��Ŀ����
18�����Ѿ��еľƾ������ѹ�ʵ�е��Ƿ��ͺ�IJ��C6H12O6$\stackrel{��ĸ}{��}$2CH3CH2OH+2CO2����������֪��ʵ��������ϩԭ��ΪCH3CH2OH$��_{170��}^{Ũ����}$CH2=CH2��+H2O��������������ʹBr2 �����Ȼ�̼��Һ��ɫ���ס���ͬѧ������ʵ����֤�����������Ѽ��飬���ּг�װ���ԣ���
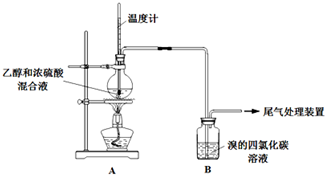
ʵ�����������
| �� �� | �� �� |
| ��ȼ�ƾ��ƣ�������170�� | ��A����ƿ��Һ�彥����� ��B����������ð������Һ����ɫ |
| �� | |
| ʵ����ϣ���ϴ��ƿ | ��A����ƿ�ڸ���������ɫ����״��д̼�����ζ�ݳ� |
��2������������Ϊ��C2H4������Ϊ�����ų�SO2�����ã�
�ٸ��ݼĹ۵㣬ʹB����Һ��ɫ��Ӧ�Ļ�ѧ����ʽ��CH2=CH2+Br2��CH2Br-CH2Br��
���Ҹ���������Ϊʵ���в�����SO2��H2O��ʹB����ɫ���ʷ�Ӧ��ɫ��
��Ϊ֤ʵ���Թ۵㣬�ס�������ʵ�飬������������£�
�ף���A��B������һ��װ��ij���Լ���ϴ��ƿ������Br2��CCl4��Һ��ɫ��
�ң�������װ�ð�һ��˳����A���ӣ���β������װ���ԣ�

����C����Һ�ɺ���ɫ��Ϊdz����ɫʱ��E����Һ��ɫ��
��ش��������⣺
a�������ʵ����A��B��ϴ��ƿ��ʢ�ŵ��Լ���NaOH��Һ������Ƶ�ʵ��D��ʢ�ŵ��Լ���Ũ���ᣬװ������˳��Ϊ��d��cabe��f����
b����˵��ȷʵ��SO2ʹE����Һ��ɫ��ʵ���Ǽ����Ѿ���ɫ��Ʒ����Һ������ɫ�ָ���֤����SO2ʹƷ����Һ��ɫ������Br2��
c����Ϊ��һ����֤��۵㣬ȡ����C����Һ�����뼸��BaCl2��Һ��������������ɫ������dz����ɫ��ʧ��������Ӧ�����ӷ���ʽ��SO2+2H2O+Br2�T4H++2Br-+SO42-��SO42-+Ba2+�TBaSO4����SO2+2H2O+Br2+Ba2+�T4H++2Br-+BaSO4�����ɴ˿ɵó��ĸ����SO2 ����ʹBr2 �����Ȼ�̼��Һ��ɫ
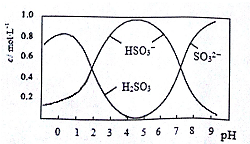
�����Ѿ��г���Na2S2O5������������
��3��0.5molNa2S2O5����ˮ���1L��Һ������Һ��pH=4.5����Һ�в�����Ũ������Һ����Ա仯����ͼ��ʾ��д��Na2S2O5����ˮʱ�����Ļ�ѧ����ʽNa2S2O5+H2O=2NaHSO3��
��4����֪��Ksp[BaSO4]=1��10-10��Ksp[BaSO3]=5��10-7���Ѳ��ֱ����������ĸ���Һ��pH��Ϊ10������Һ�еμ�BaCl2��ҺʹSO42-������ȫ[c��SO42-����1��10-5mol•L-1]����ʱ��Һ��c��SO32-����0.05mol•L-1��
���� ��1��Ũ���������ˮ�ԡ���ˮ�Ժ�ǿ�����ԣ���ʹ�Ҵ�̼����
��2������ϩ������ˮ�����ӳɷ�Ӧ��
���嵥�ʿ��ԺͶ���������������ԭ��Ӧ��
��a���������ƿ��ԺͶ�����������Ӧ��Ũ���������ˮ�ԣ�������������������ҵ���ƣ���Ҫ֤������Ķ���������ʹ������Ȼ�̼��ɫ������ʹƷ����ɫ���ݴ�ȷ��װ������˳��
b����������ʹƷ����ɫ������ʱ�ָֻ���ɫ��������һ�����ж��Ƿ�Ϊ��������ʹƷ����ɫ��
c���嵥�ʿ��ԺͶ���������������ԭ��Ӧ����������Ӻͱ����ӷ�Ӧ�����ɰ�ɫ������
��3������ͼ��֪��pH=4.5ʱ����Һ����Ҫ�����������������ʽ���ڣ��ݴ���дˮ�ⷽ��ʽ��
��4������Ksp[BaSO4]=c��Ba2+��•c��SO42-�����ɼ������Ҫ����c��Ba2+���������������Ũ��c��SO32-�����ݴ˴��⣻
��� �⣺��1��Ũ���������ˮ�ԣ���ʹ�Ҵ���Һ��������ڡ����ʴ�Ϊ����ˮ��
��2���ٸ��ݼĹ۵㣬��ˮ����ʹB����Һ��ɫ����Ӧ�Ļ�ѧ����ʽ��CH2=CH2+Br2��CH2Br-CH2Br��
�ʴ�Ϊ��CH2=CH2+Br2��CH2Br-CH2Br��
���Ҹ����������Ϊ������SO2��ˮ����B���嵥�ʿ��ԺͶ�������ˮ����������ԭ��Ӧ�����廯������ᣬ
�ʴ�Ϊ��H2O��
��a�����ݼ���ƣ�ϴ��ƿ��ʢ�ŵ��Լ����������ƣ����ԺͶ�������Ӧ�����ն��������ų���������ĸ��ţ������ҵ���ƣ�C��ʢ�ŵ��Լ��ǣ�Ũ���������ˮ�ԣ�������������������ҵ���ƣ���Ҫ֤������Ķ���������ʹ������Ȼ�̼��ɫ������ʹƷ����ɫ������װ������˳��Ϊ��d��cabe��f����
�ʴ�Ϊ��NaOH ��Һ��Ũ�����d��cabe��f����
b��֤��SO2ʹE����Һ��ɫ��ʵ���Ǽ����Ѿ���ɫ��Ʒ����Һ������ɫ�ָ���֤����SO2ʹƷ����Һ��ɫ������Br2��
�ʴ�Ϊ�������Ѿ���ɫ��Ʒ����Һ������ɫ�ָ���֤����SO2ʹƷ����Һ��ɫ������Br2��
c���嵥�ʿ��ԺͶ���������������ԭ��Ӧ��������������Ӻ������ӣ���������Ӻͱ����ӷ�Ӧ�����ɰ�ɫ��������ط�Ӧ�����ӷ���ʽΪSO2+2H2O+Br2�T4H++2Br-+SO42-��SO42-+Ba2+�TBaSO4����SO2+2H2O+Br2+Ba2+�T4H++2Br-+BaSO4����
�ʴ�Ϊ��SO2+2H2O+Br2�T4H++2Br-+SO42-��SO42-+Ba2+�TBaSO4����SO2+2H2O+Br2+Ba2+�T4H++2Br-+BaSO4����
��3������ͼ��֪��pH=4.5ʱ����Һ����Ҫ�����������������ʽ���ڣ�����ˮ�ⷽ��ʽΪNa2S2O5+H2O=2NaHSO3��
�ʴ�Ϊ��Na2S2O5+H2O=2NaHSO3��
��4������Ksp[BaSO4]=c��Ba2+��•c��SO42-������֪��Ҫc��Ba2+��=$\frac{Ksp��BaS{O}_{4}��}{c��S{O}_{4}^{2-}��}$=$\frac{1��1{0}^{-10}}{1��1{0}^{-5}}$=10-5mol•L-1������Һ��SO32-�����Ũ��c��SO32-��=$\frac{Ksp��BaS{O}_{3}��}{c��B{a}^{2+}��}$=$\frac{5��1{0}^{-7}}{1{0}^{-5}}$=0.05mol•L-1��
�ʴ�Ϊ��0.05��
���� ���⿼��ѧ���Ҵ��Ļ�ѧ���ʡ�ͼ�����������Ũ�ȵļ���ȣ����ʵ�鿼����������Ŀ���Ѷȣ��ۺ��Խ�ǿ��Ҫ��ѧ�����з����ͽ�������������

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| A�� | ������̼��K+��Na+��SiO32-��Cl- | B�� | ������Ag+��Al3+��Na+��NO3- | ||
| C�� | �Ȼ��⣺Ca2+��Fe3+��NO3-��Cl- | D�� | ������Na+��Ba2+��I-��HSO3- |
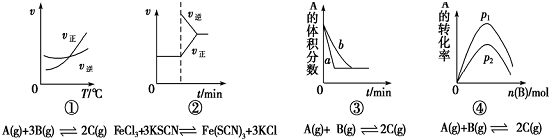
| A�� | ͼ������������һ��ʱ��Ӧ�������¶ȱ仯��ͼ��ͼ��������ϵ��¶�������ƽ�������ƶ����ϸ��¶�������ƽ�������� | |
| B�� | ͼ������ƽ����ϵ����Һ�м�������KCl�����ѧ��Ӧ������ʱ��ı仯 | |
| C�� | ͼ�ۿ����Ǵ����Ի�ѧƽ���Ӱ�죬Ҳ�������������ضԻ�ѧƽ���Ӱ�� | |
| D�� | ͼ����һ�������£�����һ����A������������Bʱ��ͼ��ѹǿp1��p2 |
| ������Ϊ���� | �����Լ� | ���Ӳ��� | |
| A | Na2O2��Na2O�� | O2 | �ڴ����м��� |
| B | Na2CO3��NaHCO3�� | CO2 | ͨ�������CO2 |
| C | FeCl��FeCl2�� | Cl2 | ͨ�������Cl2 |
| D | FeSO4��CuSO4�� | Fe | ���������Fe�ۣ���ַ�Ӧ����� |
| A�� | A | B�� | B | C�� | C | D�� | D |

| A�� | �Ʊ�AlCl3��FeCl3��CuCl2�����ܲ��ý���Һֱ�����ɵķ��� | |
| B�� | �ڶƼ��ϵ��п����п������ | |
| C�� | ����ͼ1װ��������������Һ��NaClO�� | |
| D�� | ����ͼ2װ��ΪǦ���س� |
| �¶�/�� | 400 | 500 | 830 | 1 000 |
| ƽ�ⳣ��K | 10 | 9 | 1 | 0.6 |
��1��������Ӧ������Ӧ�Ƿ��ȷ�Ӧ������ȡ������ȡ������÷�Ӧ��ƽ�ⳣ������ʽΪK=$\frac{c��{H}_{2}��•c��C{O}_{2}��}{c��CO��•c��{H}_{2}O��}$����
��2�����жϸ÷�Ӧ�Ƿ�ﵽ��ѧƽ��״̬��������bc������ţ�
a��������ѹǿ���� b����������� c��CO������
c��������H2��=������H2O�� d��c��CO2��=c��CO��
��3����ʵ�������У��÷�Ӧ����������ΪC��
A����ѹ��400��500�������B����ѹ��830��1000�����
C����ѹ��400��500�����D����ѹ��830��1000�����
��4����830��ʱ��2L���ܱ������м���4molCO��g����6molH2O��g����10min��ﵽƽ��ʱ��CO2��ƽ��Ũ��Ϊ1.2mol/L����H2Ũ�ȱ仯����ʾ��ƽ����Ӧ����Ϊ0.12mol/��L•min����CO��ת������60%��
��5��Ϊʹ�÷�Ӧ�ķ�Ӧ����������ƽ��������Ӧ�����ƶ�����a������ĸ��ţ�
a������COŨ�� b�������¶�
c��������������ȥ d��ʹ�ø�Ч������
����˵������ȷ���ǣ�������
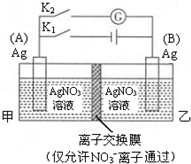
| A�� | �պ�K1���Ͽ�K2��A�缫���� | |
| B�� | �պ�K1���Ͽ�K2���ҳ���Һ��Ag+Ũ������ | |
| C�� | �Ͽ�K1���պ�K2��B�缫����������Ӧ | |
| D�� | �Ͽ�K1���պ�K2��NO3-��A�缫�ƶ� |
| A�� | �������������ˮ���� | |
| B�� | ����ʱ���Ż��������Ϲ��� | |
| C�� | ����ú���ܵ�©���������رշ��Ų�����ͨ�� | |
| D�� | ͼ�����ͼ���Ż�������Һ̬������̼��������� |
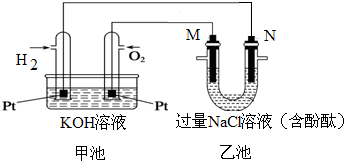 ��ͼ��ʾ������ȼ�ϵ�ع���ʱ��װ�ã��ҳ��е������缫����ʯī�缫����ش��������⣺
��ͼ��ʾ������ȼ�ϵ�ع���ʱ��װ�ã��ҳ��е������缫����ʯī�缫����ش��������⣺