��Ŀ����
11��ij��ѧ�о�С��̽����ˮ������������K3[Fe��C2O4��3]���ȷֽ�IJ����������������������������֤������ͼװ�ý���ʵ�飨�г���������ȥ����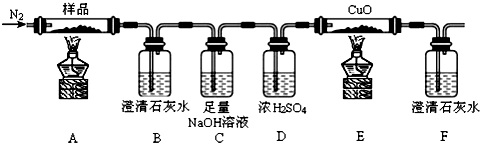
��1�����Ӻ�װ�ú���ҩƷǰ��Ӧ�ý��е�ʵ������Ǽ��װ�õ������ԣ���������ƣ���
��2��ʵ�鿪ʼʱ����ͨ�뵪������һ��ʱ���Ժ��ȷ�Ӧ��A��E������Ӧ��ȫ��ֹͣ���ȣ��Լ���ͨ������ֱ����Ӧ����ȴ��ֹͣ����ǰ�Ƿ���Ҫ�ȶϿ�A��B�����Ӵ���E��F�����Ӵ���Ϊʲô������Ҫ����ΪN2����ͨ�룬B��Fƿ��Һ���ᵹ����
��3��ʵ���й۲쵽B�г���ʯ��ˮ����ǣ���˵�������������CO2��д��ѧʽ����
��֤���ֽ��������CO�������ɵ�ʵ��������E�к�ɫ�����ɺ�ɫ��F����Һ����ǣ�
��4����С��ͬѧ�������Ϻ��֪����������в�����+3��FeԪ�أ�����������ֻ��K2CO3��
����֤��������м�Ԫ�ش��ڵķ�������ɫ��Ӧ������������ɫ���ܲ����۲쵽��ɫ���森
���о�С��Ϊ̽����Ԫ�صĴ�����ʽ��ȡA�г�ַ�Ӧ�����ù������Թ��У�������������ˮ�����ˡ�ϴ�ӡ�����õ���ɫ��ĩ��ȡ4.4g��ɫ��ĩ��������ϡ�����У��ռ�����״����NO����1.12Lͨ�������жϺ�ɫ��ĩ�ijɷ�ΪFe��FeO��д��ѧʽ����д����ɫ��ĩ��������ϡ���ᷢ����Ӧ�����ӷ���ʽ��3FeO+10H++NO3-=3Fe3++NO��+5H2O��Fe+4H++NO3-=Fe3++NO��+2H2O��
���� ̽����ˮ������������K3[Fe��C2O4��3]���ȷֽ�IJ�����Ӻ�װ�ú���ҩƷǰ��Ӧ�ý��е�ʵ������Ǽ���װ�������ԣ����õ����ṩ���Ի�����������ŷֽ����ķ�������֤��װ��A����ˮ������������K3[Fe��C2O4��3]���ȷֽ⣬װ��B�����������ͨ���Μ[ʯ��ˮ���۲쵽����ʯ��ˮ����ǣ���CO2���ɣ�װ��C�����������ƽ�CO2������װ��D�����������ʣ��������壬װ��E������CO����CuO����죩��װ��F��������CO2���ɣ�CO�����������
��1������װ��ͼ��֪�����õ����ṩ���Ի�����������ŷֽ����ķ�������֤������ҩƷǰ��Ӧ�ý��е�ʵ������Ǽ��װ�������ԣ�
��2������ʵ���������Ҫ����ͨ�뵪��ֱ����������ȴ��������
��3��B���dz���ʯ��ˮ�����ɳ���֤�����ɶ�����̼��֤��һ����̼���ɿ�������װ��E�к�ɫ�����ɺ�ɫ��F����Һ����ǣ�˵��һ����̼��ԭ����ͭ��Ӧ���ɶ�����̼ͨ��F����ʯ��ˮ����ǣ�
��4������֤��������м�Ԫ�ش��ڵ�ʵ�鷽������ɫ��Ӧ��������Ҫ����ɫ���ܲ����۲쵽��ɫ�������жϣ�
���ȸ���n=$\frac{V}{{V}_{m}}$�����NO�����ʵ������ٸ��ݻ��ϼ۱仯���������һ������ת�Ƶĵ��ӵ����ʵ�����Ȼ�����ü�ֵ���͵����غ��ж�4.4g�������ɣ����д����Ӧ�����ӷ���ʽ��
��� �⣺��1��װ��ͼ������֪���������ɵ����ṩ���Ի�����������ŷֽ����ķ�������֤������ҩƷǰ��Ӧ�ý��е�ʵ������Ǽ��װ�������ԣ�
�ʴ�Ϊ�����װ�������ԣ�
��2��ʵ���������Ҫ����ͨ�뵪��ֱ����������ȴ�������������B��F����Һ�У����Բ�����ֵ�������
�ʴ�Ϊ������ΪN2����ͨ�룬B��Fƿ��Һ���ᵹ����
��3��B���dz���ʯ��ˮ�����ɳ���֤�����ɶ�����̼��֤��һ����̼���ɿ�������װ��E�к�ɫ�����ɺ�ɫ��F����Һ����ǣ�˵��һ����̼��ԭ����ͭ��Ӧ���ɶ�����̼ͨ��F����ʯ��ˮ����ǣ�
�ʴ�Ϊ��CO2��E�к�ɫ�����ɺ�ɫ��F����Һ����ǣ�
��4������֤��������м�Ԫ�ش��ڵ�ʵ�鷽������ɫ��Ӧ��������Ҫ����ɫ���ܲ����۲쵽��ɫ���棬
�ʴ�Ϊ����ɫ��Ӧ������ɫ���ܲ����۲쵽��ɫ���棻
�ڱ����1.12Lһ�����������ʵ���Ϊ��n��NO��=$\frac{1.12L}{22.4L/mol}$=0.05mol������0.05mol������Ҫת�Ƶĵ��ӵ����ʵ���Ϊ����5-2����0.05mol=0.15mol�����ݵ����غ㣬���������ȫΪ��������������Ϊ��56gmol��$\frac{0.15mol}{3}$=2.8g���������ΪFeO������������Ϊ��72g/mol��$\frac{0.15mol}{3-2}$=10.8g������4.4gΪFe��FeO�Ļ���
Fe�����ᷴӦ�����ӷ���ʽΪ��3FeO+NO3-+10H+�T3Fe3++5H2O+NO����FeO�����ᷴӦ�����ӷ���ʽΪ��Fe+NO3-+4H+�TFe3++2H2O+NO�����ܷ�ӦΪ��3FeO+Fe+2NO3-+14H+�T4Fe3++7H2O+2NO����
�ʴ�Ϊ��Fe��FeO��3FeO+NO3-+10H+�T3Fe3++5H2O+NO����Fe+NO3-+4H+�TFe3++2H2O+NO����3FeO+Fe+2NO3-+14H+�T4Fe3++7H2O+2NO����
���� ���⿼����������ʵ�鷽������ƣ��漰��Ԫ�ؼ��仯�������ʡ����ӷ���ʽ��д��֪ʶ����Ŀ�Ѷ��еȣ�����֪ʶ��ϴ��ۺ��Խ�ǿ����һ�������Ϻõ���Ŀ�����������ѧ��������ѧ֪ʶ����������ѧʵ��������

| A�� | ��Fe��N03��2��Ʒ����ϡH2SO4�μ�KSCN��Һ����Һ��죬��Fe��N03��2�������������� | |
| B�� | �����£�pH��Ϊ11������������Һ�Ͱ�ˮ����ˮϡ��100����pH����Ϊ9 | |
| C�� | 25��ʱ��Ksp�� BaC03����Ksp�� BaCrO4���������ˮ���е�Ba2+ѡ��Na2CrO4��Na2CO3�� | |
| D�� | ��CH3CH2Br��NaOH��Һ���ȣ���ȴ��ȡ���ϲ���Һ������AgNO3��Һ��������������CH3CH2Br��NaOH��Һ�з�����ˮ�� |
| A�� | CaCl2 | B�� | NaOH | C�� | CCl4 | D�� | CO2 |
| A�� | NH3 | B�� | NO | C�� | SO3 | D�� | CO2 |
| A�� | �ɵ����ZnΪ������̿Ϊ���� | |
| B�� | �ɵ�ع���ʱ��������Zn�������·����̿�� | |
| C�� | �ɵ�س�ʱ������ʹ��ʱ��װ��״�����������ʴ���� | |
| D�� | �ɵ�ؿ���ʵ�ֻ�ѧ������ܵ�ת���͵�����ѧ�ܵ�ת�� |

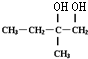 +O2 $��_{��}^{����}$
+O2 $��_{��}^{����}$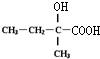 +H2O��
+H2O��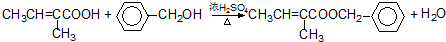
 ��
��