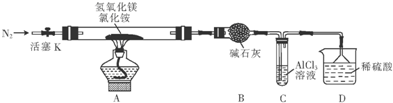
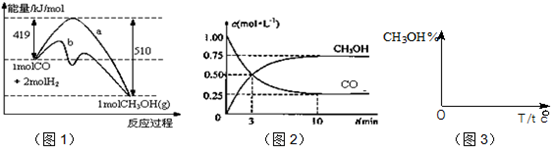
��Ŀ����
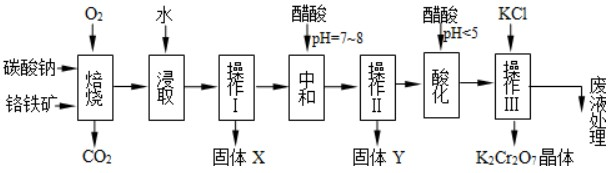
��������Ҫ�ɷ�ΪFeO?Cr2O3������������Al2O3��һ���������Cr2O3��������ԼΪ40%���ɸ������Ʊ��ظ���صķ������£�
��֪��
��4FeO?Cr2O3+8Na2CO3+7O2
8Na2CrO4+2Fe2O3+8CO2����
��Na2CO3+Al2O3
2NaAlO2+CO2����
��Cr2O72-+H2O?2CrO42-+2H+
��������ش��������⣺
��1������IΪ ������X����Ҫ���� ����д��ѧʽ����Ҫ����ữ��������Һ��pH�Ƿ����4.5��Ӧ��ʹ�� ����д�������Լ����ƣ���
��2���ữ�����ô��������ҺpH��5����Ŀ���� ��
��3���������жಽ��ɣ����K2Cr2O7����IJ��������ǣ�����KCl���塢����Ũ���� �����ˡ� �����
��4������Y����Ҫ����������������д��������Һ��pH=7��8ʱ�����������������ӷ���ʽ ��

��֪��
��4FeO?Cr2O3+8Na2CO3+7O2
| ||
��Na2CO3+Al2O3
| ||
��Cr2O72-+H2O?2CrO42-+2H+
��������ش��������⣺
��1������IΪ
��2���ữ�����ô��������ҺpH��5����Ŀ����
��3���������жಽ��ɣ����K2Cr2O7����IJ��������ǣ�����KCl���塢����Ũ����
��4������Y����Ҫ����������������д��������Һ��pH=7��8ʱ�����������������ӷ���ʽ
���㣺���ʷ�����ᴿ�ķ����ͻ��������ۺ�Ӧ��,�Ʊ�ʵ�鷽�������
ר�⣺ʵ�������
������������ͨ�����գ�����Na2CrO4��Fe2O3��MgO��NaAlO2�Ļ����ϵ��Ȼ���ˮ�ܽ�ù���Fe2O3��MgO����ҺNa2CrO4��NaAlO2���ٵ�����Һ��PH��ʹƫ��������ȫ����������������Һ��PHʹCrO42-ת��ΪCr2O72-�������������Һ�м����Ȼ��أ������ܽ�ȼ�С��K2Cr2O7��
��1����������ͼ����ɷ�Ӧ�����жϹ���X�ijɷ֣�ͨ��PH��ֻֽ�ܲⶨ��ҺPH���������ⶨPH��С������Ҫ�þ�ȷ��ֽ��pH�ƣ�
��2���������ͼ�ͷ�Ӧ�����еõ����ʷ������ữ�����ô��������ҺpH��5Ϊ��ת��CrO42-����ΪCr2O72-��
��3����Һ�еõ����ʾ���ķ���������Ũ������ȴ�ᾧ������ϴ�Ӳ�������ش�
��4��������Һ��pH=7��8ʱ��������Ժ�ƫ���������֮�䷴Ӧ������������������
��1����������ͼ����ɷ�Ӧ�����жϹ���X�ijɷ֣�ͨ��PH��ֻֽ�ܲⶨ��ҺPH���������ⶨPH��С������Ҫ�þ�ȷ��ֽ��pH�ƣ�
��2���������ͼ�ͷ�Ӧ�����еõ����ʷ������ữ�����ô��������ҺpH��5Ϊ��ת��CrO42-����ΪCr2O72-��
��3����Һ�еõ����ʾ���ķ���������Ũ������ȴ�ᾧ������ϴ�Ӳ�������ش�
��4��������Һ��pH=7��8ʱ��������Ժ�ƫ���������֮�䷴Ӧ������������������
���
�⣺��1�����������Ҫ�ɷֿɱ�ʾΪFeO?Cr2O3��������MgO��Al2O3��Fe2O3�����ʣ�����������̼���ƣ�������ӦΪ��
��4FeO?Cr2O3+8Na2CO3+7O2
8Na2CrO4+2Fe2O3+8CO2����
��Na2CO3+Al2O3
2NaAlO2+CO2����
��Cr2CO72-+H2O?2CrO42-+2H+��
�������ǹ��ˣ��õ�����XΪFe2O3��MgO��Ҫ����ữ��������Һ��pH�Ƿ����4.5����ͨpH��ֻֽ�ܲⶨ��ҺpH���������ǽ��Ʋⶨ��ȷ�ⶨ��Ҫ��pH�ƻ�ȷpH��ֽ��
�ʴ�Ϊ�����ˣ�Fe2O3��MgO��pH�ƻ���pH��ֽ��
��2���ữ�����ô��������ҺpH��5����������ͼ�����ʵ�ת�����Ʊ�Ŀ�Ŀ�֪����Ϸ�Ӧƽ��Cr2O72-+H2O?2CrO42-+2H+�������ᣬ������Ũ������ƽ�����ƣ�������ʹCrO42-ת��ΪCr2O72-���ʴ�Ϊ��ʹCrO42-ת��ΪCr2O72-��
��3���������жಽ��ɣ����K2Cr2O7����IJ��������ǣ�����KCl���塢����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ�����õ����壬
�ʴ�Ϊ����ȴ�ᾧ��ϴ�ӣ�
��4��������Һ��pH=7��8ʱ��������Ժ�ƫ���������֮�䷴Ӧ��������������������ӦΪ��CH3COOH+AlO2-+H2O=Al��OH��3��+CH3COO-���ʴ�Ϊ��CH3COOH+AlO2-+H2O=Al��OH��3��+CH3COO-��
��4FeO?Cr2O3+8Na2CO3+7O2
| ||
��Na2CO3+Al2O3
| ||
��Cr2CO72-+H2O?2CrO42-+2H+��
�������ǹ��ˣ��õ�����XΪFe2O3��MgO��Ҫ����ữ��������Һ��pH�Ƿ����4.5����ͨpH��ֻֽ�ܲⶨ��ҺpH���������ǽ��Ʋⶨ��ȷ�ⶨ��Ҫ��pH�ƻ�ȷpH��ֽ��
�ʴ�Ϊ�����ˣ�Fe2O3��MgO��pH�ƻ���pH��ֽ��
��2���ữ�����ô��������ҺpH��5����������ͼ�����ʵ�ת�����Ʊ�Ŀ�Ŀ�֪����Ϸ�Ӧƽ��Cr2O72-+H2O?2CrO42-+2H+�������ᣬ������Ũ������ƽ�����ƣ�������ʹCrO42-ת��ΪCr2O72-���ʴ�Ϊ��ʹCrO42-ת��ΪCr2O72-��
��3���������жಽ��ɣ����K2Cr2O7����IJ��������ǣ�����KCl���塢����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ�����õ����壬
�ʴ�Ϊ����ȴ�ᾧ��ϴ�ӣ�
��4��������Һ��pH=7��8ʱ��������Ժ�ƫ���������֮�䷴Ӧ��������������������ӦΪ��CH3COOH+AlO2-+H2O=Al��OH��3��+CH3COO-���ʴ�Ϊ��CH3COOH+AlO2-+H2O=Al��OH��3��+CH3COO-��
���������⿼���������Ʊ����̺ͷ����ķ����жϣ��������ʵ�Ӧ�ã������Ϣ�ķ������⣬���������ע������ͻ������������ǽ���ؼ�����Ŀ�Ѷȴ�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
���з�Ӧ�����ӷ���ʽ��ȷ���ǣ�������
| A��������SO2ͨ����İ�ˮ�У�SO2+NH3?H2O�THSO3-+NH4+ |
| B���ѹ�����NaHCO3��Һ��Ca��OH��2��Һ��ϣ�HCO3-+Ca2++OH-�TCaCO3��+H2O |
| C��������Һˮ�⣺S2-+2H2O�TH2S+2OH- |
| D����������Һ�������Һ��ϣ�SiO32-+2H+�TH2 SiO3�� |
�����봦��������Ӧ�ķ���ʽ����ȷ���ǣ�������
| A�������缫����KOH��ҺΪ�������Һ����--����ȼ�ϵ�أ��ŵ�ʱ�ĸ�����Ӧʽ��N2H4-4e-+4OH-=N2+4H2O | ||||
B��ȼú�м�������ʯ���Լ���SO2���ŷţ�O2+2CaO+2SO2
| ||||
| C���������ʴ�̲�����SiO2+4HF=SiF4��+2H2O | ||||
| D��������������ǵĺ�ˮ�У���ʹˮ���壺Al3++3H2O=Al��OH��3��+3H+ |
ֻ���Թܺͽ�ͷ�ιܾͿ��Լ�������и�����Һ�ǣ�������
��AlCl3��Һ��NaOH��Һ����
��Ca��HCO3��2��Һ��NaOH��Һ
��NaAlO2��Һ��NaHSO4����
��Al2��SO4��3��Һ�Ͱ�ˮ ��
��AgNO3��Һ�Ͱ�ˮ��
��AlCl3��Һ��NaOH��Һ����
��Ca��HCO3��2��Һ��NaOH��Һ
��NaAlO2��Һ��NaHSO4����
��Al2��SO4��3��Һ�Ͱ�ˮ ��
��AgNO3��Һ�Ͱ�ˮ��
| A���٢ۢ� | B���٢ڢ� |
| C���ڢۢ� | D���ۢܢ� |
ij�������ӵ�����������Ϊ2���й�����������ȷ���ǣ�������
| A��һ���ڵڢ�A |
| B�������ǽ���Ԫ�� |
| C��һ��������Ԫ�ػ�0��Ԫ�� |
| D��ֻ����һ�����Ӳ� |