��Ŀ����
��14�֣���ѧ��ȤС���ijƷ��������Ħ�����ɷּ��京����������̽����
������ϣ�������Ħ������̼��ơ�����������ɣ������������ɷ���������ʱ�����������
��Ħ���������������Ķ��Լ��顣ȡ����������Ʒ����ˮ��ֽ��衢���ˡ�
��1���������м������NaOH��Һ�����ˣ���Һ��ͨ�����������̼����д��ͨ�����������̼��Ӧ�����ӷ���ʽ��___________��______________��
��������Ʒ��̼��ƵĶ����ⶨ��������ͼ��ʾװ��(ͼ�мг�������ȥ)����ʵ�飬��ַ�Ӧ�ⶨC �����ɵ�BaCO3������������ȷ��̼��Ƶ�����������

����ʵ����̻ش��������⣺
��2��ʵ����������������ͨ������������ó��˿ɽ���B��C�еķ�Ӧ���⣬����_________________
��3��C�з�Ӧ����BaCO3�Ļ�ѧ����ʽ��_____________________________________
��4�����и����ʩ�У�������߲ⶨȷ�ȵ���________(����)��
a���ڼ�������֮ǰ��Ӧ�ž�װ���ڵ�CO2����
b���μ�����˹���
c����A��B֮������ʢ��Ũ�����ϴ��װ��
d����B��C֮������ʢ�б���̼��������Һ��ϴ��װ��
��5��ʵ����ȷ��ȡ8.00 g��Ʒ���ݣ��������βⶨ�����BaCO3ƽ������Ϊ3.94 g������Ʒ��̼��Ƶ���������Ϊ______��
��6��������Ϊ���زⶨC�����ɵ�BaCO3������ֻҪ�ⶨװ��C������CO2ǰ���������Ϳ���ȷ��̼��Ƶ�����������ʵ��֤�����˷����ⶨ�Ľ������ƫ�ߣ�ԭ����________��
��1��OH����CO2=HCO3����AlO2-+CO2+2H2O��Al��OH��3��+HCO3-
��2���������ɵ�CO2����ȫ������C�У�ʹ֮��ȫ��Ba��OH��2��Һ����
��3��CO2+Ba��OH��2��BaCO3��+H2O ��4��cd ��5��25%
��6��B�е�ˮ�������Ȼ�������Ƚ���װ��C��
��������
�����������1���������ƹ��������������Һ���������ƺ�ƫ�����ƵĻ��Һ������������Һ��ͨ�����������̼��������ӦΪOH����CO2=HCO3����AlO2-+CO2+2H2O��Al��OH��3��+HCO3-��
��2��װ���в������ֶ�����̼�����ܱ���ȫ���գ����²ⶨ��̼�ᱵ������ƫС����������ͨ�����������Ϊ�������ɵ�CO2����ȫ������C�У�ʹ֮��ȫ��Ba��OH��2��Һ���գ��ʴ�Ϊ��
��3��������̼������������Ӧ����̼�ᱵ��ˮ����Ӧ����ʽΪCO2+Ba��OH��2��BaCO3��+H2O��
��4��a���ڼ�������֮ǰ��Ӧ�ž�װ���ڵ�CO2���壬��ֹӰ��̼�ᱵ�����IJⶨ��������߲ⶨȷ�ȣ���a�����ϣ�b���μ��������CO2��CO2�����������գ����ų�װ��C���μ�����˹��죬ʹ������̼������ȫ��������߲ⶨȷ�ȣ���b�����ϣ�c����AB֮������ʢ��Ũ�����ϴ��װ�ã�����ˮ�֣���Ӱ��CO2�����������߲ⶨȷ�ȣ���c���ϣ�d����BC֮������ʢ�б���̼��������Һ��ϴ��װ��Ba��OH��2����������CO2�е�HCl��Ӱ��CO2��������߲ⶨȷ�ȣ���d���ϣ���ѡ��cd��
��5��BaCO3����Ϊ3.94g����n��BaCO3����3.94g��197g/mol��0.02mol����n��CaCO3����0.02mol������Ϊ0.02mol��100g/mol��2g��������Ʒ��̼��Ƶ���������Ϊ��100%��25%��
��6��B�е�ˮ�������Ȼ�������Ƚ���װ��C�У����²ⶨ������̼������ƫ�ⶨ��̼��Ƶ�����ƫ��̼��Ƶ���������ƫ�ߡ�
���㣺�����ʵ��ԭ���������������⼰���ۡ����û�ѧ�����ѧ���㡢������ɵIJⶨ��

��֪�������ʵ����Ʒֱ���
���� | ��������� | ���� | �������� | �������� |
��ѧʽ | Na2S2O3 | CaSx | BaO2 | KO2 |
���������������ɻش�K2CS3Ӧ����Ϊ
A�������̼��� B������̼��� C������̼��� D������̼���
��14�֣�����������ͨ���÷��������������Ʊ����������ı����Ƚ��ȶ������������������ϵ�֧�����۳��̣���ǿ������ʱ�����ն��������Ȼ���ijͬѧ�üױ���������Ӧ�Ʊ������ᡣ
��Ӧԭ����
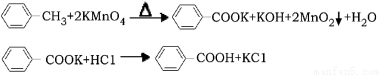
��Ӧ�Լ������������������
���� | ��Է� ������ | ��״ | �۵� | �е� | �ܶ� | �ܽ�� | ||
ˮ | �Ҵ� | ���� | ||||||
�ױ� | 92 | ��ɫҺ����ȼ�ӷ� | -95 | 110.6 | 0.8669 | ���� | ���� | ���� |
������ | 122 | ��ɫƬ״����״���� | 122.4 | 248 | 1.2659 | �� | ���� | ���� |
��Ҫʵ��װ�ú��������£�
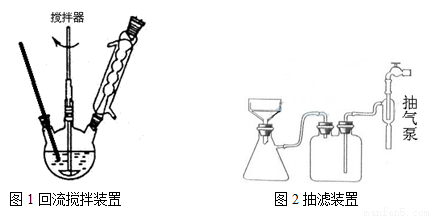
ʵ�鷽����һ�����ļױ���KMnO4��Һ����ͼ1װ���У���90��ʱ�� ��Ӧһ��ʱ�䣬��ֹͣ��Ӧ�����������̷����������ͻ���δ��Ӧ�ļױ���
��1����ɫҺ��A�Ľṹ��ʽΪ ��������Ϊ ��
��2�������Һ����ɫ��Ҫ�ȼ���������أ�Ȼ���ټ���Ũ�����ữ������������ص�Ŀ���� ��
��3�����й�����������װ����ʹ����ȷ���� ��
A�����˿��Լӿ�����ٶȣ��õ��ϸ���ij���
B����װ�綯������ʱ���������¶˲�����������ƿ�ס��¶ȼƵȽӴ�
C��ͼ1��������װ��Ӧ����ֱ�Ӽ��ȵķ���
D����������ˮ���������½��ϳ�
��4����ȥ�����ڱ������еļױ�Ӧ�ȼ��� ����Һ��ˮ���ټ��� ��Ȼ����ˣ����T�ɵõ������ᡣ
��5�����Ȳⶨ����ȡ1.220g��Ʒ�����100ml��Һ��ȡ����25.00ml��Һ�����еζ� ������KOH���ʵ���Ϊ2.4��10-3mol����Ʒ�б�������������Ϊ ��
 ���������Һ���Ƿ������������6�����ӣ���SiO
���������Һ���Ƿ������������6�����ӣ���SiO ����NH
����NH ����Fe2������Na������HCO
����Fe2������Na������HCO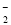 ��2H2O
��2H2O