��Ŀ����
�л���A��C10H20O2������������ζ��������������ϴ���㲨�ķ��㸳�������֪
��B������û��֧����
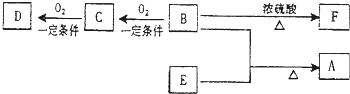
��D����̼��������Һ��Ӧ�ų�������̼��
��D��E��Ϊ������ͬ�����ŵ�ͬ���칹�壮E���������ϵ�������Clȡ������һ�ȴ���ֻ��һ�֣�
��F����ʹ������Ȼ�̼��Һ��ɫ��
��1��A�Ľṹ��ʽ ��
��2��B���Է����ķ�Ӧ�� ������ţ���
��ȡ����Ӧ���ڼӾ۷�Ӧ����������Ӧ
��3��C��F���������Ĺ����ŵ����������� ��
��4��д����D��E������ͬ�����ŵ�ͬ���칹��Ŀ��ܽṹ��ʽ �� ��
��5��E����������������ù�صȣ���֪E���Ʊ�������ͬ���䳣����ͬϵ��ݱ���������2-��-1-������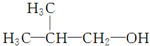 ���ͼ�����һ����������ȡE���÷�Ӧ�Ļ�ѧ����ʽ�� ��
���ͼ�����һ����������ȡE���÷�Ӧ�Ļ�ѧ����ʽ�� ��

��B������û��֧����
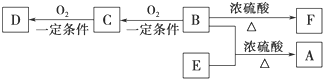
��D����̼��������Һ��Ӧ�ų�������̼��
��D��E��Ϊ������ͬ�����ŵ�ͬ���칹�壮E���������ϵ�������Clȡ������һ�ȴ���ֻ��һ�֣�
��F����ʹ������Ȼ�̼��Һ��ɫ��
��1��A�Ľṹ��ʽ
��2��B���Է����ķ�Ӧ��
��ȡ����Ӧ���ڼӾ۷�Ӧ����������Ӧ
��3��C��F���������Ĺ����ŵ�����������
��4��д����D��E������ͬ�����ŵ�ͬ���칹��Ŀ��ܽṹ��ʽ
��5��E����������������ù�صȣ���֪E���Ʊ�������ͬ���䳣����ͬϵ��ݱ���������2-��-1-������
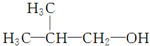 ���ͼ�����һ����������ȡE���÷�Ӧ�Ļ�ѧ����ʽ��
���ͼ�����һ����������ȡE���÷�Ӧ�Ļ�ѧ����ʽ�����㣺�л�����ƶ�
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
������D����̼��������Һ��Ӧ�ų�������̼��D�����Ȼ�����ת����ϵ�ɵ�BΪ����CΪȩ��D��E��Ϊ������ͬ�����ŵ�ͬ���칹�壬D��EΪ���ᣬ��AΪ������B��C��D��E��F������̼ԭ������ͬ��A�ķ���ʽΪC10H20O2����B��E����ʽ����ΪC5H12O��C5H10O2��B��֧������B�ṹ��ʽΪCH3��CH2��3CH2OH��CΪCH3��CH2��3CHO��DΪCH3��CH2��3COOH��E���������ϵ�������Clȡ������һ�ȴ���ֻ��һ�֣���E�Ľṹ��ʽΪ��CH3��3CCOOH��B��E����������Ӧ����A����AΪC��CH3��3COOCH2��CH2��3CH3��F����ʹ������Ȼ�̼��Һ��ɫ����B��Ũ���ᡢ���������·�����ȥ��Ӧ����F��F�Ľṹ��ʽΪCH3��CH2��2CH=CH2���ݴ˽��
���
�⣺D����̼��������Һ��Ӧ�ų�������̼��D�����Ȼ�����ת����ϵ�ɵ�BΪ����CΪȩ��D��E��Ϊ������ͬ�����ŵ�ͬ���칹�壬D��EΪ���ᣬ��AΪ������B��C��D��E��F������̼ԭ������ͬ��A�ķ���ʽΪC10H20O2����B��E����ʽ����ΪC5H12O��C5H10O2��B��֧������B�ṹ��ʽΪCH3��CH2��3CH2OH��CΪCH3��CH2��3CHO��DΪCH3��CH2��3COOH��E���������ϵ�������Clȡ������һ�ȴ���ֻ��һ�֣���E�Ľṹ��ʽΪ��CH3��3CCOOH��B��E����������Ӧ����A����AΪC��CH3��3COOCH2��CH2��3CH3��F����ʹ������Ȼ�̼��Һ��ɫ����B��Ũ���ᡢ���������·�����ȥ��Ӧ����F��F�Ľṹ��ʽΪCH3��CH2��2CH=CH2��
��1��������������֪��A�Ľṹ��ʽΪ��C��CH3��3COOCH2��CH2��3CH3���ʴ�Ϊ��C��CH3��3COOCH2��CH2��3CH3��
��2��B�ṹ��ʽΪCH3��CH2��3CH2OH������-OH�����Է���ȡ����Ӧ�����ǻ�������̼ԭ�����ڵ�̼ԭ���Ϻ���Hԭ�ӣ����Է�����ȥ��Ӧ�����ǻ�������̼ԭ���Ϻ���Hԭ�ӣ����Է������������ܷ����Ӿ۷�Ӧ���ʴ�Ϊ���٢ڣ�
��3��CΪCH3��CH2��3CHO�����еĹ�����Ϊȩ������FΪCH3��CH2��2CH=CH2�����������Ĺ�������̼̼˫�����ʴ�Ϊ��ȩ����̼̼˫����
��4����3����CH3��CH2��3COOH����CH3��3CCOOH������ͬ�����ŵ�ͬ���칹�壬�������ᣬ���ܽṹ��ʽ�У�CH3CH��CH3��CH2COOH��CH3CH2CH��CH3��COOH��
�ʴ�Ϊ��CH3CH��CH3��CH2COOH��CH3CH2CH��CH3��COOH��
��5��E�Ľṹ��ʽΪ��CH3��3CCOOH����2-��-1-������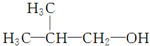 ���ͼ�����һ����������ȡ���÷�Ӧ�Ļ�ѧ����ʽ�ǣ�
���ͼ�����һ����������ȡ���÷�Ӧ�Ļ�ѧ����ʽ�ǣ� ��
��
�ʴ�Ϊ�� ��
��
��1��������������֪��A�Ľṹ��ʽΪ��C��CH3��3COOCH2��CH2��3CH3���ʴ�Ϊ��C��CH3��3COOCH2��CH2��3CH3��
��2��B�ṹ��ʽΪCH3��CH2��3CH2OH������-OH�����Է���ȡ����Ӧ�����ǻ�������̼ԭ�����ڵ�̼ԭ���Ϻ���Hԭ�ӣ����Է�����ȥ��Ӧ�����ǻ�������̼ԭ���Ϻ���Hԭ�ӣ����Է������������ܷ����Ӿ۷�Ӧ���ʴ�Ϊ���٢ڣ�
��3��CΪCH3��CH2��3CHO�����еĹ�����Ϊȩ������FΪCH3��CH2��2CH=CH2�����������Ĺ�������̼̼˫�����ʴ�Ϊ��ȩ����̼̼˫����
��4����3����CH3��CH2��3COOH����CH3��3CCOOH������ͬ�����ŵ�ͬ���칹�壬�������ᣬ���ܽṹ��ʽ�У�CH3CH��CH3��CH2COOH��CH3CH2CH��CH3��COOH��
�ʴ�Ϊ��CH3CH��CH3��CH2COOH��CH3CH2CH��CH3��COOH��
��5��E�Ľṹ��ʽΪ��CH3��3CCOOH����2-��-1-������
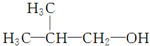 ���ͼ�����һ����������ȡ���÷�Ӧ�Ļ�ѧ����ʽ�ǣ�
���ͼ�����һ����������ȡ���÷�Ӧ�Ļ�ѧ����ʽ�ǣ� ��
���ʴ�Ϊ��
 ��
��
���������⿼���л�����ƶϣ��漰����ȩ�������������ת���ȣ��Ѷ��еȣ�������Ŀ��Ϣ����Ϸ�Ӧ���������ƶϣ�ȷ��B��C��D��E��F������̼ԭ������ͬ�ǽ���Ĺؼ���

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
��-��ԭ��ؾ���������ʽϸߣ�ѭ�����������ŵ㣮�乤��ԭ���ɱ�ʾΪ��2Na+xS
Na2Sx���������¶ȹ��������ָ����ܵ�ص�ȱ�ݣ���ѧ���о����֣����ö����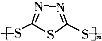 ����Ϊ�缫��Ӧ���ϣ�����Ч�ؽ��͵�صĹ����¶ȣ���ԭ���ϼ������Ͷ����������オ���ԣ������й�������ȷ���ǣ�������
����Ϊ�缫��Ӧ���ϣ�����Ч�ؽ��͵�صĹ����¶ȣ���ԭ���ϼ������Ͷ����������オ���ԣ������й�������ȷ���ǣ�������
| �ŵ� |
| ��� |
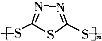 ����Ϊ�缫��Ӧ���ϣ�����Ч�ؽ��͵�صĹ����¶ȣ���ԭ���ϼ������Ͷ����������オ���ԣ������й�������ȷ���ǣ�������
����Ϊ�缫��Ӧ���ϣ�����Ч�ؽ��͵�صĹ����¶ȣ���ԭ���ϼ������Ͷ����������オ���ԣ������й�������ȷ���ǣ�������A�������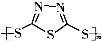 ����һ���������ǽ������� ����һ���������ǽ������� |
B�������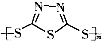 �������ﲻ�ܷ����ӳɷ�Ӧ �������ﲻ�ܷ����ӳɷ�Ӧ |
C��ԭ��صĸ�����Ӧ�ǽ����� 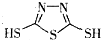 ת��Ϊ ת��Ϊ  �Ĺ��� �Ĺ��� |
| D������·����0.02mol����ͨ��ʱ��ԭ��صĸ���������ԭ��0.46g |
ijͬѧ��ʵ���ҽ�������ͼ��ʾ��ʵ�飬����˵���д�����ǣ�������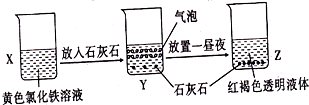

| A�����ù��˵ķ������ɽ�Z�й�����Һ����� |
| B��X��Z�ձ��з�ɢ����ͬ |
| C��Y�з�Ӧ�����ӷ���ʽΪ3CaCO3+2Fe3++3H2O=2Fe��OH��3+3 CO2��+3 Ca2+ |
| D��Z�з�ɢϵ�ܲ��������ЧӦ |
����ͼʾ���Ӧ��������������ǣ�������
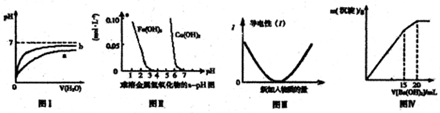

| A��ͼ���ʾpH��ͬ������������зֱ����ˮ����ҺpH�ı仯����������b��Ӧ�������� |
| B������ͼ���֪������ȥCuSO4��Һ�е�Fe3+��������Һ�м���CuO2����pH��4-5֮�伴�� |
| C��ͼ��ɱ�ʾ������Һ��ͨ�백���������Ĺ�������Һ�����Եı仯 |
| D��ͼ����ʾij������Һ�м���Ba��OH��2��Һ�����������������Ba��OH��2��Һ����Ĺ�ϵ���ڼ���20mLBa��OH��2��Һʱ������ȫ����BaSO4 |