��Ŀ����
3����֪��ѧƽ�⡢����ƽ�⡢ˮ��ƽ����ܽ�ƽ���������������ԭ������ش��������⣺��1�������£�Ũ�Ⱦ�Ϊ0.1mol•L-1������������Һ��pH���±���
| ���� | CH3COONa | NaHCO3 | Na2CO3 | NaClO | NaCN |
| pH | 8.8 | 9.7 | 11.6 | 10.3 | 11.1 |
�ڸ��ݱ��������жϣ�Ũ�Ⱦ�Ϊ0.01mol•L-1�������������ʵ���Һ�У�������ǿ����D��������Һ�ֱ�ϡ��100����pH�仯��С����A�����ţ���
A��HCN B��HClO C��H2CO3 D��CH3COOH
�۾��ϱ����ݣ������ж����з�Ӧ���ܳ�������C�����ţ���
A��CH3COOH+Na2CO3�TNaHCO3+CH3COONa
B��CH3COOH+NaCN�TCH3COONa+HCN
C��CO2+H2O+2NaClO�TNa2CO3+2HClO
��Ҫ������ˮ��HClO��Ũ�ȣ�������ˮ�м���������̼������Һ����Ӧ�����ӷ���ʽΪ2Cl2+CO32-+H2O=CO2��+2Cl-+2HClO��
��2����֪������Cu��OH��2��Ksp=2��10-20����֪������ijCuSO4��Һ��c��Cu2+��=0.02mol•L-1�����Ҫ����Cu��OH��2��������Ӧ������ҺpH����5��Ҫʹ0.2mol•L-1��CuSO4��Һ��Cu2+������Ϊ��ȫ��ʹCu2+Ũ�Ƚ���ԭ����ǧ��֮һ����Ӧ����Һ���NaOH��Һ��ʹ��ҺpHΪ6��
���� ��1�������������ˮ��̶�Խ����ͬŨ�ȵ�������Һ��pHԽ����������ӽ����������Խ��
���������Խǿ����ĵ���̶�Խ�����������ˮ��̶�ԽС����ͬŨ�ȵ�������Һ��pHԽС����ˮϡ�ʹٽ�������룬��ͬŨ�ȵIJ�ͬ��ϡ����ͬ�ı������������Խ�������PH�仯ԽС��
�۸���ǿ����ȡ�����жϣ�
��HClO��̼���Ʋ���Ӧ�������̼���Ʒ�Ӧ���ɶ�����̼��
��2������Ksp=c��Cu2+��•[c��OH-��]2=2��10-20������Cu2+���ӵ�Ũ�ȼ���OH-���ӵ�Ũ�ȣ�����ˮ�����ӻ���������������Ũ�ȣ����Դ˼�����ҺpHֵ��Cu2+������Ϊ��ȫŨ�Ƚ���ԭ����ǧ��֮һ������Ksp���OH-���ӵ�Ũ�ȣ�����pH��
��� �⣺��1�������������ˮ��̶�Խ����ͬŨ�ȵ�������Һ��pHԽ����������ӽ����������Խ������ҺpH֪��CO32-��ˮ��̶������CO32-�������������ǿ���ʴ�Ϊ��CO32-��
���������Խǿ����ĵ���̶�Խ�����������ˮ��̶�ԽС����ͬŨ�ȵ�������Һ��pHԽС�����ݱ�������֪��������ǿ�Ĵ��ᡢ��������������HCN����ˮϡ�ʹٽ�������룬��ͬŨ�ȵIJ�ͬ��ϡ����ͬ�ı������������Խ�������PH�仯ԽС����������������HCN����pH�仯��С����HCN����ѡ��D��A��
��A���������Դ���̼�ᣬ����CH3COOH+Na2CO3=NaHCO3+CH3COONa�ܷ������ʲ�ѡ��
B���������Դ��������ᣬ����CH3COOH+NaCN=CH3COONa+HCN�ܷ������ʲ�ѡ��
C������������С��̼�������̼��������ӣ�����CO2+H2O+NaClO=NaHCO3+HClO�ܷ�������ѡ��
�ʴ�Ϊ��C��
��HClO��̼���Ʋ���Ӧ�������̼���Ʒ�Ӧ���ɶ�����̼�����Ե�����ˮ��HClOŨ���������ӷ���ʽΪ2Cl2+CO32-+H2O=CO2��+2Cl-+2HClO��
�ʴ�Ϊ��2Cl2+CO32-+H2O=CO2��+2Cl-+2HClO��
��2��ijCuSO4��Һ��c��Cu2+��=0.02mol/L���������Cu��OH��2��������Ӧ��c��OH-����$\sqrt{\frac{2��1{0}^{-20}}{0.02}}$mol/L=10-9mol/L����c��H+����$\frac{1��1{0}^{-14}}{1{0}^{-9}}$mol/L=10-5mol/L��
����pH��-lg��10-5��=5��Cu2+������Ϊ��ȫ�ǵ�Ũ��Ϊ$\frac{0.2mol/L}{1000}$=2��10-4mol/L��
��c��OH-��=$\sqrt{\frac{2��1{0}^{-20}}{2��1{0}^{-4}}}$mol/L�T10-8mol/L��
��c��H+��=$\frac{1��1{0}^{-14}}{1{0}^{-8}}$mol/L=10-6mol/L��
����pH=-lg��10-6��=6��
�ʴ�Ϊ��5��6��
���� ���⿼����ƽ�ⳣ��������ˮ�⣬�ܶȻ������ļ��㣬��Ŀ�ۺ��Խ�ǿ���ѶȲ���ע����ռ��㹫ʽ�����á�H2CO3��HClO��HCO3-����ǿ����ϵ��

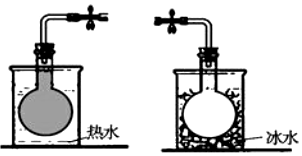 ���ܷ����ƿ�г���һ������NO2���������淴Ӧ��2NO2?N2O4����ͬ�¶�����ƿ���������ɫ��dz��ͼ��ʾ�������й�˵����ȷ���ǣ�������
���ܷ����ƿ�г���һ������NO2���������淴Ӧ��2NO2?N2O4����ͬ�¶�����ƿ���������ɫ��dz��ͼ��ʾ�������й�˵����ȷ���ǣ�������| A�� | �÷�Ӧ������ӦΪ���ȷ�Ӧ | |
| B�� | ��Ӧ�ﵽƽ��ʱ���ٳ�������NO2��ƽ���NO2��ת�������� | |
| C�� | ��Ӧ�ﵽƽ��ʱ���ٳ�������N2O4��������ɫ��dz | |
| D�� | ��Ӧ�ﵽƽ��ʱ������ѹǿ��ƽ��������Ӧ�����ƶ����淴Ӧ���ʼ�С |
| A�� | ������Br2��CCl4�м�NaOH��Һ����������������Ϊ��ɫ | |
| B�� | ��pH��ֽ�ϵμ���ˮ���ȱ����� | |
| C�� | �����ȵ�ͭ˿���뵽ʢ�������ļ���ƿ�У���������ɫ���� | |
| D�� | ����ˮ�м���NaHCO3���壬��������� |
| A�� | ��ͭ˿Ͷ��ϡ�����У�Cu+4 H++NO3-=Cu2++NO2��+2H2O | |
| B�� | Ũ������MnO2��Ӧ��ȡ������4H++2Cl-+MnO2$\frac{\underline{\;\;��\;\;}}{\;}$Cl2��+Mn2++2H2O | |
| C�� | �ö��Ե缫���MgCl2��Һ��2Cl-+2H2O$\frac{\underline{\;���\;}}{\;}$2OH-+Cl2��+H2�� | |
| D�� | ��NH4HCO3��Һ�мӹ�����NaOH��Һ�����ȣ�NH4++OH-$\frac{\underline{\;\;��\;\;}}{\;}$ NH3��+H2O |
| A�� | һ����Ag+ | B�� | һ����SO42- | C�� | ����Ag+��SO42- | D�� | ����Ag+��SO42- |

����˵��������ǣ�������
| A�� | ����������ʽ��������Һ��������ӦΪ2SO42--2e-�TS2O82- | |
| B�� | S2O82-����ǿ�����ԣ�H2S2O8�Ƕ�Ԫǿ�� | |
| C�� | ��ѹ�����Ŀ����Ϊ�˼���H2O2�ֽ⣬����õ�����һ��ֿ�ѭ������ | |
| D�� | ����ҺAת��Ϊ��ҺB������������ԭ��Ӧ |
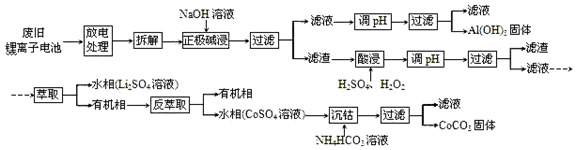
 A��B��C�ǵ��ʣ�����A�ǽ������������ʼ��ת����ϵ��ͼ��
A��B��C�ǵ��ʣ�����A�ǽ������������ʼ��ת����ϵ��ͼ��