��Ŀ����
��11�֣�A��B��C��D��E��F��G��H����I����ѧ��ѧ�г��������壬���Ǿ��ɶ�����Ԫ����ɣ������������ʣ�
��A��B��E��F��G��ʹʪ�����ɫʯ����ֽ��죬I��ʹʪ��ĺ�ɫʯ����ֽ������C��D��H����ʹ ʪ���ʯ����ֽ��ɫ��
��A��I������������
��B��E����ʹƷ����Һ��ɫ
�ܽ����ȵ�ͭ˿����װ��B��ƿ�У�ƿ�ڳ����ػ�ɫ����
�� ����ȼ��þ������װ��F��ƿ�У�þ������ȼ�գ����ɰ�ɫ��ĩ��ƿ�ڱڸ��ź�ɫ������
�� C��D�������ɺ���ɫ���壻
�� G��D��ȼ�տ��Բ���E��H2O
�� ��B��H ��ƿ�л�Ϻ��ڹ��������ü����ӣ�ƿ�ڱڳ�����״Һ�β�����A��
�ش��������⣺
��1��A�Ļ�ѧʽ�� �����а��̵Ļ�ѧʽ�� ��
��2�� ���з�����Ӧ�Ļ�ѧ����ʽ�� ��
��3�����з�����Ӧ�Ļ�ѧ����ʽ�� ��
��4��C�Ļ�ѧʽ�� ��D�ĵ���ʽ�� ��
��5�� ���з�����Ӧ�Ļ�ѧ����ʽ�� ��
��6�� H�Ľṹʽ��
����11�֣���1��HCl NH4Cl ��2��Cu+Cl2 CuCl2 ��3��2Mg+ CO2
CuCl2 ��3��2Mg+ CO2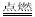 2MgO+ C
2MgO+ C
��4��NO 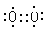 ��5��2H2S+ 3O2
��5��2H2S+ 3O2 2H2O+ 2SO2 ��6��
2H2O+ 2SO2 ��6��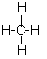
��������
�����������1����ʹʪ�����ɫʯ����ֽ��������Ϊ�������壬I��ʹʪ��ĺ�ɫʯ����ֽ������˵��I�ǰ���������A��HCl�������İ������Ȼ�淋Ĺ���С�����������̵Ļ�ѧʽ��NH4Cl��
��2�����ȵ�ͭ˿��������ȼ�ղ����ػ�ɫ���̣�����B����������ѧ����ʽΪCu+Cl2 CuCl2
CuCl2
��3��þ���ڶ�����̼��ȼ�ղ�����ɫ������þ���ɫ�ĵ���̼������F�Ƕ�����̼����ѧ����ʽΪ2Mg+ CO2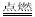 2MgO+ C��
2MgO+ C��
��4��NO��O2�������ɶ��������Ǻ���ɫ���壬��Ϣ�G��D��ȼ�գ�����D��������C��NO�������õ���ʽΪ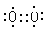 ��
��
��5��E��ʹƷ����Һ��ɫ����E��SO2, G��D��ȼ�տ��Բ���E��H2O,˵��G�к���HԪ�أ�����G��H2S�����з�����Ӧ�Ļ�ѧ����ʽ��2H2S+ 3O2 2H2O+ 2SO2
2H2O+ 2SO2
��6��B���������������������ڹ���������״Һ�γ��֣�����H�Ǽ��飬��ṹʽΪ ��
��
���㣺�����������ۺ�Ӧ��

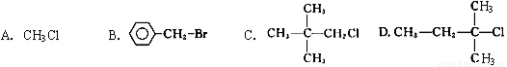
 ��Cu2����Ba2����Al3����Ag����Fe3���������ӿ�����Cl����NO
��Cu2����Ba2����Al3����Ag����Fe3���������ӿ�����Cl����NO ��SO
��SO ��CO
��CO ����֪��
����֪��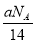
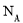 ��ʾ�����ӵ������������ж���ȷ����
��ʾ�����ӵ������������ж���ȷ���� 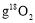 �к���
�к��� ����ԭ��
����ԭ�� 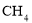 �к���4
���4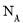 ��
��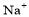 ��
��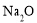 �ܽ���1Lˮ�У�
�ܽ���1Lˮ�У� �����ʵ���Ũ��Ϊ1 mol��L-1
�����ʵ���Ũ��Ϊ1 mol��L-1