��Ŀ����
7����ʯ����ʪ�����������ų��Ĺ�ҵ��������Ҫ�ɷ���CaSO4•2H2O����1��CaSO4•2H2O��ˮ��Ӧ��ص��Ȼ�ѧ����ʽΪ��
CaSO4•2H2O��s���TCaSO4•$\frac{1}{2}$H2O��s��+$\frac{3}{2}$H2O��g����H1=83.2kJ•mol-1
CaSO4•2H2O��s���TCaSO4��s��+2H2O��l����H2=26kJ•mol-1
H2O��g��=H2O��l����H3=-44kJ•mol-1
��ӦCaSO4•$\frac{1}{2}$H2O��s���TCaSO4��s��+$\frac{1}{2}$H2O��g���ġ�H4=30.8kJ•mol-1��
��2���ú��ʵĻ�ԭ�����Խ�CaSO4��ԭ������SO2�����ڹ�ҵ�������ᣮ
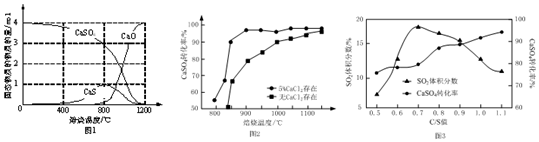
����CO����ԭ�����ı䷴Ӧ�¶ȿɵõ���ͬ�IJ����ͬ�¶��·�Ӧ�����ù���ɷֵ����ʵ�����ͼ1��ʾ���ڵ���800��ʱ��Ҫ��ԭ����ΪCaS������800��ʱ��Ҫ�����ķ�Ӧ�Ļ�ѧ����ʽΪCaS+3CaSO4$\frac{\underline{\;����\;}}{\;}$4CaO+4SO2����
���Ը���úΪ��ԭ������2.5 Сʱ����ͬ�����������ת���ʵ�Ӱ����ͼ2��ʾ��CaCl2�������������������¶ȸ���1200��ʱ����������CaCl2��CaSO4��ת����������ͬ����ԭ������������·�Ӧ���ﵽƽ�⣬����CaCl2���ı�ƽ����ƶ���
����C����ԭ�������ܱ������м�����ͬ�����ļ��鲻ͬ$\frac{C}{S}$ֵ��̿����CaSO4�����ʵ���֮�ȣ��Ļ������1100����ȣ������ͼ3��ʾ����$\frac{C}{S}$ֵΪ0.5ʱ����Ӧ����ΪCaO��SO2��CO2����$\frac{C}{S}$ֵ����0.7ʱ����Ӧ����������SO2�����������������������Ҫԭ������Ǹ����£�������C��CO2��Ӧ����CO��ʹ�������������ת��Ϊ�����������ʣ���
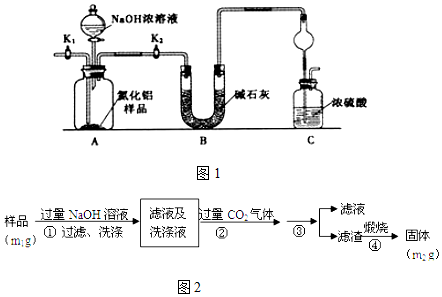
��3����֪Ksp��CaSO4��=7.10��10-5��Ksp��CaCO3��=6.2125��10-7�����÷�ӦCaSO4��s��+��NH4��2CO3��aq��?CaCO3��s��+��NH4��2SO4��aq�����Խ���ʯ��ת��Ϊ����泥�����Ӧ�ﵽƽ�����Һ��c��SO42-��=2.0mol•L-1����ʱ��Һ��c��CO32-��=1.75��10-2 mol•L-1��
���� ��1��������֪�Ȼ�ѧ����ʽ����Ŀ�귽��ʽ�����ݸ�˹���ɼ���õ�Ŀ�귽��ʽ�ķ�Ӧ�ȣ�
��2���ٴ�ͼʾ1��֪�ڵ���800��ʱ��Ҫ��ԭ����ΪCaS������800��ʱ��Ҫ�����ƺ�����Ʒ�Ӧ���������ƺͶ�������
�����Ȼ��ƺ����Ȼ��ƴ��ڵķ�Ӧ������ƽ����ͬ�����ı䷴Ӧ���̣�����CaCl2Ϊ���������¶ȸ���1200��ʱ���ﵽƽ�⣬��������ܸı�ƽ�⣬������������CaCl2��CaSO4��ת����������ͬ��
��̼�Ͷ�����̼�ڸ��������·�Ӧ��������һ����̼�����Ե�$\frac{C}{S}$ֵ����0.7ʱ����Ӧ����������SO2�������������������Ҳ������ת�����������ʣ�
��3����Ӧ�����ӷ���ʽΪCaSO4��s��+CO32-��aq��?SO42-��aq��+CaCO3��s��������Ksp��CaSO4��=c��Ca2+����c��SO42-����Ksp��CaCO3��=c��Ca2+����c��CO32-������c��CO32-����
��� �⣺��1����֪��CaSO4•2H2O��s���TCaSO4•$\frac{1}{2}$H2O��s��+$\frac{3}{2}$H2O��g����H1=83.2kJ•mol-1
��CaSO4•2H2O��s���TCaSO4��s��+2H2O��l����H2=26kJ•mol-1
��H2O��g��=H2O��l����H3=-44kJ•mol-1
����-�ۡ�2-�ٵ�CaSO4•$\frac{1}{2}$H2O��s���TCaSO4��s��+$\frac{1}{2}$H2O��g�������H4=[26kJ•mol-1-��-44kJ•mol-1����2-83.2kJ•mol-1]=30.8kJ•mol-1��
�ʴ�Ϊ��30.8��
��2���ٴ�ͼʾ1��֪���ڵ���800��ʱ��CaS�ɷֵ����ʵ���Ϊ1����CaO�����ʵ�������Ϊ0�������ڵ���800��ʱ��Ҫ��ԭ����ΪCaS������800��ʱ��Ҫ�����ƺ�����Ʒ�ӦCaS+3CaSO4$\frac{\underline{\;����\;}}{\;}$4CaO+4SO2���������Ƶ����ʵ������ӣ��Ƶ����ʵ�����С��
�ʴ�Ϊ��CaS��CaS+3CaSO4$\frac{\underline{\;����\;}}{\;}$4CaO+4SO2����
�ڴ������ܸı䷴Ӧ�����ʣ��ı䷴Ӧ���̣������ܸı仯ѧƽ�⣬��ͼʾ��֪�����¶ȸ���1200��ʱ����������CaCl2��CaSO4��ת����������ͬ������CaCl2�������������������¶ȸ���1200��ʱ����������CaCl2��CaSO4�ķ�Ӧ��������·�Ӧ���ﵽƽ�⣬����CaCl2���ı�ƽ����ƶ���
�ʴ�Ϊ������������������·�Ӧ���ﵽƽ�⣬����CaCl2���ı�ƽ����ƶ���
�۵�$\frac{C}{S}$ֵ����0.7ʱ�������£�������C��CO2��Ӧ����CO���壬��Ӧ����ΪCaO��SO2��CO2��CO��ʹ�������������Ҳ��������Ʋ���ת��Ϊ�����������ʣ�
�ʴ�Ϊ�������£�������C��CO2��Ӧ����CO��ʹ�������������ת��Ϊ�����������ʣ���
��3����Ӧ�����ӷ���ʽΪCaSO4��s��+CO32-��aq��?SO42-��aq��+CaCO3��s��������Ӧ�ﵽƽ�����Һ��c��SO42-��=2.0mol•L-1����֪Ksp��CaSO4��=c��Ca2+����c��SO42-��=7.10��10-5��c��Ca2+��=3.55��10-5mol•L-1��Ksp��CaCO3��=6.2125��10-7��Ksp��CaCO3��=c��Ca2+����c��CO32-����c��CO32-��=1.75��10-2 mol•L-1��
�ʴ�Ϊ��1.75��10-2 mol•L-1��
���� ���⿼�鷴Ӧ�ȵļ��㡢��ѧƽ���ƶ�Ӱ�����ء������ܽ�ƽ��ļ��㣬���ո�˹���ɵ�����Ӧ���Լ�������ƽ���Ӱ���ǽ��ؼ�����Ŀ�Ѷ��еȣ�

| A�� | CO2H2S | B�� | NH3 HCl | C�� | P4 CCl4 | D�� | C2H4 CH4 |
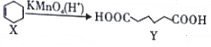 ������˵��������ǣ�������
������˵��������ǣ�������| A�� | X����������̼ԭ��һ����ͬһƽ���� | |
| B�� | X����ϩ������ͬϵ�� | |
| C�� | Y�ɷ���ȡ����Ӧ��������Ӧ | |
| D�� | Y�ĺ�����ͬ�����ŵ�ͬ���칹����8�֣������������칹�� |
| ѡ�� | �缫��Ӧʽ | ���ֵĻ��� |
| A | O2+2H2O+4e-�T4OH- | ���Ի���������ȼ�ϵ�صĸ�����Ӧ |
| B | 4OH--4e-�TO2��+2H2O | �����Ի����¸�����������ʴ |
| C | Cu-2e-�TCu2+ | ��ͭ���缫���NaOH��Һ��������Ӧ |
| D | H2-2e-�T2H+ | �ö��Ե缫���H2SO4��Һ��������Ӧ |
| A�� | A | B�� | B | C�� | C | D�� | D |
 ��֪��298K��101kPa�����£������·�Ӧ��
��֪��298K��101kPa�����£������·�Ӧ����Ӧ��C��s��+O2��g��=CO2��g����H1=-393.5kJ•mol-1
��Ӧ��2C��s��+O2��g��=2CO��g����H2=-221kJ•mol-1
��Ӧ��N2��g��+O2��g��=2NO��g����H3=+180.5kJ•mol-1
�Իش��������⣺
��1������β������ԭ��Ϊ��Ӧ����2NO��g��+2CO��g��?N2��g��+2CO2��g����H=-746.5 kJ•mol-1���÷�Ӧ���Է����е������ǵ��£�������¡��������¡��������¶ȡ�����
��2�������һ���¶��£����Ϊ 2 �����ܱ������з�����ѧ��Ӧ����0��4min���������ʵ����ı仯���±���ʾ��
| ���ʣ�mol�� ʱ�� | NO | CO | N2 | CO2 |
| ��ʼ | 0.40 | 1.0 | ||
| 2min ĩ | 2.0 | 0.80 | 1.6 | |
| 4min ĩ | 1.6 |
���Լ�����¶��·�Ӧ���Ļ�ѧƽ�ⳣ�� K=1.6��
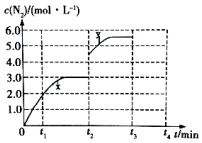
��3����һ���¶��£����ݻ��ɱ���ܱ������У�������Ӧ���ﵽƽ��״̬����ʱ�ݻ�Ϊ3L��c��N2����ʱ�� t �ı仯���� x ��ͼ��ʾ��
������ t2minʱ�ı�һ��������c��N2���淴Ӧʱ�� t �ı仯������ y ��ʾ����ı�������ǿ��ٽ����������3Lѹ����2L��
������t2minʱ�����¶ȣ�t3minʱ���´ﵽƽ�⣬����ͼ�л����� t2��t4�� c��N2���ı仯���ߣ�
| A�� | Ϊ��ȥ����CuCl2��Һ�к��е�����Fe3+�����ڼ��ȡ�����������¼��백ˮ�����˺�������Һ�м����������� | |
| B�� | �ڹ�ҵ�ϳ�NH3�ķ�Ӧ�У���ȡ���´�ʩ | |
| C�� | �����£���ƽ����ϵH2��g��+Br2��g��?2HBr��g����ѹ����ʹ������ɫ���� | |
| D�� | ���Ȼ�����Һ�в��ϻ�����ͨ��HCl���壬���������� |
| A�� | �����ĵ���ʽ��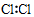 | B�� | �����Ľṹʽ��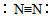 | ||
| C�� | Fһ�Ľṹʾ��ͼ�� | D�� | H2O2�ĵ���ʽ��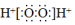 |