��Ŀ����
��һ���������ʣ�Ϊ�˲ⶨ����ɣ��������²��ԣ���ͬ��ͬѹ�����£���������ܶ���H2�ܶȵ�43����
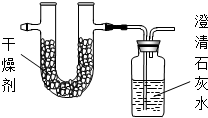
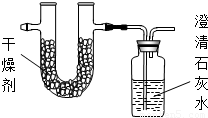
�ڽ�1.72�˸�����ȫȼ�գ���ʹ�����IJ���ȫ������ͨ����ͼװ�ã��õ��±����е�ʵ������������������������ȫ�����գ���
| ʵ��ǰ | ʵ��� | |
| �������+U�ܣ������� | 101.1g | 103.62g |
| ��ʯ��ˮ+���ƿ�������� | 312.0g | 317.28g |
��2��д���������п��ܵĽṹ��ʽ��
���𰸡���������1����������ܶ�֮�ȵ�����Է�������֮�ȼ����������Է�����������������1.72�˸���ȼ������ˮ�Ͷ�����̼���������ɼ��㻯ѧʽ��
��2����̼���칹�ĽǶ���д���ܵ�ͬ���칹��Ľṹ��ʽ��
����⣺��1����n=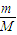 =
= ��֪����ܶ�֮�ȵ�����Է�������֮�ȣ�ȷ����������Է�������Ϊ2×43=86��
��֪����ܶ�֮�ȵ�����Է�������֮�ȣ�ȷ����������Է�������Ϊ2×43=86��
����1.72�˸��������ʵ���Ϊ =0.02mol��
=0.02mol��
�ɱ������ݿ�֪����������ص�����Ϊˮ����������n��H2O��= =0.14mol��n��H��=0.28mol��
=0.14mol��n��H��=0.28mol��
���ƿ���ص�����Ϊ������̼����������n��CO2��=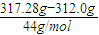 =0.12mol��
=0.12mol��
��1mol���к��� =14molHԭ�ӣ�����
=14molHԭ�ӣ����� =6molCԭ�ӣ�
=6molCԭ�ӣ�
���Է���ʽΪC6H14��
��ȷ����������Է�������Ϊ86�ͻ�ѧʽΪC6H14��
��2������Ϊ���飬���ܵĽṹ��ʽ�У�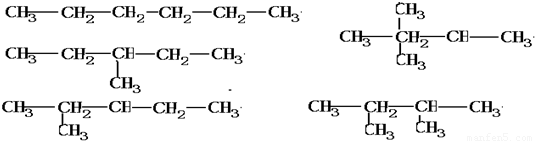 ��
��
�𣺿��ܵĽṹ��ʽ�У�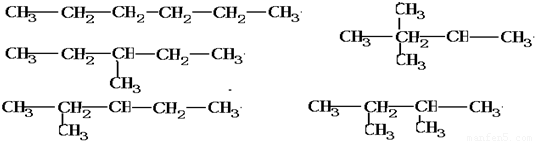 ��
��
���������⿼���л������ʽ��ȷ������Ŀ�ѶȲ���ע�⼺���ͬ���칹�����Ŀ���ṹ��ʽ��
��2����̼���칹�ĽǶ���д���ܵ�ͬ���칹��Ľṹ��ʽ��
����⣺��1����n=
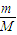 =
= ��֪����ܶ�֮�ȵ�����Է�������֮�ȣ�ȷ����������Է�������Ϊ2×43=86��
��֪����ܶ�֮�ȵ�����Է�������֮�ȣ�ȷ����������Է�������Ϊ2×43=86������1.72�˸��������ʵ���Ϊ
 =0.02mol��
=0.02mol���ɱ������ݿ�֪����������ص�����Ϊˮ����������n��H2O��=
 =0.14mol��n��H��=0.28mol��
=0.14mol��n��H��=0.28mol�����ƿ���ص�����Ϊ������̼����������n��CO2��=
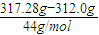 =0.12mol��
=0.12mol����1mol�����
 =14molHԭ�ӣ�����
=14molHԭ�ӣ����� =6molCԭ�ӣ�
=6molCԭ�ӣ����Է���ʽΪC6H14��
��ȷ����������Է�������Ϊ86�ͻ�ѧʽΪC6H14��
��2������Ϊ���飬���ܵĽṹ��ʽ�У�
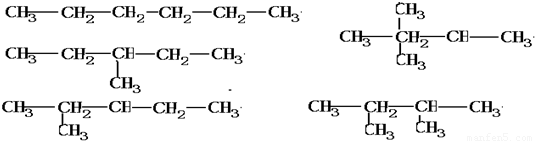 ��
���𣺿��ܵĽṹ��ʽ�У�
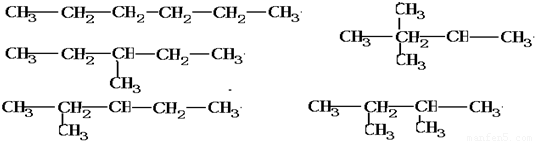 ��
�����������⿼���л������ʽ��ȷ������Ŀ�ѶȲ���ע�⼺���ͬ���칹�����Ŀ���ṹ��ʽ��

��ϰ��ϵ�д�
 �ƸԴ��ž�ϵ�д�
�ƸԴ��ž�ϵ�д�
�����Ŀ
 ��һ���������ʣ�Ϊ�˲ⶨ����ɣ��������²��ԣ�
��һ���������ʣ�Ϊ�˲ⶨ����ɣ��������²��ԣ�