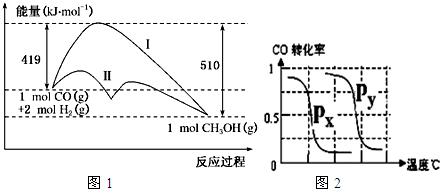
��Ŀ����
�����̾��й㷺����;��ͭ�������CuMn2O4�����ڳ����´����������е�һ����̼�ͼ�ȩ��HCHO����
��1����д��Mn2+��̬�ĵ����Ų�ʽ ����Ԫ�����ڱ�ǰ������Ԫ���гɵ���������Mn2+���Ԫ���� ��
��2����ȩ��HCHO���ķе�Ϊ-21�棬���״���CH3OH���ķе�Ϊ65�棬������ֲ�����Ҫԭ����
��3����֪ͭ�ĵ縺��Ϊ1.9���ȵĵ縺��Ϊ3.0������ˮ�Ȼ�ͭ��CuCl2������Ũ���ᣬ��[CuCl4]2-
���ӣ���ˮ�Ȼ�ͭ���ڰ���Һ�У�������[Cu��NH3��4]Cl2?2H2O���壮˼������˵����ȷ����
a��CuCl2�����ӻ������Һ�л����Cu2+��Cl-
b����[CuCl4]2-��[Cu��NH3��4]2+Ϊ�������幹�ͣ���Cu2+��ȡsp3�ӻ���ʽ
c��[Cu��NH3��4]Cl2?2H2O �������Ӽ������ۼ�����λ��
d��CuCl2���۷е��CaCl2�ĸ�
��4�������̵�һ�־���Ϊ���������ѻ����侧������ ����ԭ�ӣ�ÿ����ԭ����Χ�� ����ԭ�ӵȾ������ڣ�
��1����д��Mn2+��̬�ĵ����Ų�ʽ
��2����ȩ��HCHO���ķе�Ϊ-21�棬���״���CH3OH���ķе�Ϊ65�棬������ֲ�����Ҫԭ����
��3����֪ͭ�ĵ縺��Ϊ1.9���ȵĵ縺��Ϊ3.0������ˮ�Ȼ�ͭ��CuCl2������Ũ���ᣬ��[CuCl4]2-
���ӣ���ˮ�Ȼ�ͭ���ڰ���Һ�У�������[Cu��NH3��4]Cl2?2H2O���壮˼������˵����ȷ����
a��CuCl2�����ӻ������Һ�л����Cu2+��Cl-
b����[CuCl4]2-��[Cu��NH3��4]2+Ϊ�������幹�ͣ���Cu2+��ȡsp3�ӻ���ʽ
c��[Cu��NH3��4]Cl2?2H2O �������Ӽ������ۼ�����λ��
d��CuCl2���۷е��CaCl2�ĸ�
��4�������̵�һ�־���Ϊ���������ѻ����侧������
���㣺�����ļ���,ԭ�Ӻ�������Ų�,�����ijɼ����,����Ĵ��ڶ��������ʵ�Ӱ��
ר�⣺
��������1��Mn��ԭ������Ϊ25�������������ԭ����д��Mn�Ļ�̬ԭ�ӵĵ����Ų�ʽ��������ȷ��Mn2+��̬�ĵ����Ų�ʽ��Mn2+�гɵ�������Ϊ5��
��2����������������ʵķе���죬�״�����֮�������γɷ��Ӽ����������ȩ���Ӽ䲻���������
��3��a��CuCl2�����ӻ������ֻ��[CuCl4]2-���ӣ�������Һ�в������Cu2+��Cl-��
b����[CuCl4]2-��[Cu��NH3��4]2+Ϊ�������幹�ͣ���Cu�ĵ��Ӷ���Ϊ4������sp3�ӻ���
c��[Cu��NH3��4]Cl2?2H2O ��N-H���ڹ��ۼ���Cu��NH3����λ����ϣ�[CuCl4]2-���Ӻ������Ӽ��γɵ������Ӽ���
d�����Ӿ�����۷е�����Ӱ뾶����ɶ����йأ�
��4�������������ѻ���Mnԭ�Ӵ��ڶ����������ϣ����ݾ�̯�����㾧���к��е�Mnԭ����Ŀ������������ÿ�������϶���һ��ԭ�ӣ������϶���һ��ԭ�ӣ�
��2����������������ʵķе���죬�״�����֮�������γɷ��Ӽ����������ȩ���Ӽ䲻���������
��3��a��CuCl2�����ӻ������ֻ��[CuCl4]2-���ӣ�������Һ�в������Cu2+��Cl-��
b����[CuCl4]2-��[Cu��NH3��4]2+Ϊ�������幹�ͣ���Cu�ĵ��Ӷ���Ϊ4������sp3�ӻ���
c��[Cu��NH3��4]Cl2?2H2O ��N-H���ڹ��ۼ���Cu��NH3����λ����ϣ�[CuCl4]2-���Ӻ������Ӽ��γɵ������Ӽ���
d�����Ӿ�����۷е�����Ӱ뾶����ɶ����йأ�
��4�������������ѻ���Mnԭ�Ӵ��ڶ����������ϣ����ݾ�̯�����㾧���к��е�Mnԭ����Ŀ������������ÿ�������϶���һ��ԭ�ӣ������϶���һ��ԭ�ӣ�
���
�⣺��1��Mn��ԭ������Ϊ25����̬ԭ�ӵĵ����Ų�ʽΪ��1s22s22p63s23p63d54s2����Mn2+��̬�ĵ����Ų�ʽ�ɱ�ʾΪ1s22s22p63s23p63d5����[Ar]3d5����
Mn2+�гɵ�������Ϊ5��������Ԫ�����ڱ�ǰ������Ԫ���гɵ���������Mn2+���Ԫ����Cr��
�ʴ�Ϊ��1s22s22p63s23p63d5����[Ar]3d5����Cr��
��2����״�����֮�������γɷ��Ӽ��������е�ߣ�����ȩ���Ӽ䲻���������
�ʴ�Ϊ���״���CH3OH�����Ӽ��γ������ʹ���Ӽ��������ȼ�ȩ��HCHO���Ĵ�����е�ߣ�
��3��a��CuCl2�����ӻ������ֻ��[CuCl4]2-���ӣ�������Һ�в������Cu2+��Cl-���ʴ���
b����[CuCl4]2-��[Cu��NH3��4]2+Ϊ�������幹�ͣ���Cu�ĵ��Ӷ���Ϊ4������sp3�ӻ�������ȷ��
c��[Cu��NH3��4]Cl2?2H2O ��N-H���ڹ��ۼ���Cu��NH3����λ����ϣ�[CuCl4]2-���Ӻ������Ӽ��γɵ������Ӽ�������ȷ��
d�����Ӿ�����۷е�����Ӱ뾶����ɶ����йأ�ͭ���Ӱ뾶�ȸ����Ӱ뾶��CuCl2���۷е�ͣ��ʴ���
�ʴ�Ϊ��bc��
��4��Mnԭ�Ӵ��ڶ����������ϣ������к��е�Mnԭ����ĿΪ8��
+6��
=4��
�������������У�����������ÿ�������϶���һ��ԭ�ӣ������϶���һ��ԭ�ӣ�����ÿ������ԭ����Χ���ڵĽ���ԭ����3��8��
=12��
�ʴ�Ϊ��4��12��
Mn2+�гɵ�������Ϊ5��������Ԫ�����ڱ�ǰ������Ԫ���гɵ���������Mn2+���Ԫ����Cr��
�ʴ�Ϊ��1s22s22p63s23p63d5����[Ar]3d5����Cr��
��2����״�����֮�������γɷ��Ӽ��������е�ߣ�����ȩ���Ӽ䲻���������
�ʴ�Ϊ���״���CH3OH�����Ӽ��γ������ʹ���Ӽ��������ȼ�ȩ��HCHO���Ĵ�����е�ߣ�
��3��a��CuCl2�����ӻ������ֻ��[CuCl4]2-���ӣ�������Һ�в������Cu2+��Cl-���ʴ���
b����[CuCl4]2-��[Cu��NH3��4]2+Ϊ�������幹�ͣ���Cu�ĵ��Ӷ���Ϊ4������sp3�ӻ�������ȷ��
c��[Cu��NH3��4]Cl2?2H2O ��N-H���ڹ��ۼ���Cu��NH3����λ����ϣ�[CuCl4]2-���Ӻ������Ӽ��γɵ������Ӽ�������ȷ��
d�����Ӿ�����۷е�����Ӱ뾶����ɶ����йأ�ͭ���Ӱ뾶�ȸ����Ӱ뾶��CuCl2���۷е�ͣ��ʴ���
�ʴ�Ϊ��bc��
��4��Mnԭ�Ӵ��ڶ����������ϣ������к��е�Mnԭ����ĿΪ8��
| 1 |
| 8 |
| 1 |
| 2 |
�������������У�����������ÿ�������϶���һ��ԭ�ӣ������϶���һ��ԭ�ӣ�����ÿ������ԭ����Χ���ڵĽ���ԭ����3��8��
| 1 |
| 2 |
�ʴ�Ϊ��4��12��
���������⿼���˺�����ӵ��Ų�����������Լ������ļ��㣬�ۺ��Խ�ǿ���ѶȽϴ�ע������ӻ����͵��жϷ�����

��ϰ��ϵ�д�
 ���Ž�������С״Ԫϵ�д�
���Ž�������С״Ԫϵ�д�
�����Ŀ
����ʵ��װ�ã����̶ֹ�װ����ȥ�����й���������ȷ���ǣ�������
A�� ���Բ���п��������ķ�Ӧ���� |
B�� ���Խ����к��ȵIJⶨʵ�� |
C�� ����֤�¶ȶԻ�ѧƽ���Ӱ�� |
D�� ���о������Ի�ѧ��Ӧ���ʵ�Ӱ�� |
ʵ���ǻ�ѧ�о��Ļ��������й��ڸ�ʵ��װ�õ�������ȷ���ǣ�������


| A��װ�âٳ����ڷ����Ҵ�����ˮ��ɵĻ���� |
| B��װ�âڿ���������NH3��HCl���壬����ֹ���� |
| C��װ�âۿ������ռ�H2��CO2��Cl2��NO������ |
| D��װ�âܿ����ڸ���ռ������������ն���İ��� |
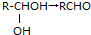 +H2O������C�ܷ���������Ӧ������C��Ũ�������¿��Է�������������ԭ��Ӧ����E��F����E��F������̼ԭ�ӵ���Ŀ��ͬ��
+H2O������C�ܷ���������Ӧ������C��Ũ�������¿��Է�������������ԭ��Ӧ����E��F����E��F������̼ԭ�ӵ���Ŀ��ͬ��
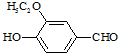 ����ʳƷ���Ӽ�������ԭ�ϣ�����ζ�����ȩ����Ũ����
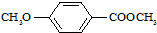
����ʳƷ���Ӽ�������ԭ�ϣ�����ζ�����ȩ����Ũ����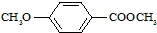 ����һ��ҽҩ�м��壬����ƺ�������������ȩ��
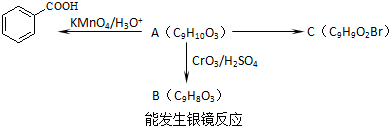
����һ��ҽҩ�м��壬����ƺ�������������ȩ�� 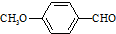 ���ϳ�X���÷�Ӧ����ͼ��ʾ���£�
���ϳ�X���÷�Ӧ����ͼ��ʾ���£�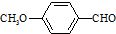 ��
��
 ��ͼΪ10mLһ�����ʵ���Ũ�ȵ����ᣨHCl������һ��Ũ�ȵ�NaOH��Һ�ζ���ͼʾ������ͼʾ�Ƴ�HCl��NaOH�����ʵ���Ũ���ǣ�������
��ͼΪ10mLһ�����ʵ���Ũ�ȵ����ᣨHCl������һ��Ũ�ȵ�NaOH��Һ�ζ���ͼʾ������ͼʾ�Ƴ�HCl��NaOH�����ʵ���Ũ���ǣ�������