��Ŀ����
��1����֪������ˮ�ķ�Ӧ�ǿ��淴Ӧ���������ֻ�ѧ�����������ˮ�м���NaHCO3�������ˮ��ɱ����Ư��������ǿ��ԭ�� ��
��2���ҹ�����������ȷ��������ö����������������������ˮ��������
��ClO2�е�ClԪ�صĻ��ϼ�Ϊ+4�ۣ��Ʋ�÷��ӵĿռ乹��Ϊ �ͣ��÷���Ϊ ���ӣ�����ԡ��Ǽ��ԡ�����
��ClO2���ȶ�������NaOH��Һ��H2O2��Ӧ��ת��Ϊ�Ƚ��ȶ����������ƣ�NaClO2�����÷�Ӧ�Ļ�ѧ����ʽΪ ��
����ClO2������������ˮ�л����ClO2������FeCl2���仹ԭΪCl-��Fe2+ת��ΪFe��OH��3������V L���ѻ���Ϊ��״����ClO2����һ��������ˮ���ټ���a mol FeCl2�ɽ�������ClO2ǡ�ó�ȥ���������ˮ�в���ClO2�����ʵ�����Ϊ g��
��2���ҹ�����������ȷ��������ö����������������������ˮ��������
��ClO2�е�ClԪ�صĻ��ϼ�Ϊ+4�ۣ��Ʋ�÷��ӵĿռ乹��Ϊ
��ClO2���ȶ�������NaOH��Һ��H2O2��Ӧ��ת��Ϊ�Ƚ��ȶ����������ƣ�NaClO2�����÷�Ӧ�Ļ�ѧ����ʽΪ
����ClO2������������ˮ�л����ClO2������FeCl2���仹ԭΪCl-��Fe2+ת��ΪFe��OH��3������V L���ѻ���Ϊ��״����ClO2����һ��������ˮ���ټ���a mol FeCl2�ɽ�������ClO2ǡ�ó�ȥ���������ˮ�в���ClO2�����ʵ�����Ϊ
���㣺�����Ļ�ѧ����,�ȡ��塢�⼰�仯������ۺ�Ӧ��
ר�⣺±��Ԫ��
��������1��������ˮ��Ӧ��������ʹ����ᣬ�÷�ӦΪ���淴Ӧ������Ӱ��ƽ���ƶ������ؽ��
��2����Clԭ����sp2�ӻ�����γɦҼ�������ΪV�η��ӣ��ṹ�Գ�������������غϵ����ڷǼ��Է��ӣ���ϲ��Գ�������ɲ��غϵ����ڼ��Է��ӣ�
��ClO2��NaOH��Һ��H2O2��Ӧ��ת��Ϊ�Ƚ��ȶ����������ƣ�NaClO2�������У�Cl�Ļ��ϼ۴�+4�۽�Ϊ+3�ۣ�����˫��ˮ�ǻ�ԭ���������к�����������Ԫ�صĻ��ϼ۴�-1�۽�Ϊ-2�ۣ����ݵ�ʧ�����غ���ƽ����ʽ��
�۸���Ԫ�ػ��ϼ۱仯��ϵ����غ������㣮
��2����Clԭ����sp2�ӻ�����γɦҼ�������ΪV�η��ӣ��ṹ�Գ�������������غϵ����ڷǼ��Է��ӣ���ϲ��Գ�������ɲ��غϵ����ڼ��Է��ӣ�
��ClO2��NaOH��Һ��H2O2��Ӧ��ת��Ϊ�Ƚ��ȶ����������ƣ�NaClO2�������У�Cl�Ļ��ϼ۴�+4�۽�Ϊ+3�ۣ�����˫��ˮ�ǻ�ԭ���������к�����������Ԫ�صĻ��ϼ۴�-1�۽�Ϊ-2�ۣ����ݵ�ʧ�����غ���ƽ����ʽ��
�۸���Ԫ�ػ��ϼ۱仯��ϵ����غ������㣮
���
�⣺��1��������ˮ��Ӧ��������ʹ����Cl2+H2O HCl+HClO���÷�ӦΪ���淴Ӧ������NaHCO3��������������ӣ�����������Ũ�ȣ�ƽ���������ƶ���HClOŨ��������ˮ��ɱ����Ư��������ǿ��
�ʴ�Ϊ��������ˮ��Ӧ����HCl��HClO������NaHCO3�������H+��Ũ�ȣ�ƽ�������� ����HClOŨ������
��2����Clԭ����sp2�ӻ�����γɦҼ�������ΪV�η��ӣ�������ɲ��غϣ����ڼ��Է��ӣ��ʴ�Ϊ��V�����ԣ�
��ClO2��NaOH��Һ��H2O2��Ӧ��ת��Ϊ�Ƚ��ȶ����������ƣ�NaClO2�������У�Cl�Ļ��ϼ۴�+4�۽�Ϊ+3�ۣ�����˫��ˮ�ǻ�ԭ���������к�����������Ԫ�صĻ��ϼ۴�-1�۽�Ϊ-2�ۣ����ݵ�ʧ�����غ㡢ԭ�Ӹ����غ�÷���ʽΪ��2ClO2+2NaOH+H2O2=2NaClO2+O2��+2H2O��
�ʴ�Ϊ��2ClO2+2NaOH+H2O2=2NaClO2+O2��+2H2O��
�۸���ClO2�ܱ�FeCl2��ԭΪCl-��ClԪ�ػ��ϼ۽�����5�ۣ�FeCl2������ΪFeCl3��FeԪ�ػ��ϼ�������1�ۣ��裺������ˮ�в���ClO2�����ʵ���Ϊx��
���ݵ����غ㣬��a��3-2��=5x�����x=0.2amol��ClO2������m=nM=0.2amol��67.5g/mol=13.5ag��
�ʴ�Ϊ��13.5a��
�ʴ�Ϊ��������ˮ��Ӧ����HCl��HClO������NaHCO3�������H+��Ũ�ȣ�ƽ�������� ����HClOŨ������
��2����Clԭ����sp2�ӻ�����γɦҼ�������ΪV�η��ӣ�������ɲ��غϣ����ڼ��Է��ӣ��ʴ�Ϊ��V�����ԣ�
��ClO2��NaOH��Һ��H2O2��Ӧ��ת��Ϊ�Ƚ��ȶ����������ƣ�NaClO2�������У�Cl�Ļ��ϼ۴�+4�۽�Ϊ+3�ۣ�����˫��ˮ�ǻ�ԭ���������к�����������Ԫ�صĻ��ϼ۴�-1�۽�Ϊ-2�ۣ����ݵ�ʧ�����غ㡢ԭ�Ӹ����غ�÷���ʽΪ��2ClO2+2NaOH+H2O2=2NaClO2+O2��+2H2O��
�ʴ�Ϊ��2ClO2+2NaOH+H2O2=2NaClO2+O2��+2H2O��
�۸���ClO2�ܱ�FeCl2��ԭΪCl-��ClԪ�ػ��ϼ۽�����5�ۣ�FeCl2������ΪFeCl3��FeԪ�ػ��ϼ�������1�ۣ��裺������ˮ�в���ClO2�����ʵ���Ϊx��
���ݵ����غ㣬��a��3-2��=5x�����x=0.2amol��ClO2������m=nM=0.2amol��67.5g/mol=13.5ag��
�ʴ�Ϊ��13.5a��
���������⿼�������������ʣ���Ŀ�Ѷ��еȣ���Ϥ������ˮ���Ӧ�����ʡ�������ԭ��Ӧ����ʽ��ƽ�ķ����ǽ���ؼ���

��ϰ��ϵ�д�
 ÿ��10���ӿ�����������������ϵ�д�
ÿ��10���ӿ�����������������ϵ�д�
�����Ŀ
�����£���0.1mol?L-1��������Һ����μ������ʵ���Ũ����ͬ������������Һ��װ����ͼ1�������ɳ����������������������Һ�������ϵ��ͼ2��ʾ��a��b��c��d�ֱ��ʾʵ��IJ�ͬ�Σ������й�˵���в���ȷ���ǣ�������
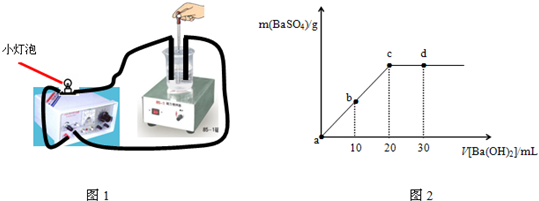

| A��ʵ�������С���������䰵ֱ��Ϩ�𣬺������� |
| B��ʵ���з�Ӧ���������������������ʺ��ѵ����������� |
| C����Һ��pHֵ��a��b��c��d |
| D��С���ݵ����ȣ�a��b=d��c |
��0.2mol/L�Ĵ�������Һ10mL��0.1mol/L����10mL��Ϻ���Һ�����ԣ�����Һ���й�����Ũ�ȹ�ϵ��ȷ���ǣ�������
| A��c��Na+��+c��H+���Tc��CH3COO-��+c��OH-�� |
| B��c��CH3COOH����c��Cl-����c��CH3COO-����c��H+�� |
| C��c��CH3COO-����c��Cl-����c��CH3COOH����c��H+�� |
| D��c��CH3COO-���Tc��Cl-���Tc��CH3COOH����c��H+�� |


 ����Ϊ
����Ϊ ����Ϊ
����Ϊ ����Ϊ
����Ϊ