��Ŀ����
������KxFe(C2O4)y��3H2O��FeΪ��3�ۣ���һ�ֹ������ϣ�ʵ���ҿ��������µķ������Ʊ����ֲ��ϲ��ⶨ���ֲ��ϵ���ɡ�
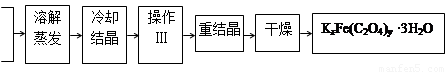 ���Ʊ�
���Ʊ�
K2C2O4
FeCl3
��1���ᾧʱӦ����Һ�ñ�ˮ��ȴ���ںڰ����ȴ����������������������ԭ���ǣ�__________________��
��2���������������___________ ��
����ɲⶨ
��ȡһ�������ľ���������ƿ�У���������������ˮ��ϡH2SO4����C2O42��ת��ΪH2C2O4����0.1000 mol��L��1KMnO4��Һ�ζ���������KMnO4��Һ24.00mLʱǡ����ȫ��Ӧ��������Һ�м��������Ļ�ԭ����ǡ�ý�Fe3+��ȫת��ΪFe2+����KMnO4��Һ�����ζ�����Fe2+��ȫ����ʱ����ȥKMnO4��Һ4.00mL���ڶ��εζ������ӻ�ѧ����ʽΪ��
__________________________ ___ __________________________
��3������100mL 0.1000 mol��L��1KMnO4��Һ���ζ�ʵ��������IJ����������ձ�������������ͷ�ιܡ���Ͳ����ƿ���_______________________________�����������ƣ���
��4���û�����KxFe(C2O4)y��3H2O�У�x��_______��
��1���ñ�ˮ��ȴ��������������ľ��壬�ڰ����Է�ֹ����ֽ⣨2�֣�
��2�����ˡ�ϴ�ӣ�2�֣�MnO4��+5Fe2++8H+��Mn2++5Fe3++4H2O��2�֣�
��3��100mL����ƿ����ʽ�ζ��ܣ�2�֣�
��4��3��2�֣�
��ʾ��2MnO4�� + 5H2C2O4 + 6H+��2Mn2++10CO2��+8H2O
2mol 5mol
0.1000 mol��L��1��24.00��10��3L n ��ʽ��ã�n=6��10��3 mol
���� KxFe(C2O4)y��3H2O
KxFe(C2O4)y��3H2O
MnO4�� + 5Fe2+ + 8H+��Mn2++5Fe3++4H2O
1mol 5mol

 ��ʽ��ã�y=3����x=3
��ʽ��ã�y=3����x=3
��������

 ��Ǭ����������ҵ���ּ����ӱ����������ϵ�д�
��Ǭ����������ҵ���ּ����ӱ����������ϵ�д�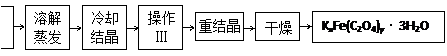 K2C2O4
K2C2O4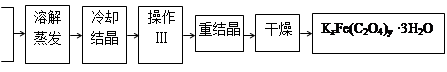 ���Ʊ�
���Ʊ� 4Fe(OH)3��8OH����3O2��
4Fe(OH)3��8OH����3O2��

 4Fe(OH)3��8OH����3O2��
4Fe(OH)3��8OH����3O2��
