��Ŀ����
| |||||||||||||||||||
������
(1) |
H+��OH?/SUP>��H2O(1��)��NH4+��OH?IMG SRC="http://thumb.zyjl.cn/pic7/pages/62A2/0165/0027/90857e727a917aaedc3927fdaceaefdd/C/Image34.gif" width=37 HEIGHT=3>NH3����H2O(1��) |
(2) |
3.7gΪ������Ʒ����7.4g�� |
(3) |
���������ݼ��㣬��n[(NH4)2SO4]��n(NH4HSO4)�ֱ�Ϊx��y����������
�ʣ�n[(NH4)2SO4]��n(NH4HSO4)��0.0125��0.05��1��4(4��) |
(4) |
ӦѡNaOH����ȫ��Ӧ�ĵڢ���(1��)����(3)�����ݣ�22.2g��Ʒ�й����� n(H+)��n(NH4HSO4)��0.05�� 0.15mol����NH4+��OH?IMG SRC="http://thumb.zyjl.cn/pic7/pages/62A2/0165/0027/90857e727a917aaedc3927fdaceaefdd/C/Image39.gif" width=50 HEIGHT=9>NH3����H2O֪����1.12LNH3����NaOH ��NaOH��Һ�����ʵ���Ũ��Ϊ��
|

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
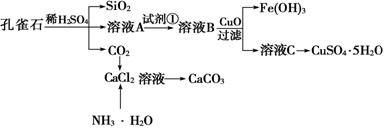
Сѧ��10����Ӧ����ϵ�д�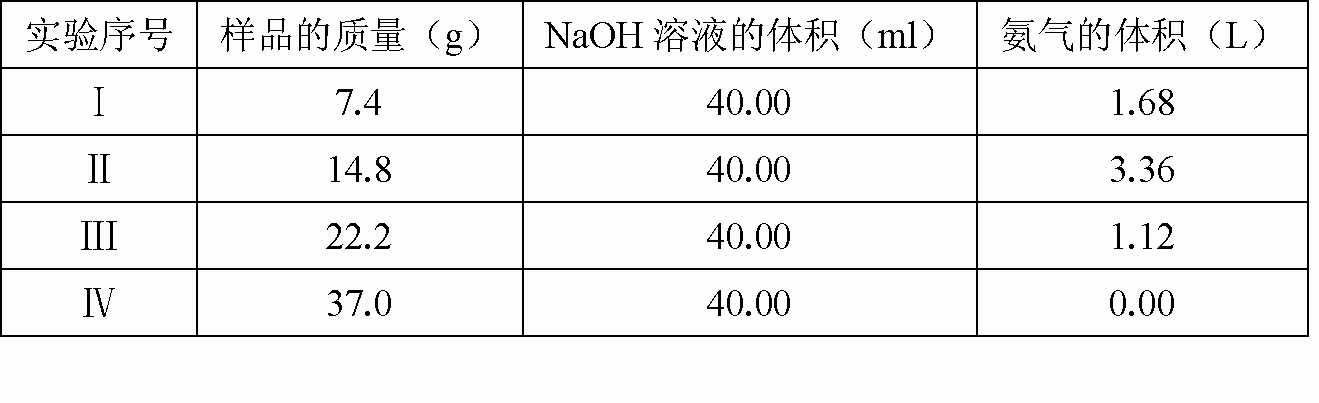


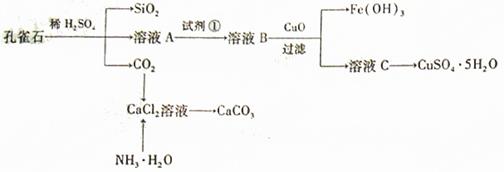
 ��
�� 


 ��
��