��Ŀ����
9��ij�����Ĺ�ҵ��ˮ�к��д�����FeSO4���϶��CuSO4������Na2SO4��Ϊ�˼�����Ⱦ�����Ϊ���������ƻ��Ӹ÷�ˮ�л������������ͽ���ͭ���������������ͼ����ɻ�������������ͭ��ʵ�鷽�������ɹ�ѡ����Լ�Ϊ���ۡ�ϡH2SO4��NaOH��Һ���Լ���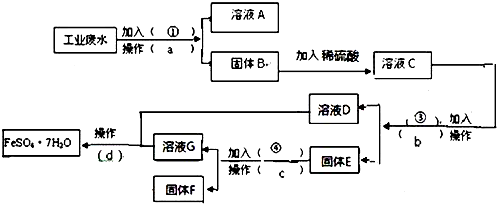
��1������a������Ϊ���ˣ�����Ҫ�IJ�������Ϊ©�������������ձ���
��2������E�ijɷ�ΪFe��Cu��������Լ���Ϊϡ���ᣬ�����Ļ�ѧ����ʽΪFe+H2SO4=FeSO4+H2����
��3�������Լ��ٵ�Ŀ���ǽ���Һ�е�Fe2+��Cu2+ת��Ϊ�����������뺬��Na+����Һ���룮
��4������ҺD����ҺG�еõ�FeSO4.7H2O����IJ���Ϊ����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ����
��5������ˮ����������������ͭ����������ɵĻ����Һ������c��Cu2+��=0.1mol/L��c��Fe2+��=0.4mol/L��c��SO42-��=0.6mol/L����֪������Ũ�Ⱥ��Բ��ƣ���c��Na+��Ϊ0.2mol/L��
���� �ȼӹ�������˵õ�Cu��OH��2��Fe��OH��2��������Һ��Na2SO4��NaOH��Һ����Cu��OH��2��Fe��OH��2�мӹ���ϡH2SO4����ҺC�ijɷ�ΪCuSO4��FeSO4��H2SO4��
����Һ�мӹ����������˳�Fe��Cu����ҺD��FeSO4���ڹ���E�мӹ���ϡH2SO4�����˳�Cu��ʵ���˻��ս���ͭ����Һ��FeSO4��H2SO4�������Һ��һ�飬����������Ũ������ȴ�ᾧ�����ˣ��͵õ�FeSO4•7H2O�����������������Դ˽����⣮
��� �⣺�ȼӹ�������˵õ�Cu��OH��2��Fe��OH��2��������Һ��Na2SO4��NaOH��Һ����Cu��OH��2��Fe��OH��2�мӹ���ϡH2SO4����ҺC�ijɷ�ΪCuSO4��FeSO4��H2SO4������Һ�мӹ����������˳�Fe��Cu����ҺD��FeSO4���ڹ���E�мӹ���ϡH2SO4�����˳�Cu��ʵ���˻��ս���ͭ����Һ��FeSO4��H2SO4�������Һ��һ�飬����������Ũ������ȴ�ᾧ�����ˣ��͵õ�FeSO4•7H2O����������������
��1������aΪ���ˣ����ڷ�������Һ�壬�õ��IJ���������©�������������ձ����ʴ�Ϊ�����ˣ�©�������������ձ���
��2������EΪFe��Cu���ɼ������ᣬ�������ᷴӦ����������������Ӧ�Ļ�ѧ����ʽΪFe+H2SO4=FeSO4+H2����
�ʴ�Ϊ��Fe��Cu��ϡ���Fe+H2SO4=FeSO4+H2����
��3���Լ���ӦΪ�������ƣ��ɽ���Һ�е�Fe2+��Cu2+ת��Ϊ�����������뺬��Na+����Һ���룬�ʴ�Ϊ������Һ�е�Fe2+��Cu2+ת��Ϊ�����������뺬��Na+����Һ���룻
��4������ҺD����ҺG�еõ�FeSO4.7H2O����IJ���Ϊ ����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ����
�ʴ�Ϊ������Ũ�������ˣ�
��5����Һ�ʵ����ԣ����ݵ���غ��֪��2c��Cu2+��+c��Na+��+2c��Fe2+��=2c��SO42-����c��Na+��=2��0.6mol/L-2��0.1mol/L-2��0.4mol/L=0.2 mol/L��
�ʴ�Ϊ��0.2 mol/L��
���� ���⿼���������ƶϼ����ʵķ��롢�ᴿ��Ϊ��Ƶ���㣬������ѧ���ķ�����ʵ�������Ŀ��飬�������ʵ����ʡ�����֮��ķ�Ӧ����������ѶȲ������ʱҪע������ķ���Ҫ����Ϊ���ֿ�����������Լ������Ժ�һ������Ҫ��֮��ȥ��

| A�� | 20 mL 12 mol/L������ | B�� | 10 mL 18 mol/L������ | ||
| C�� | 80 mL 2 mol/L������ | D�� | 40 mL 14 mol/L������ |
��1��������Һ��������100mL1.5mol/L-1��Һ����ȡ20mL 36.5%������ϡ�͵�200mL������KOH��Һʱ��Ҫ�õIJ��������У��ձ�������������ͷ�ιܺ�100mL����ƿ��
��2���ζ������Է�̪��ָʾ�����жϵ���ζ��յ��ʵ����������Һ����ɫ��Ϊdz��ɫ���Ұ�����ڲ���ɫ��
�ڲⶨ�Ľ�����±�
| �ⶨ���� | ��������/mL | KOH��Һ����� | |
| �ζ�ǰ�̶�/mL | �ζ���̶�/mL | ||
| 1 | 25.00 | 1.02 | 21.03 |
| 2 | 25.00 | 2.00 | 21.99 |
| 3 | 25.00 | 0.20 | 20.20 |
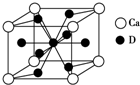 A��B��C��D��Ԫ�����ڱ���ǰ36��Ԫ�أ����ǵĺ˵�����������ڶ�����Ԫ��Aԭ�ӵĺ���ɶԵ�������δ�ɶԵ�������2������3���ܼ���Bԭ�ӵ������p����ĵ���Ϊ������ṹ��C�ǵؿ��к�������Ԫ�أ�D�ǵ�������Ԫ�أ���ԭ�Ӻ�����������������ԭ����ͬ�����������Ӿ���������ش��������⣺
A��B��C��D��Ԫ�����ڱ���ǰ36��Ԫ�أ����ǵĺ˵�����������ڶ�����Ԫ��Aԭ�ӵĺ���ɶԵ�������δ�ɶԵ�������2������3���ܼ���Bԭ�ӵ������p����ĵ���Ϊ������ṹ��C�ǵؿ��к�������Ԫ�أ�D�ǵ�������Ԫ�أ���ԭ�Ӻ�����������������ԭ����ͬ�����������Ӿ���������ش��������⣺ ��
��
 ��
��
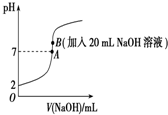 25���£���20mL 0.2mol•L-1��������еμ�0.2mol•L-1��NaOH��Һʱ����Һ��pH�仯��ͼ��ʾ����ش��������⣺
25���£���20mL 0.2mol•L-1��������еμ�0.2mol•L-1��NaOH��Һʱ����Һ��pH�仯��ͼ��ʾ����ش��������⣺