ�����䣬���ƽ�ⳣ����֪��ƽ��ʱ��Ӧ�����ֵ�Ũ�ȶ�����A����
B��ѹǿ���������淴Ӧ���ʶ���������Ӧ����������࣬��B����
C��ѹǿ����ƽ��������Ӧ�����ƶ���CH3OH �����ʵ������ӣ���C��ȷ��
D��ѹǿ����ƽ��������Ӧ�����ƶ������������ʵ�����С���״������ʵ�����������ƽ��$\frac{c��{H}_{2}��}{c��C{H}_{3}OH��}$��С����D��ȷ��
�ʴ�Ϊ��CD��
��3����2CH3OH��l��+3O2��g���T2CO2��g��+4H2O��g����H=-1272.8kJ•mol-1
��2CO��g��+O2��g���T2CO2��g����H=-552.6kJ•mol-1
��H2O��g���TH2O��l����H=-44.0kJ•mol-1
���ݸ�˹���ɣ�����֪��Ӧ$\frac{1}{2}$������-��+�ۡ�4���õ�CH3OH��l��+O2��g��=CO��g��+2H2O��l�������Ը÷�Ӧ�ġ�H=$\frac{1}{2}$��[��-1272.8kJ/mol��-��-552.6kJ/mol��+��-44.0kJ/mol����4]=-448.1 kJ•mol-1����CH3OH��l��+O2��g��=CO��g��+2H2O��l����H=-448.1 kJ•mol-1��
�ʴ�Ϊ��CH3OH��l��+O2��g��=CO��g��+2H2O��l����H=-448.1 kJ•mol-1��
��4����ͨ���Co2+������Co3+������������ʧ���ӷ���������Ӧ���缫��ӦΪCo2+-e-=Co3+��
�ʴ�Ϊ��Co2+-e-=Co3+��
����Co3+����������ˮ�еļ״�������CO2����������������ԭΪCo2+�����ԭ���غ������غ��֪�����ӷ���ʽΪ��6Co3++CH3OH+H2O=CO2��+6Co2++6H+��˵����ԭ����H+���ʴ�Ϊ�����ɣ�
���� ���⿼���Ȼ�ѧ����ʽ��д����ѧƽ��ͼ��Ӱ�컯ѧƽ�����ء�ƽ�ⳣ���뷴Ӧ���ʼ��㡢ԭ��صȣ����ջ����ǽⱾ��Ĺؼ�����Ŀ�Ѷ��еȣ�

 һ����ʦ�����Ծ�ϵ�д�
һ����ʦ�����Ծ�ϵ�д� �����Ծ���Ԫ���Ծ�ϵ�д�
�����Ծ���Ԫ���Ծ�ϵ�д�| A�� | 2���SO2������O2��Ӧ��һ����������2���SO3 | |
| B�� | �����������䣬����ѹǿ������Ӧ���������淴Ӧ����Ҳ���� | |
| C�� | ����2molSO2��1molO2�ų��������Ǽ���1molSO2��0.5molO2�ų�������2�� | |
| D�� | �ڸ���������Ͷ��1molSO3��ƽ����ټ���1molSO3������ƽ���SO2�����������ԭƽ���С |
| A�� | ����Ӧ���Ũ�ȣ�������λ����ڻ���ӵİٷ������Ӷ�ʹ��Ч��ײ�������� | |
| B�� | ������μӵĻ�ѧ��Ӧ��������ѹǿ������С��Ӧ������������������ӻ���ӵİٷ������Ӷ�ʹ��Ӧ�������� | |
| C�� | �����¶���ʹ��ѧ��Ӧ����������Ҫԭ���������˷�Ӧ������л���ӵİٷ��� | |
| D�� | ������Ȼ���μӻ�ѧ��Ӧ����������λ����ڻ���ӵ���Ŀ���Ӷ�����Ӧ���� |
 CO2��һ���������壬�ݿ�ѧ��Ԥ�⣬��21������Ҷ��ȫ�����½�����1.5-4.5�棬�������µ���������ƽ�����ߣ�����������滷���������Ӱ�죮��κ���������CO2�ǰ��ڿ�ѧ����ǰ��һ���ش���⣮�ش��������⣺
CO2��һ���������壬�ݿ�ѧ��Ԥ�⣬��21������Ҷ��ȫ�����½�����1.5-4.5�棬�������µ���������ƽ�����ߣ�����������滷���������Ӱ�죮��κ���������CO2�ǰ��ڿ�ѧ����ǰ��һ���ش���⣮�ش��������⣺��1����ҵ�����ø��¡���ѹ����������CO2��NH3�ϳ�����[CO��NH2��2]���÷�Ӧ�Ļ�ѧ����ʽΪCO2+NH3 $\frac{\underline{\;���¸�ѹ\;}}{\;}$CO��NH2��2+H2O��
��2��һ�������£���ͬ����CO2�벻ͬ����NaOH��ַ�Ӧ�ų����Ⱦ����±���ʾ��
| CO2���� | NaOH��Һ���� | �ų������� | |
| �� | 22.0g | 750mL1.0mol•L-1 | xkJ |
| �� | 1.0mol | 2.0mL1.0mol•L-1 | ykJ |
��3����һ���¶Ⱥʹ��������£��ɽ�CO2ת��Ϊȼ��CH4����Ӧ����ʽΪCO2��g��+4H2��g��?CH4��g��+2H2O��g����H�� 300��ʱ��һ������CO2��H2����������ݻ�Ϊ2L�ĺ����ܱ���
���з���������Ӧ��5min��ﵽƽ�⣬��ʱ�����ʵ�Ũ�����±���
| ���� | CO2��g�� | H2��g�� | CH4��g�� | H2O��g�� |
| Ũ��/mol•L-1 | 0.2 | 0.8 | a | 1.6 |
��4��CO2�������������״���һ�������£�������ӦCO2 ��g��+3H��g��?CH3OH��g��+H2O��g����H�������ݻ�Ϊ2L�ĺ����ܱ������У�ͨ��2molCO2��3mol H2����������Ӧ����ա̼���ܹ������ÿ��淴Ӧ�ﵽƽ��״̬����de������ĸ����
a������3mol H2��g��ʱ����lmol CH3OH��g������
b��ת��3mol����ʱ����Ӧ��CO2Ϊ11.2L����״����
c����ϵ��������ܶȲ���
d��ˮ����������������ֲ���
e����λʱ��������H2��g��������H2O��g�������ʵ���֮��Ϊ3��1
���ö��ʯī���缫��30% KOH��Һ���������Һ���������ͼ����ʾ�ļ״�ȼ�ϵ�أ��õ�صĸ�����ӦʽΪCH3OH-6e-+8OH-�TCO32-+6H2O��������ȼ�ϵ�����ⱥ��ʳ��ˮ��װ�ý��д�������ͼ��������0.12mol���ӷ���ת��ʱ���Ͽ���Դ������Һ��ȴ�����£����ʳ����ҺΪ120mL�����ʱ��װ������Һ��pH=14����ʳ��ˮ����������NaCl����Cl2��ȫ�ݳ�����
| �¶ȡ� | 700 | 800 | 880 | 1000 | 1200 |
| ƽ�ⳣ�� | 1.0 | 10.0 | 15.0 | 16.1 | 17.7 |
��2��800��ʱ����һ��5L���ܱ������г���0.4molA��0.7molB������Ӧ��ʼ2mim��A��ƽ����Ӧ����Ϊ0.01mol•L-1•min-1����2minʱc��A��=0.06mol•L-1��C�����ʵ���Ϊ0.1mol����ʱ���ÿ��淴Ӧ�Ƿ�ﵽƽ�⣿����ǡ���
��3���ڴ��ܱ������У�����ѡ������Ϊ�жϸ÷�Ӧ�ﵽƽ���������abd
a��ѹǿ����ʱ��ı� b��������ܶȲ���ʱ��ı�
c����λʱ��������A��B�����ʵ������ d��C�İٷֺ������ֲ���
��4��880��ʱ����Ӧ C��g��+D��s��?A��g��+B��g����ƽ�ⳣ����ֵΪ0.067��
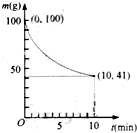 �����������������ͻ�ѧ���ԣ����������������Ǹ�����ҵ����Ҫԭ�ϣ��ʻ����ᴿ�����漰��������Ӧ����Ϊ��
�����������������ͻ�ѧ���ԣ����������������Ǹ�����ҵ����Ҫԭ�ϣ��ʻ����ᴿ�����漰��������Ӧ����Ϊ�� �������ѧ֪ʶ�ش��������⣺
�������ѧ֪ʶ�ش��������⣺