��Ŀ����
ʯ����һ�ֽ��ܽ������ϡ�ijʯ����Ʒ���ɶ�ˮʯ�ࣨCaSO4��2H2O������ˮʯ�ࣨCaSO4�� H2O������ˮʯ�ࣨCaSO4�����ֳɷ���ɣ�����ֺ����������з����ⶨ��
H2O������ˮʯ�ࣨCaSO4�����ֳɷ���ɣ�����ֺ����������з����ⶨ��
�ٳ�ȡ8.70 g��ʯ����������400�����ʧȥȫ���ᾧˮ��������Ϊ8.16 g��
���ٳ�ȡ8.70 g�ĸ�ʯ������������80%�ľƾ���Һ��ʹCaSO4��ȫˮ����CaSO4�� H2O����ʱ�������������ƾ���Һ�������ã�������ȡ����������������������Ϊ8.97 g��
H2O����ʱ�������������ƾ���Һ�������ã�������ȡ����������������������Ϊ8.97 g��
���ڢ�������8.97 g�����м�������ˮ��ʹȫ����CaSO4�� H2O��ˮ����CaSO4��2H2O��ȡ��������ٴγ�����������Ϊ10.32 g��
H2O��ˮ����CaSO4��2H2O��ȡ��������ٴγ�����������Ϊ10.32 g��
��1�������нᾧˮ����������Ϊ__________________����С����ʾ����
��2����������ˮʯ�ࣨCaSO4�������ʵ���Ϊ____________ mol��
��3������������ʯ������ʵ���֮��Ϊ��
n(CaSO4��2H2O)��n(CaSO4�� H2O)��n(CaSO4)=
__________________��
H2O)��n(CaSO4)=
__________________��
���𰸡�
��1��0.062 ��2��0.03 ��3��1��2��3
����������

��ϰ��ϵ�д�
�����Ŀ
 H2O)��n(CaSO4)=__________________��
H2O)��n(CaSO4)=__________________��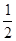 H2O������ˮʯ�ࣨCaSO4�����ֳɷ���ɣ�����ֺ����������з����ⶨ��
H2O������ˮʯ�ࣨCaSO4�����ֳɷ���ɣ�����ֺ����������з����ⶨ��