��Ŀ����
12�������£�����˵����ȷ���ǣ�������| A�� | 0.1 mol/L��NaHA��ҺpH=5������Һ��c��HA-����c��H+����c��A2-����c��H2A�� | |
| B�� | ������������ʵ���Ũ�ȵ�NaCl��aq�� ���������NaClO��aq���������� | |
| C�� | pH=3��������Һ��ˮ�ĵ���̶ȴ���pH=11�İ�ˮ��Һ��ˮ�ĵ���̶� | |
| D�� | �����ͬ��0.1 mol/L��ˮ��0.1 mol/L NaOH��Һ�к�������������ߴ� |
���� A����Һ��ʾ���ԣ�HA-�ĵ���̶ȴ���ˮ��̶ȣ���c��A2-����c��H2A���������ӻ�����ˮ�ĵ��룬��c��H+����c��A2-����
B��NaCl��Һ�к��������ӡ������ӡ����������ӡ������Ӻ�ˮ���ӣ��ܹ���5�֣�����������Һ�д��������ӡ�����������ӡ������ӡ������ӡ����������Ӻ�ˮ���ӣ�
C������Ͱ�ˮ����������ˮ�ĵ���̶ȣ�����Һ�������ӡ�����������Ũ�ȴ�СӰ��ˮ�ĵ��룻
D����ˮ���������Ƶ�Ũ����ͬ���������ͬʱ���ĵ�HCl�����ʵ�����ͬ��
��� �⣺A.0.1 mol/L��NaHA��ҺpH=5��˵��HA-�ĵ���̶ȴ���ˮ��̶ȣ���Һ��ʾ���ԣ��������Ӵ�С��ϵ�ǣ�c��HA-����c��H+����c��A2-����c��H2A������A��ȷ��
B��NaCl��Һ�к��������ӡ������ӡ����������ӡ������Ӻ�ˮ���ӣ��ܹ���5�����ӣ�NaClO��Һ�д��������ӡ�����������ӡ������ӡ������ӡ����������Ӻ�ˮ���ӣ��ܹ�����6�����ӣ�����NaCl��Һ�е�������С��NaClO��Һ����B����
C��pH=3��������Һ��������Ũ��Ϊ0.001mol/L��pH=11�İ�ˮ��Һ������������Ũ��Ϊ0.001mol/L������Һ��������ˮ�ĵ��룬�����������������백ˮ������������Ũ����ȣ�������Һ��ˮ�ĵ���̶�Ӱ����ͬ����C����
D�������ͬ��0.1 mol/L��ˮ��0.1 mol/L NaOH��Һ�к������ʵ����ʵ�����ͬ��������Һ�к������������ͬ����D����
��ѡA��
���� ���⿼��������Ũ�ȴ�С�Ƚϣ���Ŀ�Ѷ��еȣ���ȷ�ε�ˮ��ԭ������Ӱ��Ϊ���ؼ���ע�������ж�����Ũ�ȴ�С�ij��÷���������������ѧ���ķ������������Ӧ��������

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | CO2 | B�� | N2 | ||
| C�� | NO2 | D�� | ֱ���dz�С�Ŀ�������� |
| A�� | HClˮ��Һ��ǿ�ᣬ�κ�ǿ�ᶼ�����г������� | |
| B�� | Cl-Ũ������ʹƽ���������Ȼ��Ƶķ����ƶ�������NaCl��s�� | |
| C�� | ��Ĵ��ڽ�����Ksp��NaCl������ֵ | |
| D�� | Ksp��NaCl���������Ӱ�죬������Cl-Ũ�ȣ���ʹKsp��NaCl����С |
| A�� | ʵ�������ռ���Һ��ȥβ���е�Cl2��Cl2+OH-�TCl-+HClO | |
| B�� | ϡ�����м���������ۣ�Fe+4H++NO3-=Fe3++NO��+2H2O | |
| C�� | ʯ��ˮ�м������С�մ���Һ��2HCO3-+Ca2++2OH-=CaCO3��+CO32+2H2O | |
| D�� | ��NaAlO2��Һ��ͨ�����CO2��2AlO2-+CO2+3H2O��2Al��OH��3��+CO32- |

�������ܼʼ��������ȫ������pH�����
| ������ | ��ʼ���� | ��ȫ���� |
| Al��OH��3 | 3.8 | 5.2 |
| Fe��OH��3 | 2.7 | 3.2 |
| Fe��OH��2 | 7.6 | 9.7 |
| Ni��OH��2 | 7.1 | 9.2 |
��1����Һ���к�����Ԫ�ص�������AlO2-��
��2�������ӷ���ʽ��ʾ����˫��ˮ��Ŀ��2Fe2++H2O2+2H+=2Fe3++2H2O��˫��ˮ���������������������A��
A������ B��������� C������ D������
��3��ȡ2.0000g������������Ʒ��������ˮ����0.2mol•L-1��EDTA��Na2H2Y������Һ�ζ����յ㣬����EDTA����ҺΪ34.50mL���ζ���ӦΪNi2++H2Y2-=NiY2-+2H+��������Ʒ����Ϊ97.0%������֪��NiSO4•7H2O��Է�������Ϊ281�����������ʷ�Ӧ����
��4������b������Һ��ΧΪ3.2-7.1����Ŀ���dz�ȥFe3+������ڵĻ�ѧʽΪFe��OH��3��
��5������a��c��Ҫʹ�õ�����������̨������Ȧ�����ձ����ƾ��ơ����������Ҫ����Ҫ����������©�������������У��������ʱ�����������H2SO4���ѧʽ����
| A�� | C1=C2 | B�� | C1��C2 | C�� | C1��C2 | D�� | ��ȷ�� |
| A�� | ʵ����������еķ�Һ������ˮ���ų�ʵ���ң�������Ⱦʵ���� | |
| B�� | Ϊ��ʹ���ֻ������ֲ��ոѷ��մ��� | |
| C�� | PM2.5��ָ������ֱ���ӽ���2.5��10-6m��1m=109nm���Ŀ������Щϸ�������ɢ�ڿ������γɵķ�ɢϵ�ǽ��� | |
| D�� | ʵ����Դ�ġ�3R�����ã�����������Դ���ģ�Reduce����������Դ���ظ�ʹ�ã�Reuse���������Դ��ѭ�����ã�Recycle�� |
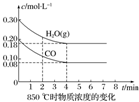 ��֪��CO��g��+H2O��g��?CO2��g��+H2��g����H=Q����ƽ�ⳣ�����¶ȱ仯���±���ʾ��
��֪��CO��g��+H2O��g��?CO2��g��+H2��g����H=Q����ƽ�ⳣ�����¶ȱ仯���±���ʾ��| �¶�/�� | 400 | 500 | 850 |
| ƽ�ⳣ�� | 9.94 | 9 | 1 |
��1��������Ӧ�Ļ�ѧƽ�ⳣ������ʽΪ$\frac{c��C{O}_{2}����c��{H}_{2}��}{c��CO����c��{H}_{2}O��}$���÷�Ӧ��Q��0���������������
��2��850��ʱ�����Ϊ10L�ķ�Ӧ���У�ͨ��һ������CO��H2O��g��������������Ӧ��CO��H2O��g�������ʵ���Ũ�ȱ仯��ͼ��ʾ����0��4minʱƽ����Ӧ����v��CO��=0.03mol/��L•min����
��3������500��ʱ���У���CO��H2O��g������ʼŨ�Ⱦ�Ϊ0.020mol•L-1���������£�CO�����ת����Ϊ75%��
��4������850��ʱ���У�����ʼʱCO��H2O��g����Ϊ1mol������H2O��g�����������Ϊx��ƽ��ʱCO��ת����Ϊy�����Ƶ�y��x֮��Ĺ�ϵx=y��