��Ŀ����
8���л���A��B��C��D��E��F��ֻ��C��H��O����Ԫ�أ���һ��������������ת����ϵ��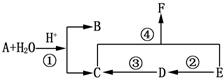
��������Ϣ�ɹ��ο���
��A�����й���24��ԭ�ӣ��������ܶ�����ͬ״���������ܶȵ�95����
��B�����к��б�����������������ȡ�������ڼ��������¿�������������ͭ����Һ��Ӧ����FeCl3��Һ����������ɫ��
��A��B��Է�������֮�����54��
��������Ƶ����ش��������⣺
��1��A�ķ���ʽC11H10O3��C��Ħ������72g/mol��E�й����ŵ�����̼̼˫�����ǻ���
��2����Ӧ�ٵ�������ȡ����Ӧ����Ӧ�ڵ�������������Ӧ��
��3��D+Cu��OH��2��Ӧ�Ļ�ѧ����ʽCH2=CHCHO+2Cu��OH��2$\frac{\underline{\;\;��\;\;}}{\;}$CH2=CHCOOH+2Cu2O��+2H2O����Ӧ�ܵĻ�ѧ����ʽ
 ��
����4��д����������Ҫ���B��ͬ���칹�壨������B������
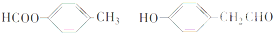 ��
���ٱ����ϵ�һ�����ֻ�����ֽṹ��
�ڿɷ���������Ӧ��
�ۿ���NaOH��Һ��Ӧ��
���� A�����ܶ�����ͬ״���������ܶȵ�95������A����Է�������Ϊ95��2=190��A�����������·���ˮ�ⷴӦ��B��C����A�к���������B�����к��б�����������������ȡ�������ڼ��������¿�������������ͭ����Һ��Ӧ����B�к���-CHO��B��FeCl3��Һ����������ɫ����B�в������ǻ���Eת���õ�C��C��Eת���õ�F��EӦ����-CH2OH�ṹ��C�������Ȼ���F������������֪B�л����д��ǻ���B����Է�������Ϊ190-54=136��B�ı�������-CHO��-CH2OH���ţ���B�ķ���ʽΪC8H8O2�������Է���������֪��C��ֻ�ܺ���1���Ȼ�����1molAˮ��õ�1molB��1mol��A��B��Է�������֮��Ϊ54����C����Է�������Ϊ54+18=72��ȥ��1��-COOHʣ��������ԭ������Ϊ72-45=27����$\frac{27}{12}$=2��3����CΪCH2=CHCOOH�����Ƶ�DΪCH2=CHCHO��EΪCH2=CHCH2OH����FΪCH2=CHCOOCH=CH2�����C��B�ķ���ʽ��֪A�ķ���ʽΪC11H10O3������A�����й���24��ԭ�ӣ�
��� �⣺A�����ܶ�����ͬ״���������ܶȵ�95������A����Է�������Ϊ95��2=190��A�����������·���ˮ�ⷴӦ��B��C����A�к���������B�����к��б�����������������ȡ�������ڼ��������¿�������������ͭ����Һ��Ӧ����B�к���-CHO��B��FeCl3��Һ����������ɫ����B�в������ǻ���Eת���õ�C��C��Eת���õ�F��EӦ����-CH2OH�ṹ��C�������Ȼ���F������������֪B�л����д��ǻ���B����Է�������Ϊ190-54=136��B�ı�������-CHO��-CH2OH���ţ���B�ķ���ʽΪC8H8O2�������Է���������֪��C��ֻ�ܺ���1���Ȼ�����1molAˮ��õ�1molB��1mol��A��B��Է�������֮��Ϊ54����C����Է�������Ϊ54+18=72��ȥ��1��-COOHʣ��������ԭ������Ϊ72-45=27����$\frac{27}{12}$=2��3����CΪCH2=CHCOOH�����Ƶ�DΪCH2=CHCHO��EΪCH2=CHCH2OH����FΪCH2=CHCOOCH=CH2�����C��B�ķ���ʽ��֪A�ķ���ʽΪC11H10O3������A�����й���24��ԭ�ӣ�
��1����������ķ�����֪��A�ķ���ʽΪ C11H10O3��C��Ħ������Ϊ72g/mol��EΪCH2=CHCH2OH��E�й����ŵ�����Ϊ̼̼˫�����ǻ���
�ʴ�Ϊ��C11H10O3��72g/mol��̼̼˫�����ǻ���
��2����Ӧ�ٵ�������ȡ����Ӧ����Ӧ�ڵ�������������Ӧ��
�ʴ�Ϊ��ȡ����Ӧ��������Ӧ��
��3��D+Cu��OH��2��Ӧ�Ļ�ѧ����ʽ��CH2=CHCHO+2Cu��OH��2$\frac{\underline{\;\;��\;\;}}{\;}$CH2=CHCOOH+2Cu2O��+2H2O��
��Ӧ�ܵĻ�ѧ����ʽΪ  ��
��
�ʴ�Ϊ��CH2=CHCHO+2Cu��OH��2$\frac{\underline{\;\;��\;\;}}{\;}$CH2=CHCOOH+2Cu2O��+2H2O�� ��
��
��4��B�ķ���ʽΪC8H8O2��B��ͬ���칹���������Ҫ�ٱ����ϵ�һ�����ֻ�����ֽṹ��˵�������ϵ�����ȡ�������ڶ�λ���ڿɷ���������Ӧ��˵����ȩ�����ۿ���NaOH��Һ��Ӧ��˵�����Ȼ�����ǻ��������ȣ�������B��ͬ���칹����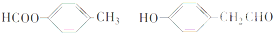 ��������B��������
��������B��������
�ʴ�Ϊ��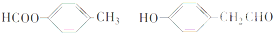 ��
��
���� ���⿼���л����ƶϣ���������л�����Է����������й����ʵ����ʽ������ʵ��ƶϣ���Ҫѧ���������չ����ŵ�������ת�������ؿ���ѧ�����������������Ѷ��еȣ�

 ������ʱ����ҵ����ϵ�д�
������ʱ����ҵ����ϵ�д� ��ĩ���ƾ�ϵ�д�
��ĩ���ƾ�ϵ�д� ���ɿ��ñ���ϵ�д�
���ɿ��ñ���ϵ�д� ��
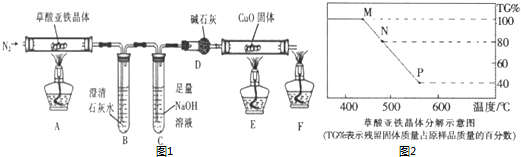
��
 ��3��þ����Mg2+��4��������
��3��þ����Mg2+��4��������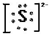 ��
��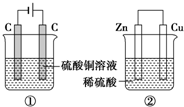 ��ͼ����װ�ã�����ʱת�Ƶĵ�������ȣ���Ӧ��ɺ������м���19.6gCu��OH��2���壬��Һǡ���ָܻ������ǰ��Ũ�Ⱥ��������������Һ����ı仯����
��ͼ����װ�ã�����ʱת�Ƶĵ�������ȣ���Ӧ��ɺ������м���19.6gCu��OH��2���壬��Һǡ���ָܻ������ǰ��Ũ�Ⱥ��������������Һ����ı仯����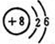
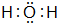 ��
��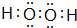 �����ڹ��ۻ����
�����ڹ��ۻ����