��Ŀ����
16����ҵ��ұ����ͭ��mCu2O•nFeS���ɵõ���ͭ���ڿ��������ձ�ͭʱ������һ���д̼�����ζ������A���������ŷŵ������У��ǵ���������γɵ���Ҫԭ��
���������գ�
��1��Ϊ�˽�����Ⱦ����A���ŷţ���ѡ�������Լ��е�cd������ţ����գ�
a��ŨH2SO4 b��ŨHNO3 c��NaOH��Һ d��ʯ����
��2����֪���ڸ������ܽ�ijЩ�����������������û���������������ͭ������Ӧ�Ӷ��õ���ͭ����ͭұ����ͭ�Ļ�ѧ����ʽ��3Cu2O+2Al$\frac{\underline{\;����\;}}{\;}$6Cu+Al2O3��ÿ����1mol���μӷ�Ӧ����ת�Ƶ��Ӹ���Ϊ3��6.02��1023
��3��SO2�������������Ʒ�Ӧʱ���õ�����ΪNa2SO3�� NaHSO3��ͨ������ش𣺽�0.3mol SO2ͨ��100mL��0.4molNaOH��Һ�У���ַ�Ӧ��õ��������ʵ����ֱ��Ƕ��٣���д��������̣�
���� ��1������AΪSO2�����ü�Һ���գ�
��2��Cu2O��Al�ڸ����·�Ӧ����Cu��Al2O3��Ϊ���ȷ�Ӧ���ɷ���ʽ��֪1mol�����μӷ�Ӧת�Ƶ��ӵ����ʵ���Ϊ3mol��
��3��0.3mol��SO2ͨ����������ȫ�����������������������Ƶ����ʵ���Ϊ��0.6mol����ȫת��Ϊ�������������������Ƶ����ʵ���Ϊ0.3mol����ʵ���������Ƶ����ʵ���Ϊ0.4mol�����Լ������������������������ƣ��ɴ˷�����⣮
��� �⣺��1������AΪSO2�����ü�Һ���գ���Ӧ��NaOH��ˮ���ʴ�Ϊ��cd��
��2��Cu2O��Al�ڸ����·�Ӧ����Cu��Al2O3����Ӧ�ķ���ʽΪ3Cu2O+2Al$\frac{\underline{\;����\;}}{\;}$6Cu+Al2O3���ɷ���ʽ��֪1mol�����μӷ�Ӧת�Ƶ��ӵ����ʵ���Ϊ3mol������ת�Ƶ�����Ϊ��3��6.02��1023���ʴ�Ϊ��3Cu2O+2Al$\frac{\underline{\;����\;}}{\;}$6Cu+Al2O3��3��6.02��1023��
��3��0.3mol��SO2ͨ����������ȫ�����������������������Ƶ����ʵ���Ϊ��0.6mol����ȫת��Ϊ�������������������Ƶ����ʵ���Ϊ0.3mol����ʵ���������Ƶ����ʵ���Ϊ0.4mol����Na2SO3�����ʵ���xmol��NaHSO3�����ʵ���Ϊ��ymol����$\left\{\begin{array}{l}{2x+y=0.4}\\{x+y=0.3}\end{array}\right.$����֮�ã�Na2SO3 �����ʵ���Ϊ0.1mol��NaHSO3�����ʵ���Ϊ0.2mol����Na2SO3 �����ʵ���Ϊ0.1mol��NaHSO3�����ʵ���Ϊ0.2mol��
���� �����ۺϿ����Ʊ�ʵ����漰�Լ����ʵķ����ᴿ֪ʶ���������ۺϿ���ѧ����ʵ�����������ݷ���������ע�����ʵ��ԭ����ʵ�鷽������Ŀ�Ѷ��еȣ�

| A�� | ���Ĺ̶������˹��̵�����Ȼ�̵� | B�� | ��ҵ�ϳɰ������˹��̵� | ||
| C�� | ��Ȼ�̵��е�Ԫ�ر����� | D�� | ���Ĺ̶��в���Ԫ������������ |
| A�� | ��FeCl2��NaI�Ļ����Һ�м������軯����Һ���ٵμ�������ˮ����Һû�б�Ϊ��ɫ��˵����ԭ�ԣ�Fe2+��I- | |
| B�� | ����AlNH4��SO4��2��Һ�е�NH4+��ȡ�����Թ��У�����Һ�м�������NaOH��Һ�����ȣ���ʪ��ĺ�ɫʯ����ֽ������������� | |
| C�� | ����Na2SO3��Na2SO4�Ļ����Һ�е�SO42-���ȼ�����ϡ���ᣬ�ټ��Ȼ�����Һ | |
| D�� | ��֤̼������Һ�Լ�����CO32-���µģ��ȼӷ�̪��Һ����Һ��죬�ټ�������Һ����ɫ��ȥ |
| A�� | MnO4-��Mn2+ | B�� | HCl��Cl2 | C�� | Fe��Fe2+ | D�� | KClO3��O2 |
| A�� | ��һ����������һ���ۣ��μ��ữ��ʳ��ˮ���ݼ�KSCN��Һ����Һ��ɫû�б仯���ж�����кõ���ʴ�� | |
| B�� | �ⶨһ�����ʵ���Ũ�ȵ�ijNaA��Һ��pH��pH��7������˵��HA������ | |
| C�� | ���Ȼ���������Һ�м���⻯����Һ��������ɫ���䣬˵���Ȼ������ܽ�ȱȵ⻯���� | |
| D�� | ������к͵ζ��У��ñ�����������Һ�ⶨ������Һ��Ũ��ʱ���ü�����ָʾ���Եζ��յ���жϱ��÷�̪�ĸ�ȷ |
| A�� | ���ö����ЧӦ��������Һ�ͽ��� | |
| B�� | ��ʹʪ��ļ�ɫʯ����ֽ����������NH3 | |
| C�� | �۲��Ԫ�ص���ɫ��ӦʱҪ����ɫ�ܲ��� | |
| D�� | ������Һ��SO42-����ʱ���ȼ������ữ���ټ��Ȼ�����Һ |
| A�� | 1molCl2������������Ӧת�Ƶĵ�������3NA | |
| B�� | ���ʵ���Ũ��Ϊ0.5mol/L��MgCl2��Һ�У�����Cl-����ΪNA | |
| C�� | 7.8g Na2O2�к�����������ĿΪ0.1NA��������ˮ��Ӧʱ����ת����0.1NA | |
| D�� | ��״����33.6L���Ȼ�̼���еķ�����Ϊ1.5 NA |
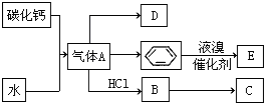 ��֪��C��һ�ֺϳ���֬�������Ʊ����Ϻͺϳ���ά��D��һ��ֲ���������ڼ����������Դ����ʵ���������»�ѧ��Ӧ��ͼ��գ�
��֪��C��һ�ֺϳ���֬�������Ʊ����Ϻͺϳ���ά��D��һ��ֲ���������ڼ����������Դ����ʵ���������»�ѧ��Ӧ��ͼ��գ�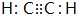 ��C�Ľṹ��ʽ
��C�Ľṹ��ʽ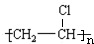 ��
�� +Br2 $\stackrel{FeBr_{3}}{��}$
+Br2 $\stackrel{FeBr_{3}}{��}$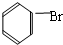 +HBr���䷴Ӧ����Ϊȡ����Ӧ��
+HBr���䷴Ӧ����Ϊȡ����Ӧ��