��Ŀ����
19�������й����ʼ����ʵ�������ȷ���ǣ�������| ѡ�� | ʵ����������� | ʵ����� |
| A | ��ij��Һ�м��������ữ���Ȼ�����Һ���а�ɫ�������� | ����Һ��һ������SO42- |
| B | ��Fe��NO3��2��Ʒ����ϡH2SO4���μ�KSCN��Һ��Һ��� | Fe��NO3��2��Ʒ���������� |
| C | ��ʢ������NaHCO3���Թ��еμӲ�����Һ�����ݲ��� | ���ԣ����̼�� |
| D | ������ij���ʵ�ϡ��Һ�еμ�ϡ���ᣬ��������ʹ����ʯ��ˮ����ǵ����� | ������һ����̼���� |
| A�� | A | B�� | B | C�� | C | D�� | D |
���� A����ɫ����ΪAgCl�����ᱵ��
B��Fe��NO3��2��Ʒ����ϡH2SO4������������ԭ��Ӧ���������ӣ�
C������ǿ����ȡ����ķ�Ӧ��
D����ʹ����ʯ��ˮ����ǵ�����Ϊ������̼���������
��� �⣺A����ɫ����ΪAgCl�����ᱵ����ԭ��Һ�п��ܺ������ӻ���������ӣ������߲���ͬʱ���ڣ���A����
B��Fe��NO3��2��Ʒ����ϡH2SO4������������ԭ��Ӧ���������ӣ����ű��ʵļ��飬Ӧ����ˮ����B����
C������ǿ����ȡ����ķ�Ӧ������Ϊ������̼�������ԣ����̼�ᣬ��C��ȷ��
D����ʹ����ʯ��ˮ����ǵ�����Ϊ������̼���������������ʲ�һ����̼���Σ���D����
��ѡC��
���� ���⿼�黯ѧʵ�鷽�������ۣ�Ϊ��Ƶ���㣬�������ʵ����ʡ����Ӽ��顢��Ӧԭ����ʵ�鼼��Ϊ���Ĺؼ������ط�����ʵ�������Ŀ��飬ע��Ԫ�ػ�����֪ʶ��ʵ��Ľ�ϣ���Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ���Ͱ�ͨ�������Сѧ��ʱͬ�����ϵ�д�
���Ͱ�ͨ�������Сѧ��ʱͬ�����ϵ�д� ���Ͱ�ͨ������ϵ�д�
���Ͱ�ͨ������ϵ�д� �ٷ�ѧ����ҵ��������ϵ�д�
�ٷ�ѧ����ҵ��������ϵ�д�
�����Ŀ
9��ijѧϰС�飬��ϡHNO3�����ʯ��Ӧ������������С�ķ�����̽��Ӱ�췴Ӧ���ʵ����أ�����HNO3Ũ��Ϊ1.00mol•L-1��2.00mol•L-1������ʯ��ϸ������ֿ������ֹ��ʵ���¶�Ϊ298K��308K��ÿ��ʵ��HNO3������Ϊ25.00mL������ʯ����Ϊ10.00g��
ʵ����������
����Ӧ��ʵ��Ŀ���������пո��У�
��1��ʵ��ٺ͢�̽��Ũ�ȶԸ÷�Ӧ���ʵ�Ӱ�죻
��2��ʵ��ٺ͢�̽���¶ȶԸ÷�Ӧ���ʵ�Ӱ�죻
��3��ʵ��ٺ͢�̽�����������С�Ը÷�Ӧ���ʵ�Ӱ�죮
ʵ����������
| ��� | T/K | ����ʯ��� | HNO3Ũ�� |
| �� | 298 | �ֿ��� | 2.00 mol•L-1 |
| �� | 298 | �ֿ��� | 1.00 mol•L-1 |
| �� | 308 | �ֿ��� | 2.00 mol•L-1 |
| �� | 298 | ϸ���� | 2.00 mol•L-1 |
��1��ʵ��ٺ͢�̽��Ũ�ȶԸ÷�Ӧ���ʵ�Ӱ�죻
��2��ʵ��ٺ͢�̽���¶ȶԸ÷�Ӧ���ʵ�Ӱ�죻
��3��ʵ��ٺ͢�̽�����������С�Ը÷�Ӧ���ʵ�Ӱ�죮
10���ó������ⶨKHCO3��Na2CO3�����������ɣ�ÿ�γ�ȡһ����������Ʒ����ˮ�Ƴ���Һ�������еμ���ͬŨ�ȵ�Ba��OH��2��Һ��ÿ��ʵ�����ַ�Ӧ����Ӧǰ����Һ����仯���Բ��ƣ�ʵ���¼������
�ش��������⣺
��1����Ʒ��KHCO3��Na2CO3�����ʵ���֮��2��1��
��2�������µڢ���ʵ��������Һ�е�OH-���ʵ���Ũ��Ϊ0.2mol/L��
| ʵ����� | �� | �� | �� | �� | �� |
| ����������g�� | 3.06 | 6.12 | 9.18 | 12.24 | 15.30 |
| Ba��OH��2��Һ�����mL�� | 300 | 300 | 300 | 300 | 300 |
| ���ɳ�����������g�� | 5.19 | 11.82 | 17.73 | 23.64 | 23.64 |
��1����Ʒ��KHCO3��Na2CO3�����ʵ���֮��2��1��
��2�������µڢ���ʵ��������Һ�е�OH-���ʵ���Ũ��Ϊ0.2mol/L��
7���������ӷ�Ӧ����ʽ����д��ȷ���ǣ�������
| A�� | ϡ���������Ƭ�ϣ�2Fe+6H+�T2Fe3++3H2�� | |
| B�� | ����ͭ��Һ������������Һ��ϣ�CuSO4+2OH-�TCu��OH��2+SO42-������þ��ϡ�����ϣ�MgO+2H+�TMg2++H2O | |
| C�� | ���������Ȼ�����Һ��Ӧ��AgNO3+Cl-�TAgCl��+NO3- | |
| D�� | ̼��������Һ��ϡ������Һ��ϣ�HCO3-+H+=H2O+CO2�� |
14����һ�������£��ֱ��Ը�����ء�����ء���������Ϊԭ����ȡ���������Ƶ�ͬ�¡�ͬѹ����ͬ���������ʱ��������Ӧ��ת�Ƶĵ�����֮��Ϊ��������
| A�� | 1��1��l | B�� | 2��2��1 | C�� | 1��1��2 | D�� | 4��3��2 |
4���������������������Ʊ����ػ��Դ�������Ҫԭ�ϣ�������110��ʱʧȥ�ᾧˮ��230��ʱ�ֽ⣮���й⣬�����¹��ձ�Ϊ��ɫ��
2K3[Fe��C2O4��3]•3H2O��2FeC2O4•2H2O+3K2C2O4+2CO2��+2H2O
��֪��
ijͬѧ�����������������ص��Ʊ�����ɽ�����ʵ��̽�����ش��������⣺
��һ�����������������ص��Ʊ�
��������ȡ4.50g FeC2O4•2H2O���壬���뱥��K2C2O4��Һ�У��õι���������3% H2O2��Һ�����Ͻ��裬�������¶���40�����ң���ַ�Ӧ����ת��Ϊ����������
���������������ڣ��μ�1mol �IJ�����Һ�������ܽ⣮�ò�������ص�����ҺpH=4-5
������С�����Ũ������ȴ�ᾧ�����ˣ�����������ˮϴ�ӣ�������ˮ�Ҵ�ϴ�ӣ����¸����������õ�9.80g ����ɫ���壮
��1����ɲ��裨i��������Ӧ�����ӷ���ʽ��
6FeC2O4•2H2O+6 C2O42-+3 H2O2�T2Fe��OH��3+4[Fe��C2O4��3]3-+12H2O
��2�����裨�����У�����ˮ�Ҵ�ϴ�Ӿ����Ŀ���ǣ�
��3����ʽ���㱾ʵ�������������������صIJ��ʣ�
����������������ඨ
ȷ��ȡ4.80g ������������������Ʒ�����Ƴ�250mL ��Һ����ȡ25.00mL ��Һ������ƿ�У�����5.0mL 1mol/L H2SO4����0.06mol/L��KMnO4��Һ�ζ����ζ��յ�ʱ����KMnO4��Һ20.00mL��
��4���ζ������У�ʢ�Ÿ��������Һ��������������ʽ�ζ��ܣ��ζ�ʱ������Ӧ�����ӷ���ʽΪ5C2O42-+2MnO4-+16H+=10CO2��+2Mn2++8H2O��5[Fe��C2O4��3]3-+6MnO4-+48H+=30CO2��+6Mn2++24H2O+5Fe3+��
��5����Ʒ�в���������ʵ���Ϊ0.03mol��������ֵ����4.80g���������������ش�Ʒ�в���������ʵ�������ԭ�������ac������ĸ����
a����ȡ�����в����������������
b����Ʒδ��ȫ����
c���ζ�ʱʢ��KMnO4��Һ������δ�ñ�Һ��ϴ
d����ƿϴ����δ���
������ijͬѧ�Ƶõ����������������أ�������淢�ƣ��Ʋ�����в��־��巢���˷ֽ⣮
��6��Ϊ����֤���Ʋ��Ƿ���ȷ����ѡ�õ��Լ����ữ��K3[Fe��CN��6]�����軯�أ���Һ��
2K3[Fe��C2O4��3]•3H2O��2FeC2O4•2H2O+3K2C2O4+2CO2��+2H2O
��֪��
| �������� | ��ѧʽ | ��Է������� | ��ɫ | �ܽ��� |
| ���������������� | K3[Fe��C2O4��3]•3H2O | 491 | ����ɫ | ������ˮ���������Ҵ� |
| �������� | FeC2O4•2H2O | 180 | ��ɫ | ������ˮ���������� |
��һ�����������������ص��Ʊ�
��������ȡ4.50g FeC2O4•2H2O���壬���뱥��K2C2O4��Һ�У��õι���������3% H2O2��Һ�����Ͻ��裬�������¶���40�����ң���ַ�Ӧ����ת��Ϊ����������
���������������ڣ��μ�1mol �IJ�����Һ�������ܽ⣮�ò�������ص�����ҺpH=4-5
������С�����Ũ������ȴ�ᾧ�����ˣ�����������ˮϴ�ӣ�������ˮ�Ҵ�ϴ�ӣ����¸����������õ�9.80g ����ɫ���壮
��1����ɲ��裨i��������Ӧ�����ӷ���ʽ��
6FeC2O4•2H2O+6 C2O42-+3 H2O2�T2Fe��OH��3+4[Fe��C2O4��3]3-+12H2O
��2�����裨�����У�����ˮ�Ҵ�ϴ�Ӿ����Ŀ���ǣ�
��3����ʽ���㱾ʵ�������������������صIJ��ʣ�
����������������ඨ
ȷ��ȡ4.80g ������������������Ʒ�����Ƴ�250mL ��Һ����ȡ25.00mL ��Һ������ƿ�У�����5.0mL 1mol/L H2SO4����0.06mol/L��KMnO4��Һ�ζ����ζ��յ�ʱ����KMnO4��Һ20.00mL��
��4���ζ������У�ʢ�Ÿ��������Һ��������������ʽ�ζ��ܣ��ζ�ʱ������Ӧ�����ӷ���ʽΪ5C2O42-+2MnO4-+16H+=10CO2��+2Mn2++8H2O��5[Fe��C2O4��3]3-+6MnO4-+48H+=30CO2��+6Mn2++24H2O+5Fe3+��
��5����Ʒ�в���������ʵ���Ϊ0.03mol��������ֵ����4.80g���������������ش�Ʒ�в���������ʵ�������ԭ�������ac������ĸ����
a����ȡ�����в����������������
b����Ʒδ��ȫ����
c���ζ�ʱʢ��KMnO4��Һ������δ�ñ�Һ��ϴ
d����ƿϴ����δ���
������ijͬѧ�Ƶõ����������������أ�������淢�ƣ��Ʋ�����в��־��巢���˷ֽ⣮
��6��Ϊ����֤���Ʋ��Ƿ���ȷ����ѡ�õ��Լ����ữ��K3[Fe��CN��6]�����軯�أ���Һ��
8����״���£����й��������˵������ȷ���ǣ�������
| A�� | 2mol������1mol���������������ԼΪ2��1 | |
| B�� | 2mol������1mol��������������ԼΪ1��1 | |
| C�� | 1mol������3mol��������������ԼΪ1��3 | |
| D�� | 1mol������3mol����������ܶȱ�ԼΪ1��3 |
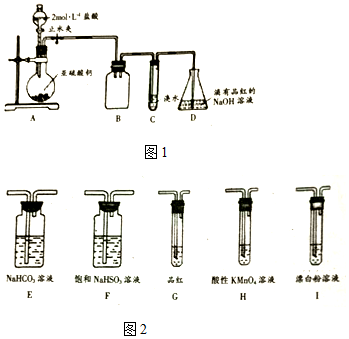
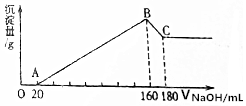 ��һ��������þ�����Ͻ�Ͷ��200mLһ��Ũ�ȵ������У��Ͻ���ȫ�ܽ⣬��������Һ�еμ�5mol/L NaOH��Һ�����ɵij������������NaOH��Һ�����ͼ��ʾ��
��һ��������þ�����Ͻ�Ͷ��200mLһ��Ũ�ȵ������У��Ͻ���ȫ�ܽ⣬��������Һ�еμ�5mol/L NaOH��Һ�����ɵij������������NaOH��Һ�����ͼ��ʾ��