��Ŀ����
��������Ԫ�أ�����A��B��C��D��EΪԭ����������������ԭ��������������36����������������Ϣ���ش����⣮
��1��AԪ�ص�һ��ͬ��������ʵ���״����ѧ������ij���Ϊ��ʯīϩ�����䲿�ֽṹ��ͼ�ף�ͼ��ÿ���������ε����ԼΪ5.24��10-20m2����12gʯīϩ�����ԼΪm2������3λ��Ч���֣�
��2��D�Ļ�̬ԭ�Ӻ���۲�����Ų�ʽ ��DCl3?6H2O�����ֲ�ͬ��ɫ��ͬ���칹�壬ȡ���е�һ�ְ���ɫ��ͬ���칹��0.0100mol�����Һ���μ�������������Һ��õ�2.87g���������ͬ���칹��ĽṹΪ ��A��[D��H2O��5]Cl3 B��[D��H2O��5Cl]Cl2?H2O C��[D��H2O��4 Cl2]Cl?2H2O
��3��CԪ��+5�۵�ԭ����-2�۵���ԭ���γ�һ��-1�۵�������X����ԭ�ӵ��ӻ���ʽΪ ��X�Ŀռ�ṹΪ ��
��4�����B�Լ�Bͬ������ԭ��������BС��ԭ�Ӱ���һ�����ܴӴ�С��˳�����У� ������Ӧ��Ԫ�ط��ű�ʾ����
��5������E������ԭ�ӵĶѻ���ʽ��ͼ����ʾ���侧��������ͼ����ʾ����Eԭ�Ӱ뾶Ϊacm���þ�����ܶ�Ϊ g��cm-3��ֻ�г�����ʽ������٤��������ֵΪNA����
| A ��̬ԭ����3���ܼ������ܼ��е�������� |
| B ��̬ԭ�Ӻ�����13�ֲ�ͬ�˶�״̬�ĵ��� |
| C��B����ͬһ���ڣ���ԭ���ڸ������е縺����� |
| D D4+�ĺ�������Ų����ԭ����ͬ |
| E ��ds��ԭ��������С��Ԫ�� |
��2��D�Ļ�̬ԭ�Ӻ���۲�����Ų�ʽ
��3��CԪ��+5�۵�ԭ����-2�۵���ԭ���γ�һ��-1�۵�������X����ԭ�ӵ��ӻ���ʽΪ

��4�����B�Լ�Bͬ������ԭ��������BС��ԭ�Ӱ���һ�����ܴӴ�С��˳�����У�
��5������E������ԭ�ӵĶѻ���ʽ��ͼ����ʾ���侧��������ͼ����ʾ����Eԭ�Ӱ뾶Ϊacm���þ�����ܶ�Ϊ
���㣺�����ļ���,Ԫ�ص����ܡ��縺�Եĺ��弰Ӧ��,ԭ�ӽṹ��Ԫ�������ɵĹ�ϵ
ר�⣺Ԫ����������Ԫ�����ڱ�ר��,��ѧ���뾧��ṹ
������A ��̬ԭ����3���ܼ������ܼ��е�������ȣ������Ų�Ϊ1s22s22p2��ΪCԪ�أ�B ��̬ԭ�Ӻ�����13�ֲ�ͬ�˶�״̬�ĵ��ӣ�ΪAlԪ�أ�C ��B����ͬһ���ڣ���ԭ���ڸ������е縺�����ΪClԪ�أ� D4+�ĺ�������Ų����ԭ����ͬ��DӦΪTi��E ��ds��ԭ��������С��Ԫ�أ�ΪCu����϶�Ӧ���ʵ������Լ���ĿҪ������⣮
���
�⣺A ��̬ԭ����3���ܼ������ܼ��е�������ȣ������Ų�Ϊ1s22s22p2��ΪCԪ�أ�B ��̬ԭ�Ӻ�����13�ֲ�ͬ�˶�״̬�ĵ��ӣ�ΪAlԪ�أ�C ��B����ͬһ���ڣ���ԭ���ڸ������е縺�����ΪClԪ�أ� D4+�ĺ�������Ų����ԭ����ͬ��DӦΪTi��E ��ds��ԭ��������С��Ԫ�أ�ΪCu��
��1�����þ�̯�����ÿ��������ʵ��ռ��̼ԭ�ӵĸ���Ϊ6��
=2�����ԣ�12gʯīϩ����1mol��ʵ��ռ�е������εĸ���Ϊ
���������ʯīϩ�����Ϊ
��6.02��1023��5.24��10-20m2=1.58��104m2���ʴ�Ϊ��1.58��104��
��2��DΪTi��λ�����ڱ��������ڡ���B�壬��̬ԭ�Ӻ���۲�����Ų�ʽΪ3d24s2��n��AgCl��=
=0.02mol����1molTiCl3?6H2O�ɵ����2molCl-��ֻ��B���ϣ�
�ʴ�Ϊ��3d24s2��B��
��3��ClԪ��+5�۵�ԭ����-2�۵���ԭ���γ�һ��-1�۵�������ΪClO3-��Cl�γ�3���ļ����µ��Ӷ���Ϊ
=1��ӦΪsp3�ӻ���Ϊ�����Σ�
�ʴ�Ϊ��sp3�������Σ�
��4��BΪAl��ͬ����ԭ��������AlС����Na��Mg��ͬ����Ԫ�ص�һ�����ܴ�������������Mg�����Ϊ���ͽṹ�����һ�����ܴ���Al������˳��ΪMg��Al��Na���ʴ�Ϊ��Mg��Al��Na��
��5��Cuԭ�Ӱ뾶Ϊacm�����߳�=2
acm���������V=��2
acm��3��ÿ��������ͭԭ�Ӹ���=8��
+6��
=4����=
=
=
g/cm3=
g/cm3��
�ʴ�Ϊ��
��
��1�����þ�̯�����ÿ��������ʵ��ռ��̼ԭ�ӵĸ���Ϊ6��
| 1 |
| 3 |
| 1 |
| 2 |
| 1 |
| 2 |
��2��DΪTi��λ�����ڱ��������ڡ���B�壬��̬ԭ�Ӻ���۲�����Ų�ʽΪ3d24s2��n��AgCl��=
| 2.87g |
| 143.5g/mol |
�ʴ�Ϊ��3d24s2��B��
��3��ClԪ��+5�۵�ԭ����-2�۵���ԭ���γ�һ��-1�۵�������ΪClO3-��Cl�γ�3���ļ����µ��Ӷ���Ϊ
| 7+1-3��2 |
| 2 |
�ʴ�Ϊ��sp3�������Σ�
��4��BΪAl��ͬ����ԭ��������AlС����Na��Mg��ͬ����Ԫ�ص�һ�����ܴ�������������Mg�����Ϊ���ͽṹ�����һ�����ܴ���Al������˳��ΪMg��Al��Na���ʴ�Ϊ��Mg��Al��Na��
��5��Cuԭ�Ӱ뾶Ϊacm�����߳�=2
| 2 |
| 2 |
| 1 |
| 8 |
| 1 |
| 2 |
| m |
| V |
| ||
| V |
| ||
(2
|
| 64��4 | ||
(2
|
�ʴ�Ϊ��
| 64��4 | ||
(2
|
���������⿼����ۺϣ��漰�����ļ��㡢�ӻ���ʽ���жϡ����ӿռ乹�͵��жϵ�֪ʶ�㣬�����ܶȹ�ʽ���۲���ӶԻ������۵�֪ʶ������������ѵ��Ǿ����ļ��㣮

��ϰ��ϵ�д�
�����Ŀ
���й��ڻ�ѧ�仯��˵���У�������ǣ�������
| A����ѧ������ʱҪ�����������γ��»�ѧ��ʱҪ�ų����� |
| B����ѧ�仯��ʵ���Ǿɻ�ѧ�����ѣ��»�ѧ���γ� |
| C���ڻ�ѧ�仯�����У�һ��Ҫ���ջ�ų����� |
| D������������ų������Ĺ��̣�һ���ǻ�ѧ�仯���� |
���ܱ������У���1.0mol CO��1.0mol H2O��ϼ��ȵ�800�棬�������з�Ӧ��CO��g��+H2O��g��?CO2��g��+H2��g����һ��ʱ���÷�Ӧ�ﵽƽ�⣬���CO�����ʵ���Ϊ0.5mol��������˵����ȷ���ǣ�������
| A��800���£��÷�Ӧ�Ļ�ѧƽ�ⳣ��Ϊ0.25 |
| B��427��ʱ�÷�Ӧ��ƽ�ⳣ��Ϊ9.4����÷�Ӧ�ġ�H��0 |
| C��800���£����������ƽ����ϵ��ͨ��1.0mol��CO��g������ƽ��ʱCO���ʵ�������Ϊ33.3% |
| D��800���£����������ƽ����ϵ��ͨ��1.0mol��H2O��g������ƽ��ʱCOת����Ϊ33.3% |
ͭ�����ֳ�����������CuO��Cu2O��ijѧϰС��ȡ0.98g���þ�����ƽ������Cu��OH��2������ȣ���ͭ�����������ɣ����������¶ȱ仯��ͼ1��ʾ�����⣬ijͬѧ������������ʾ����������������������Ԫ�ص������Ĺ�ϵ���ߣ���ͼ2��ʾ�������з�����ȷ���ǣ�������
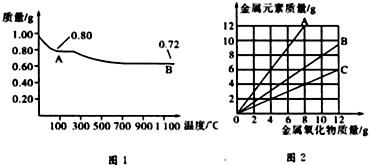

| A��ͼ1�У�A��B�Ĺ�������0.01 mol���ӷ�����ת�� |
| B��ͼ1���������й�����0.26 gˮ |
| C��ͼ2���������У���ʾCuO����������CuԪ��������ϵ��������A |
| D��ͼ1��A��B��ѧʽ�ֱ�ΪCu2O��CuO |
�ڸ��������£����мӵ�������ڻ�ѧ��Ӧ����ȫ���ĵ��ǣ�������
A����50 mL 12 mol/L��
| ||||||
B�������£���1 g
| ||||||
C����100 mL 3 mol/L��ϡ�����м���5.6 g
| ||||||
D�����³�ѹ�£�0.1 mol
|
��֪298Kʱ���ϳɰ���ӦN2��g��+3H2��g���T2NH3��g�������淴Ӧ����H=-92.2kJ?mol-1�����¶��£���1mol N2��3mol H2����һ�ܱ������У��ڴ�������ʱ���з�Ӧ����÷�Ӧ�ų�������������������ʧ�� ��������
| A��һ������92.2 kJ |
| B��һ��С��92.2 kJ |
| C��һ������92.2 kJ |
| D����ȷ�� |
��A��B��D��E���ֽ�������A��B���ԭ���ʱ��������������ΪA��B����A��D�ֱ�Ͷ���Ũ�ȵ������У�D��A��Ӧ���ң��ö��Ե缫��⺬����ͬŨ�ȵ�B2+��E2+����Һʱ��E2+�ȱ���ԭ����A��B��D��E������������ǿ������˳��Ϊ��������
| A��A��B��E��D |
| B��A��B��D��E |
| C��D��E��A��B |
| D��D��A��B��E |