��Ŀ����
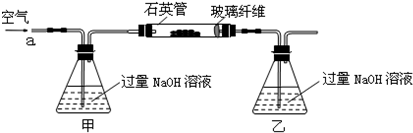
��15�֣��ס�����ͬѧΪ̽��SO2������Ա���ǿ�����ܷ�Ӧ���ɰ�ɫBaSO3����������ͼ��ʾװ�ý���ʵ�飨�г�װ�ú�A�м���װ�����ԣ��������Ѽ��飩
ʵ�����������
| ���� | ���� |
| �رյ��ɼУ��μ�һ����Ũ���ᣬ���� | A���а������ɣ�ͭƬ����������� B��������ð��������������ɫ���� C�в�����ɫ������Һ���Ϸ�����dz��ɫ������ʧ |
| ���ɼУ�ͨ��N2,ֹͣ���ȣ�һ��ʱ���ر� | ___________________ |
| ��B��C�зֱ�ȡ������ɫ��������ϡ���� | ��δ���ְ�ɫ�����ܽ� |
��1��A �з�Ӧ�Ļ�ѧ����ʽ��_________________��
��2��C�а�ɫ������__________________���ó��������ɱ���SO2����___________�ԡ�
��3��C��Һ���Ϸ�����dz��ɫ����Ļ�ѧ����ʽ��_____________________��
��4������B�в�����ϡ����ij���������ԭ����Ϊ�ǿ����гɷֹ�ͬ���ã�����Ϊ��ֻ�а������뷴Ӧ��
��Ϊ֤ʵ���ԵĹ۵㣬��ԭʵ������ϣ�����ԭ�в���֮ǰ����һ���������ò�����_____________;
����A��B������ϴ��ƿD��D��ʢ�ŵ��Լ���_____________��
�ڽ���ʵ�飬B������
| �� | ������ɫ���� |
| �� | ������ɫ���� |
�����ɫ���������־�������ϡ���ᡣ������ӷ���ʽ����ʵ��������ͬ��ԭ��_____��
��5���ϲ���4������ͬѧ�ķ����������顣B���������ɣ���C�в�����ɫ�������ɴ˵ó��Ľ�����_____________��
��15�֣�
��1��2H2SO4(Ũ)��CuCuSO4��2H2O��2SO2����
��2��BaSO4 ��ԭ
��3��2NO+O2=2NO2��
��4����ͨN2һ��ʱ�䣬�ų�װ�õĿ��� ���͵�NaHSO3��Һ
�ڼף�SO42����Ba2��=BaSO4�����ң�2Ba2����2SO2��O2��2H2O=2BaSO4��4H������������ҪԶ����װ������������
��5��SO2������Ա���ǿ���β��ܷ�Ӧ����BaSO3����
����:��

������ʯ����Ҫ�ɷ�ΪFeS2������FeS���������������в���Fe��SԪ�أ��Ҹ����²�������ѧ�仯�������ҹ���������᳧��ȡ�������Ҫԭ�ϣ�ij��ѧ��ȤС��Ըû�����ʯ��������ʵ��̽����
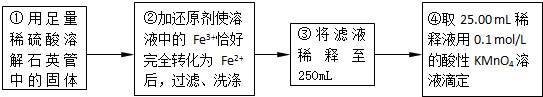
��m1g�û�����ʯ����Ʒ��������ͼװ�ã��гֺͼ���װ���ԣ���ʯӢ���У���a�����ϵػ���ͨ��������������ջ�������Ʒ����Ӧ��ȫ���䷴Ӧ�Ļ�ѧ����ʽΪ��
4FeS2+11O2 2Fe2O3+8SO2����
2Fe2O3+8SO2����
4FeS+7O2 2Fe2O3+4SO2
2Fe2O3+4SO2
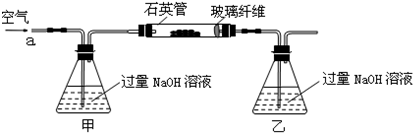
ʵ��һ���ⶨ��Ԫ�صĺ���
��Ӧ��������ƿ�е���Һ�������´�����
��1���ס�����װ�õ����÷ֱ���______��______��
��ƿ�ڷ�����Ӧ�����ӷ���ʽΪ______��______��
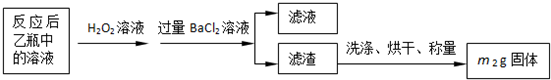
��2����Ӧ����ƿ�е���Һ�������H2O2��Һ��Ŀ����______��
��3���û�����ʯ����Ԫ�ص���������Ϊ______��
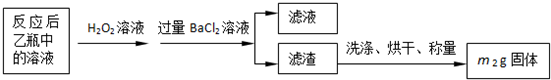
ʵ������ⶨ��Ԫ�صĺ���
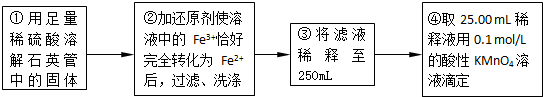
��4�����У�������������ԭ����������õ���Ԫ�صĺ���______���ƫ����ƫС������Ӱ�족����
��5�����У���Ҫ�õ����������ձ�������������ͷ�ι��⣬����______��
��6��ijͬѧһ�����������εζ�ʵ�飬����ʵ������¼���£�
| ʵ����� | ��һ�� | �ڶ��� | ������ |
| ����KMnO4��Һ���/mL | 25.00 | 25.03 | 24.97 |
| ʵ���� | ʵ���¶�/�� | c��Na2S2O3��/mol?L-1 | c��H2SO4��/mol?L-1 |
| �� | 25 | 0.1 | 0.1 |
| �� | 25 | 0.1 | 0.2 |
| �� | 50 | 0.2 | 0.1 |
| �� | 50 | 0.1 | 0.1 |
̽���¶ȶԻ�ѧ��Ӧ���ʵ�Ӱ�죬Ӧѡ��______����ʵ���ţ���
��2����֪Na2S2O3��Һ��Cl2��Ӧʱ��1mol Na2S2O3ת��8mol���ӣ��÷�Ӧ�����ӷ���ʽ��______��
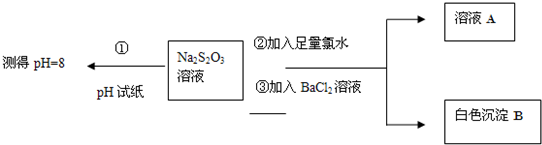
��ͬѧ�������ʵ������̽��Na2S2O3�Ļ�ѧ���ʣ�
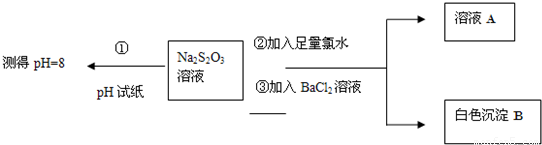
����ͬѧ���ʵ�����̵�Ŀ����֤��Na2S2O3��Һ���м��Ժ�______�ԣ�
�������ɰ�ɫ����B�����ӷ���ʽ��______��
������ͬѧ��ΪӦ�����������Тڢ������Լ�˳��ߵ�������Ϊ�ס�����ͬѧ����Ƹ���������______����ס����ҡ�����������______��