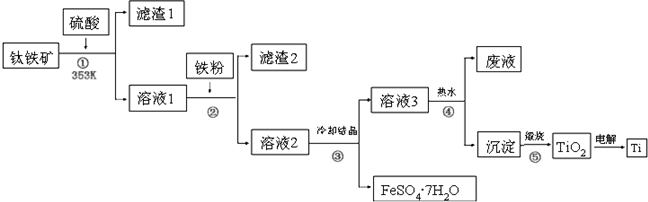
��Ŀ����
8������������SnSO4����һ����Ҫ�������Σ��㷺Ӧ���ڶ�����ҵ��ij�о�С�����SnSO4�Ʊ�·����ͼ��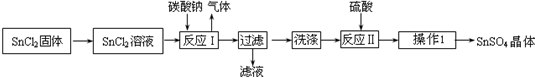
�������ϣ�
I�����������£�����ˮ��Һ����Sn2+��Sn4+������Ҫ������ʽ��
��Sn2+��������Fe2+���������ƣ�
�ش��������⣺
��1����Ԫ�������ڱ��е�λ���ǵ������ڵڢ�A�壮
��2����SnCl2��������SnCl2��Һ�ķ����ǽ�SnCl2��������Ũ���ᣬ��ˮϡ��������Ũ�ȣ��ټ�������Sn�ۣ�
��3����ӦI�õ�������SnO���÷�Ӧ�����ӷ���ʽ��Sn2++CO32-�TSnO��+CO2����
��4��ϴ��SnO�����ķ�������������м�������ˮ����û��������ˮ��ȫ�������ظ�����2-3�Σ�
��5������I�Ǽ���Ũ������ȴ�ᾧ�����ˡ�ϴ�ӣ�
��6�����������£�SnSO4����������˫��ˮȥ������������Ӧ�����ӷ���ʽ��Sn2++H2O2+2H+�TSn4++2H2O��
��7����С��ͨ�����з����ⶨ����SnCl2����Ĵ��ȣ����ʲ����뷴Ӧ����
��ȡ6.10g SnCl2�������Ƴ�100mL ��Һ��
��ȡ25.00mL��Һ������Һ�м��������FeCl3���壻
������0.10mol/L��K2Cr2O7����Һ���еζ������յ�ʱ��¼����K2Cr2O7����Һ�������
���ظ����Тڡ��������������ⶨ����K2Cr2O7����Һ��ƽ�����Ϊ25.00mL��
�Լ���SnCl2����Ĵ��ȣ�д��������̣���
���� SnCl2�ܽ�õ���Һ����̼���Ƴ��������ӣ����˵õ�����ϴ�Ӻ���������ܽ�õ���������Һ������Ũ����ȴ�ᾧ������ϴ�ӵõ����������壮
��1����ԭ�ӵĺ˵����Ϊ50����̼Ԫ������ͬһ���壬���ڢ�A�壬����ԭ��������ȥ����������Ԫ������ȷ�����ڵ����ڣ�
��2������Ϣ��֪��SnCl2��ˮ�����ɼ�ʽ�Ȼ��������������ᣬ����Sn2+ˮ�⣬Sn2+�ױ�����������Sn�۳�������ҺpH�⣬����ֹSn2+��������
��3����Ӧ��õ�������SnO��SnԪ�ػ��ϼ�Ϊ�仯�����ڷ�������ԭ��Ӧ��ͬʱ�������壬������Ϊ������̼��
��4��ϴ�ӳ����ڹ���װ���н��У�
��5��������Һ��þ���ķ���������Ũ������ȴ�ᾧ�����ˡ�ϴ�����ش�
��6�����������£�SnSO4����������˫��ˮȥ������˫��ˮ��ǿ�����ԣ���Sn2+�ױ�����ΪSn4+����������ԭΪˮ��
��7�����ݵ���ת���غ��뷽��ʽ�ɵù�ϵʽSn2+��2Fe3+��2Fe2+��$\frac{1}{3}$K2Cr2O7���ݴ˼��㣮
��� �⣺��1����Ԫ����̼Ԫ������ͬһ���壬���ڢ�A�壬ԭ�Ӻ˵����Ϊ50����50-2-8-8-18=14����Sn���ڵ������ڣ��������ڱ��е�λ��Ϊ�������ڵڢ�A�壬
�ʴ�Ϊ���������ڵڢ�A�壮
��2������Ϣ��֪��SnCl2ˮ�⣬����SnCl2+H2O?Sn��OH��Cl+HCl���������ᣬʹ��ƽ�������ƶ�������Sn2+ˮ�⣬Sn2+�ױ�����������Sn�۳�������ҺpH�⣬����ֹSn2+����������SnCl2��������SnCl2��Һ�ķ����ǣ���SnCl2��������Ũ���ᣬ��ˮϡ��������Ũ�ȣ��ټ�������Sn�ۣ�
�ʴ�Ϊ����SnCl2��������Ũ���ᣬ��ˮϡ��������Ũ�ȣ��ټ�������Sn�ۣ�
��3����Ӧ��õ�������SnO��SnԪ�ػ��ϼ�Ϊ�仯�����ڷ�������ԭ��Ӧ��ͬʱ�������壬������Ϊ������̼�����ӷ���ʽΪ��Sn2++CO32-�TSnO��+CO2����
�ʴ�Ϊ��Sn2++CO32-�TSnO��+CO2����
��4��ϴ��SnO�����ķ��������ù���װ�ý���ϴ�ӣ���������м�������ˮ����û��������ˮ��ȫ�������ظ�����2-3�Σ�
�ʴ�Ϊ����������м�������ˮ����û��������ˮ��ȫ�������ظ�����2-3�Σ�
��5������������Һ��þ��壬��ȡ�ķ����ǣ�����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӣ��ʴ�Ϊ������Ũ������ȴ�ᾧ�����ˡ�ϴ�ӣ�
��6�����������£�SnSO4����������˫��ˮȥ������˫��ˮ��ǿ�����ԣ���Sn2+�ױ�����ΪSn4+����������ԭΪˮ�����ӷ���ʽΪ��Sn2++H2O2+2H+�TSn4++2H2O��
�ʴ�Ϊ��Sn2++H2O2+2H+�TSn4++2H2O��
��7����ʵ����̿�֪�������ķ�ӦΪ��2Fe3++Sn2+=Sn4++2Fe2+��6Fe2++Cr2O72-+14H+=6Fe3++2Cr3++7H2O����
Sn2+��2Fe3+��2Fe2+��$\frac{1}{3}$K2Cr2O7
1 $\frac{1}{3}$
n 0.0250L��0.100mol/L
���n=0.0075mol��
100ml��Һ�к���SnCl2 ���ʵ���=0.0075mol��$\frac{100}{25}$=0.03mol��
SnCl2����Ĵ���=$\frac{190g/mol��0.03mol}{6.1g}$��100%=93.44%��
��SnCl2����Ĵ���Ϊ93.44%��
���� ���⿼�������ʷ���ķ�����ʵ�������ˮ�����Ӧ�ã��ζ�ʵ��ļ����жϺͼ���Ӧ�ã����ջ����ǹؼ�����Ŀ�Ѷ��еȣ�

 һ����ʦ�����Ծ�ϵ�д�
һ����ʦ�����Ծ�ϵ�д�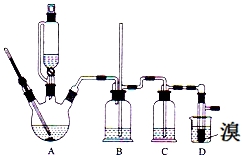 ʵ����������������������Ҵ���Ũ�����Ʊ�1��2-���������װ����ͼ��ʾ�����п��ܴ��ڵ���Ҫ����Ӧ�У��Ҵ���Ũ����Ĵ�������140����ˮ�������ѣ��й������б����£�
ʵ����������������������Ҵ���Ũ�����Ʊ�1��2-���������װ����ͼ��ʾ�����п��ܴ��ڵ���Ҫ����Ӧ�У��Ҵ���Ũ����Ĵ�������140����ˮ�������ѣ��й������б����£�| �Ҵ� | 1��2-�������� | ���� | |
| ״̬ | ��ɫҺ�� | ��ɫҺ�� | ��ɫҺ�� |
| �ܶ�/g•cm-3 | 0.79 | 2.2 | 0.71 |
| �е�/�� | 78.5 | 132 | 34.6 |
| �۵�/�� | -l30 | 9 | -1l6 |
��1��������������������Ҵ���Ũ�����Ʊ�1��2-�������������������У��ڶ�����Ӧ�Ļ�ѧ����ʽΪCH2=CH2+Br2��CH2BrCH2Br��
��2���ڴ�ʵ���У�Ҫ������Ѹ�ٵذѷ�Ӧ�¶���ߵ�170�����ң�������ҪĿ����d������ȷѡ��ǰ����ĸ����
a��������Ӧ b���ӿ췴Ӧ�ٶ� c����ֹ�Ҵ��ӷ� d�����ٸ�������������
��3����װ��C��Ӧ����c����Ŀ�������շ�Ӧ�п������ɵ��������壨����ȷѡ��ǰ����ĸ����
a��ˮ b��Ũ���� c������������Һ d������̼��������Һ
��4����1��2-��������ֲ�Ʒ���ڷ�Һ©���м�ˮ�����ã�����Ӧ���²㣨��ϡ������¡�����
��5����������������δ��Ӧ��Br2�������bϴ�ӳ�ȥ������ȷѡ��ǰ����ĸ����
a��ˮ b������������Һ c���⻯����Һ d���Ҵ�
��6�������������������������ѣ���������ķ�����ȥ��
��7����Ӧ������Ӧ����ˮ��ȴװ��D������ҪĿ������ϩ���巴Ӧʱ���ȣ���ȴ�ɱ�����Ĵ����ӷ������ֲ��ܹ�����ȴ�����ñ�ˮ������ԭ������ȴ�ɱ�����Ĵ����ӷ���1��2-������������̵�ϵͣ�9�棩��������ȴ��ʹ�����̶�ʹ��·������
���������գ�
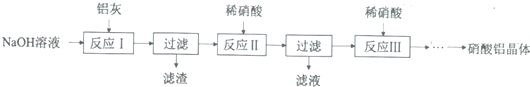
��1����NaOH��������30%��NaOH��Һ������IJ����������ձ��⣬����b��
a����ƿ b��Ͳ c����ƿ
��2����Ӧ����Ϊ����������ʧ����Ҫ�������������ο��Ʒ�Ӧ�յ㣨���������������
��3���ӷ�Ӧ��������Һ�еõ�����������IJ��������ǣ���ѹ��������ȴ�ᾧ�����ˡ�ϴ�ӡ����º�ɣ�
��4�����˽��齫��Ӧ��ϲ��Լ�����˵����ҵ�ϲ��������ַ�����ԭ�����������NaNO3�ĺ����ϸߣ�
��5��ijͬѧ��ʵ�����������Ƹ����������壬���������£�
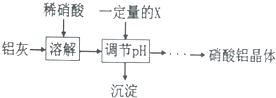
| �������� | Fe��OH��3 | Al��OH��3 |
| ��ʼ����pH | 1.9 | 4.2 |
| ������ȫpH | 3.2 | 5.4 |
Ϊʹ�õ�������������ϴ��������õ�X���ʿ�����BC�����ţ���
A����ˮ B���� c���������� D��ƫ������
��6����ʵ�����Ʒ��빤ҵ���Ʒ���ȣ���ȱ���Dz���������Ⱦ������NO�ȣ�
��7����ȡ7.392g������������Ʒ����������ʹ��ֽ���ȫ�����յõ�1.020gAl2O3������������������Ʒ�нᾧˮ����ĿΪ8.7��
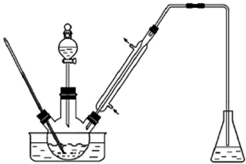 ʵ�����Ա���ȩΪԭ���Ʊ����屽��ȩ��ʵ��װ����Ҫ������ͼ��������ʵķе��������������������ʵķе㣨101kPa��
ʵ�����Ա���ȩΪԭ���Ʊ����屽��ȩ��ʵ��װ����Ҫ������ͼ��������ʵķе��������������������ʵķе㣨101kPa��| ���� | �е�/�� | ���� | �е�/�� |
| �� | 58.8 | 1��2-�������� | 83.5 |
| ����ȩ | 179 | ���屽��ȩ | 229 |
����2������Ӧ����ﻺ������һ������ϡ�����У����衢���á���Һ��
����3���л�����10%NaHCO3��Һϴ�ӣ��ټ���������ˮMgSO4���壬����һ��ʱ�����ˣ�
����4����ѹ�����л��࣬�ռ���Ӧ��֣�
��1��ʵ��װ���п��ܵĴ�������ƿ�ڿ��ܻᷢ���������������ܱ��л��︯ʴ�������ܽ�ˮ����a��
��2����ͬѧ���齫װ�����¶ȼƻ��ɽ��������ô�¶ȼ�Ӧ�Ƶ�ʲô�ط�ʹ��ˮԡ�У�
��3������2���������ǽ��衢���á���Һ��
��4������3����10%NaHCO3��Һϴ�ӵľ���������ڷ�Һ©���н��л�����С�մ���Һ������ٷ�Һ���ظ��������Σ�������ˮMgSO4����������dz�ȥ�л����ˮ��
��5������4�в���������룬����Ԥ�������¶�Ӧ��ѡ��ԼD�棬��ѡ��ԼE�森
A��50 B��100 C��150 D��200 E��250 F��300��
 S2Cl2��һ�ֽ��ɫ�ӷ���Һ�壬������������ij��ѧ��ȤС�� �����ʵ���Ʊ�������S2Cl2����������֪S2Cl2��ˮ�������绯��Ӧ��һ������Ԫ�ػ� �ϼ����ߣ���һ���ֻ��ϼ۽��ͣ����������������ʺ��������Cl2��Ӧ��������S2Cl2����Ӧ�Ļ�ѧ����ʽΪ��2S+Cl2$\frac{\underline{\;\;��\;\;}}{\;}$S2Cl2��
S2Cl2��һ�ֽ��ɫ�ӷ���Һ�壬������������ij��ѧ��ȤС�� �����ʵ���Ʊ�������S2Cl2����������֪S2Cl2��ˮ�������绯��Ӧ��һ������Ԫ�ػ� �ϼ����ߣ���һ���ֻ��ϼ۽��ͣ����������������ʺ��������Cl2��Ӧ��������S2Cl2����Ӧ�Ļ�ѧ����ʽΪ��2S+Cl2$\frac{\underline{\;\;��\;\;}}{\;}$S2Cl2����Ӧ�漰�ļ������ʵ��۷е����£�
| ���� | S | S2Cl2 |
| �е�/�� | 445 | 138 |
| �۵�/�� | 113 | -76 |
�ش��������⣺
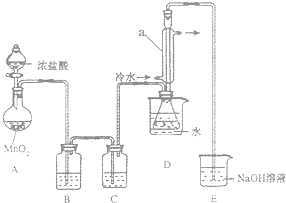
��1�����Ӻ�ʵ��װ�ú�ĵ�һ��ʵ������Ǽ��װ�õ������ԣ�
��2��ʵ������Ҫ���ȵ�������AD����д��ĸ��
��3��װ��B��C�е��Լ��ֱ��DZ���ʳ��ˮ��Ũ���
��4��װ��D������a������������������������
��5����Ӧ���������ƿ�ڻ�����з������Ʒ�ķ���������
��6����ʵ�������ȱ��Cװ�ã����ֲ�Ʒ���Dz��壬���û�ѧ����ʽ��ʾ��ԭ��2S2Cl2+2H2O=3S��+SO2��+4HCl����
��7��ʵ����ϣ�С���е�һλͬѧ��ʣ��Ũ���ᵹ��E�ձ��У������л���ɫ�ݼ�����������������ӷ���ʽ��ʾ�����������ԭ��ClO-+2H++Cl-=Cl2��+H2O��
| �� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| 2 | �� | �� | ||||||
| 3 | �� | �� | �� | �� | �� | �� |
��2���ۡ��ܡ�������Ԫ���е�ԭ�Ӱ뾶��С����O����Ԫ�ط��ţ���
��3���١��ڡ�������Ԫ���γɵ����ӣ����Ӱ뾶�ɴ�С��˳����Na+��Mg2+��Al3+�������ӷ�����д����
��4��������γɵĻ�����ĵ���ʽΪ
 ��
����5���������γɺ�������ߵĻ�����Ľṹʽ
 ��
����6��д���ٺ͢�����Ԫ�ص�����������Ӧ��ˮ�������Ӧ�����ӷ���ʽ��Al��OH��3+OH-=AlO2-+2H2O��
��7��д���١�����Ԫ���γɼȺ����Ӽ��ֺ����ۼ��Ļ�����ĵ���ʽ��
 ���γɵľ��������Ӿ��壮
���γɵľ��������Ӿ��壮 | A�� | 10min�ڣ���Ӧ�ų�������Ϊ197kJ���� | |
| B�� | 10min�ڣ�X��ƽ����Ӧ����Ϊ0.06mol•L-1•min-1 | |
| C�� | ��10minʱ��Y�ķ�Ӧ����С��0.015mol•L-1•min-1��������ϵ�¶Ȳ��䣩 | |
| D�� | ��10minʱ��ZŨ��Ϊ0.6mol•L-1 |
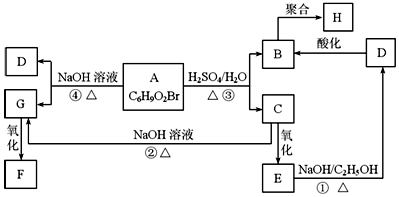 �л���A��B��C��D��E��F��G��H�ת����ϵ��ͼ��ʾ��5.2g F����100mL 1mol/L NaOH��Һǡ����ȫ�кͣ�0.1molF����������NaHCO3��Ӧ�ڱ�״���·ų�4.48LCO2��D�ķ���ʽΪC3H3O2Na��E�ķ����к����Ȼ���
�л���A��B��C��D��E��F��G��H�ת����ϵ��ͼ��ʾ��5.2g F����100mL 1mol/L NaOH��Һǡ����ȫ�кͣ�0.1molF����������NaHCO3��Ӧ�ڱ�״���·ų�4.48LCO2��D�ķ���ʽΪC3H3O2Na��E�ķ����к����Ȼ���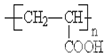 ��
��