��Ŀ����
�������岻��ȱ����Ԫ�أ�Ϊ�˷�ֹ��ȱ���������г�������һ�ּӵ��Σ������ھ���������һ������KIO3��ȥ��ij�о�С��Ϊ�˼��ij�ӵ������Ƿ��е⣬�������йص����ϣ���������ԭ���ǣ�KIO3+5KI+3H2SO4=3I2+3H2O+3K2SO4
��1���÷�Ӧ���������뻹ԭ������������
��2����ȡ�����ļӵ��μ�����ˮ�ܽ⣬Ȼ�����ϡ�����KI��Һ��������һ������CCl4������ʱ�۲쵽�������� ��
��3��ijѧ����һ�η�Һ�����з������²���Һ������ɫҺ�壬��֪����Һ©���е�Һ����һ�����л��㣬��һ����ˮ�㣬�����üķ����������������д���йز��輰�ж� ��
��4��������ƿ��ʹ�÷����У����в�����ȷ����
A��ʹ������ƿǰ�����Ƿ�©ˮ
B������ƿ��ˮϴ�������ô�����Һϴ��
C��������Һʱ����������Һ�壬����Ͳȡ�����ò�����������������ƿ�У�������ˮ���̶���1��2cm�����ý�ͷ�ιܼ�����ˮ���̶��ߣ�
D���Ǻ�ƿ������ʳָ��סƿ������һֻ����סƿ�ף�������ƿ������ת��Σ�ҡ�ȣ�
��5������ʵ�����õ�һ�����ʵ���Ũ�ȵ�ϡ���ᣬ������0.5mol/L��������Һ450mL��������Ͳ��ȡ��������Ϊ98%���ܶ�Ϊ1.84g/cm3��Ũ��������Ϊ mL�����ʵ������15mL��20mL��50mL��Ͳ��Ӧѡ�� mL��Ͳ��ã�
��6����������������ϡ������ҺŨ��ƫ�ߵ���
A���ܽ��ʱ����Һû����ȴ�����¾�ת��
B��ת��ʱû��ϴ���ձ���������
C��������ƿ��ˮ����ʱ�۾�����Һ�� D������Ͳ��ȡŨ�����ϴ����Ͳ����ϴ��Һת�Ƶ�����ƿ
E��ҡ�Ⱥ���Һ����ڿ̶��ߣ��ּ�����ˮ���̶��ߣ�
��1���÷�Ӧ���������뻹ԭ������������
��2����ȡ�����ļӵ��μ�����ˮ�ܽ⣬Ȼ�����ϡ�����KI��Һ��������һ������CCl4������ʱ�۲쵽��������
��3��ijѧ����һ�η�Һ�����з������²���Һ������ɫҺ�壬��֪����Һ©���е�Һ����һ�����л��㣬��һ����ˮ�㣬�����üķ����������������д���йز��輰�ж�
��4��������ƿ��ʹ�÷����У����в�����ȷ����
A��ʹ������ƿǰ�����Ƿ�©ˮ
B������ƿ��ˮϴ�������ô�����Һϴ��
C��������Һʱ����������Һ�壬����Ͳȡ�����ò�����������������ƿ�У�������ˮ���̶���1��2cm�����ý�ͷ�ιܼ�����ˮ���̶��ߣ�
D���Ǻ�ƿ������ʳָ��סƿ������һֻ����סƿ�ף�������ƿ������ת��Σ�ҡ�ȣ�
��5������ʵ�����õ�һ�����ʵ���Ũ�ȵ�ϡ���ᣬ������0.5mol/L��������Һ450mL��������Ͳ��ȡ��������Ϊ98%���ܶ�Ϊ1.84g/cm3��Ũ��������Ϊ
��6����������������ϡ������ҺŨ��ƫ�ߵ���
A���ܽ��ʱ����Һû����ȴ�����¾�ת��
B��ת��ʱû��ϴ���ձ���������
C��������ƿ��ˮ����ʱ�۾�����Һ�� D������Ͳ��ȡŨ�����ϴ����Ͳ����ϴ��Һת�Ƶ�����ƿ
E��ҡ�Ⱥ���Һ����ڿ̶��ߣ��ּ�����ˮ���̶��ߣ�
���㣺����һ�����ʵ���Ũ�ȵ���Һ,���ܼ��ȵ�������ʹ�÷���,���ʵļ���ͼ����ʵ�鷽�����
ר�⣺
��������1��KIO3+5KI+3H2SO4��3I2+3K2SO4+3H2O�У�IԪ�صĻ��ϼ���+5�۽���Ϊ0��IԪ�صĻ��ϼ���-1������Ϊ0�����ϼ�����ֵ=���ϼ۽���ֵ=ת�Ƶ����������ݻ��ϼ۱仯ȷ������ת�������
��2�����������л��ܼ��������Ȼ�̼�ڵ��ܽ��Զ������ˮ�У��������Ȼ�̼��ȡˮ�еĵ⣬���Ȼ�̼��ˮ�����ܣ���Һ�ֳ����㣬���Ȼ�̼���ܶȱ�ˮ���л������²㣬���������Ȼ�̼���Ϻ�ɫ���ϲ㼸����ɫ��
��3��������ȡ����ˮ�����ܽ�����ƣ��ӷ�Һ©���¿ڷų������²�Һ�����С�Թ��У������Թ��м�������ˮ�����Թ���Һ��ֲ㣬��֤���²����л��㣻�����ֲ㣬��֤���²���ˮ�㣻
��4����������ƿ��ʹ�÷�����ע���������ش����⣻
��5������ϡ��ǰ�����ʵ����������з��������㣬������Ͳ����ȡ������ѡ��
��6���������������ʵ����ʵ��������Һ�������Ӱ�죬����c=
������������ҺŨ�ȵ�Ӱ�죮
��2�����������л��ܼ��������Ȼ�̼�ڵ��ܽ��Զ������ˮ�У��������Ȼ�̼��ȡˮ�еĵ⣬���Ȼ�̼��ˮ�����ܣ���Һ�ֳ����㣬���Ȼ�̼���ܶȱ�ˮ���л������²㣬���������Ȼ�̼���Ϻ�ɫ���ϲ㼸����ɫ��
��3��������ȡ����ˮ�����ܽ�����ƣ��ӷ�Һ©���¿ڷų������²�Һ�����С�Թ��У������Թ��м�������ˮ�����Թ���Һ��ֲ㣬��֤���²����л��㣻�����ֲ㣬��֤���²���ˮ�㣻
��4����������ƿ��ʹ�÷�����ע���������ش����⣻
��5������ϡ��ǰ�����ʵ����������з��������㣬������Ͳ����ȡ������ѡ��
��6���������������ʵ����ʵ��������Һ�������Ӱ�죬����c=
| n |
| V |
���
�⣺��1��������ԭ��ӦKIO3+5KI+3H2SO4�T3K2SO4+3I2+3H2O�У�IԪ�صĻ��ϼ���+5�۽���Ϊ0��IԪ�صĻ��ϼ���-1������Ϊ0�����ϼ�����ֵ=���ϼ۽���ֵ=ת�Ƶ�����=5������ת��������£� ��KIΪ��ԭ����+5�۵�IԪ�ر���ԭ����ԭ���������������Ϊ�⣬��ԭ���غ㼰��Ӧ��֪�����ʵ���֮��Ϊ1��5���ʴ�Ϊ��5��1��
��KIΪ��ԭ����+5�۵�IԪ�ر���ԭ����ԭ���������������Ϊ�⣬��ԭ���غ㼰��Ӧ��֪�����ʵ���֮��Ϊ1��5���ʴ�Ϊ��5��1��
��2�����������л��ܼ��������Ȼ�̼�ڵ��ܽ��Զ������ˮ�У��������Ȼ�̼��ȡˮ�еĵ⣬���Ȼ�̼��ˮ�����ܣ���Һ�ֳ����㣬���Ȼ�̼���ܶȱ�ˮ���л������²㣬���������Ȼ�̼���Ϻ�ɫ���ϲ㼸����ɫ���ʴ�Ϊ����Һ�ֳ����㣬�ϲ���ɫ���²���ϣ��죩ɫ��
��3���ӷ�Һ©���¿ڷų������²�Һ�����С�Թ��У���������ˮ�����Թ���Һ��ֲ㣬��֤���²����л��㣻�����ֲ㣬��֤���²���ˮ�㣬
�ʴ�Ϊ���ӷ�Һ©���¿ڷų������²�Һ�����С�Թܣ������Թ��м�������ˮ�����Թ���Һ��ֲ㣬��֤���²����л��㣻�����ֲ㣬��֤���²���ˮ�㣻
��4��A��ʹ������ƿǰ������Ƿ�©ˮ������������Һ��Ũ��������A��ȷ��
B������ƿ������ˮϴ�������ô�����Һϴ�ӣ��������ʵ����ʵ���ƫ��������Һ��Ũ��ƫ�ߣ���B����
C������ƿֻ����������һ��Ũ�ȵ���Һ������������ƿ���ܽ����ʣ���C����
D���Ǻ�ƿ�ǣ���ʳָ��סƿ��������һֻ�ֵ���ָ��סƿ�ף�������ƿ��ת��ҡ����Σ�Ŀ����ҡ����Һ����D��ȷ��
�ʴ�Ϊ��AD��
��5����Ũ��������ΪVmL��ϡ��ǰ�����ʵ��������䣬��98%��1.84g/cm3V=0.5mol/L��0.50L��98g/mol�����V=13.6mL��
Ϊ��С��Ӧѡ��15mL����Ͳ��
�ʴ�Ϊ��13.6��15��
��6��A��δ��ȴ�����¾ͽ���Һת�Ƶ�����ƿ�����ݣ���Һ��ȴ�����ƫС�����Ƶ���ҺŨ��ƫ�ߣ���A��ȷ��
B��ת��ʱû��ϴ���ձ������������������Ƶ���Һ�����ʵ����ʵ���ƫС�����Ƶ���ҺŨ��ƫ�ͣ���B����
C������ʱ���ӿ̶��ߣ����¼��������ˮ���ƫС�����Ƶ���Һ���ƫС����ҺŨ��ƫ�ߣ���C��ȷ��
D��ϴ����Ͳ�����ʵ����ʵ���ƫ�����Ƶ���ҺŨ��ƫ�ߣ���D��ȷ��
E���ּ�����ˮ���̶��ߣ����Ƶ���Һ���ƫ����ҺŨ��ƫС����E����
�ʴ�Ϊ��ACD��
 ��KIΪ��ԭ����+5�۵�IԪ�ر���ԭ����ԭ���������������Ϊ�⣬��ԭ���غ㼰��Ӧ��֪�����ʵ���֮��Ϊ1��5���ʴ�Ϊ��5��1��
��KIΪ��ԭ����+5�۵�IԪ�ر���ԭ����ԭ���������������Ϊ�⣬��ԭ���غ㼰��Ӧ��֪�����ʵ���֮��Ϊ1��5���ʴ�Ϊ��5��1����2�����������л��ܼ��������Ȼ�̼�ڵ��ܽ��Զ������ˮ�У��������Ȼ�̼��ȡˮ�еĵ⣬���Ȼ�̼��ˮ�����ܣ���Һ�ֳ����㣬���Ȼ�̼���ܶȱ�ˮ���л������²㣬���������Ȼ�̼���Ϻ�ɫ���ϲ㼸����ɫ���ʴ�Ϊ����Һ�ֳ����㣬�ϲ���ɫ���²���ϣ��죩ɫ��
��3���ӷ�Һ©���¿ڷų������²�Һ�����С�Թ��У���������ˮ�����Թ���Һ��ֲ㣬��֤���²����л��㣻�����ֲ㣬��֤���²���ˮ�㣬
�ʴ�Ϊ���ӷ�Һ©���¿ڷų������²�Һ�����С�Թܣ������Թ��м�������ˮ�����Թ���Һ��ֲ㣬��֤���²����л��㣻�����ֲ㣬��֤���²���ˮ�㣻
��4��A��ʹ������ƿǰ������Ƿ�©ˮ������������Һ��Ũ��������A��ȷ��
B������ƿ������ˮϴ�������ô�����Һϴ�ӣ��������ʵ����ʵ���ƫ��������Һ��Ũ��ƫ�ߣ���B����
C������ƿֻ����������һ��Ũ�ȵ���Һ������������ƿ���ܽ����ʣ���C����
D���Ǻ�ƿ�ǣ���ʳָ��סƿ��������һֻ�ֵ���ָ��סƿ�ף�������ƿ��ת��ҡ����Σ�Ŀ����ҡ����Һ����D��ȷ��
�ʴ�Ϊ��AD��
��5����Ũ��������ΪVmL��ϡ��ǰ�����ʵ��������䣬��98%��1.84g/cm3V=0.5mol/L��0.50L��98g/mol�����V=13.6mL��
Ϊ��С��Ӧѡ��15mL����Ͳ��
�ʴ�Ϊ��13.6��15��
��6��A��δ��ȴ�����¾ͽ���Һת�Ƶ�����ƿ�����ݣ���Һ��ȴ�����ƫС�����Ƶ���ҺŨ��ƫ�ߣ���A��ȷ��
B��ת��ʱû��ϴ���ձ������������������Ƶ���Һ�����ʵ����ʵ���ƫС�����Ƶ���ҺŨ��ƫ�ͣ���B����
C������ʱ���ӿ̶��ߣ����¼��������ˮ���ƫС�����Ƶ���Һ���ƫС����ҺŨ��ƫ�ߣ���C��ȷ��
D��ϴ����Ͳ�����ʵ����ʵ���ƫ�����Ƶ���ҺŨ��ƫ�ߣ���D��ȷ��
E���ּ�����ˮ���̶��ߣ����Ƶ���Һ���ƫ����ҺŨ��ƫС����E����
�ʴ�Ϊ��ACD��
���������⿼����ȡ��������ԭ��Ӧ��һ�����ʵ���Ũ����Һ�������йؼ���ȣ��Ѷ��еȣ��Ƕ���ѧ֪ʶ���ۺ����ã���Ҫѧ���߱���ʵ�Ļ���֪ʶ���ۺ�����֪ʶ�������⡢��������������ѧϰ��ȫ����ջ���֪ʶ��

��ϰ��ϵ�д�
 ��ְٷְټ�����Ԫ��ĩ���Ծ�ϵ�д�
��ְٷְټ�����Ԫ��ĩ���Ծ�ϵ�д�
�����Ŀ
��10mL0.1mol?L-1NH4Al��SO4��2��Һ�У��μӵ�Ũ��Ba��OH��2��Һx mL������������ȷ���ǣ�������
| A��x=10ʱ����Һ����NH4+��Al3+��SO42-����c�� NH4+����c��Al3+�� |
| B��x=15ʱ����Һ����NH4+��SO42-����c��NH4+����2c��SO42-�� |
| C��x=20ʱ����Һ����NH4+��Al3+��OH-����c��OH-����c��Al3+�� |
| D��x=30ʱ����Һ����Ba2+��AlO2-��OH-����c��OH-��=c��Ba2+�� |
25��ʱ��ˮ�ĵ���ﵽƽ�⣬����������ȷ���ǣ�������
| A����ˮ�м�������NaOH��ƽ�������ƶ�����ƽ��ʱc��OH-������ |
| B����ˮ�м�������NH4Cl��ƽ�������ƶ�����ƽ��ʱc��OH-������ |
| C��pH=5�Ĵ����pH=9�İ�ˮ����ˮ�������c��OH-������� |
| D����ˮ���ȣ�Kw����pH���� |
�����ͬ��pH��ͬ������ʹ�����Һ����NaOH��Һ�к�ʱ��������NaOH�����ʵ�����������
| A������� | B������� |
| C��������ͬ | D�����Ƚ� |
 ��1����ͼ��ʾ���ܱ������з�Ӧ��2SO2+O2?2SO3+Q�ﵽƽ��ʱ�����������ı������Ӧ�ٶȺͻ�ѧƽ��ı仯�����a b�����иı������������
��1����ͼ��ʾ���ܱ������з�Ӧ��2SO2+O2?2SO3+Q�ﵽƽ��ʱ�����������ı������Ӧ�ٶȺͻ�ѧƽ��ı仯�����a b�����иı������������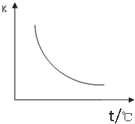 ��ҵ���Ʊ�H2��һ����Ҫ�����ǣ�CO�� g��+H2 O��g��?CO2��g��+H2��g����H=Q kJ/mol����֪�÷�Ӧ��ƽ�ⳣ�� K ���¶ȵĹ�ϵ��ͼ��ʾ������һ�̶����ܱ������У�850��ʱ����������Ӧ������������ڸ����� ��Ũ�ȣ�mol?L-1����ʱ��ı仯��ϵ�����
��ҵ���Ʊ�H2��һ����Ҫ�����ǣ�CO�� g��+H2 O��g��?CO2��g��+H2��g����H=Q kJ/mol����֪�÷�Ӧ��ƽ�ⳣ�� K ���¶ȵĹ�ϵ��ͼ��ʾ������һ�̶����ܱ������У�850��ʱ����������Ӧ������������ڸ����� ��Ũ�ȣ�mol?L-1����ʱ��ı仯��ϵ�����