��Ŀ����
19��ijͬѧΪ�ⶨά����C�ķ���ʽ��ȡά����C��Ʒ���飬��ȡ������0.352g�����ڲ��۲�����ȼ�չ��У�����ͨ�����������þƾ���Ƴ���������Ʒ�����������Ⱥ�ͨ����ˮ�Ȼ��ƺͼ�ʯ�ң���������ȫ�����գ����߷ֱ�����0.144g��0.528g���������йظ�ʵ�鼰ά����C��˵������ȷ���ǣ�������| A�� | ά����C�������������ԼΪ4.5% | |
| B�� | 0.352g����������Ԫ�ص�����Ϊ0.192g | |
| C�� | ���ø�ʵ���������ݼ���ɵ�ά����C�ķ���ʽΪC3H4O3 | |
| D�� | ά����C��һ�ֿ����������׳ƿ���Ѫ�ᣬ�������Ի������Һ���ױ����� |
���� A���Ȼ�������0.144gΪȼ������ˮ����������ʯ�����ص�������CO2������Ԫ���غ����HԪ�������������㣻
B������ע�������غ㶨�ɼ�����������Ԫ�ص�������
C���ٸ���ԭ���غ�ȷ�����ʽ�������ʽ��Hԭ���Ѿ�����̼���ļ۽ṹ�����ʽ��Ϊ����ʽ��������Ҫ֪��ά����C����Է�������ȷ������ʽ��
D��ά����C�׳ƿ���Ѫ�ᣬ�������Ի������Һ���ױ���������һ�ֿ���������
��� �⣺A��n��H2O��=$\frac{0.144g}{18g/mol}$=0.008mol��0.008molˮ�к���0.016mol Hԭ�ӣ�����Ԫ�ص�����Ϊ��1g/mol��0.016mol=0.016g����Ԫ�ص���������Ϊ��w��H��=$\frac{0.016g}{0.352g}$��100%��4.5%����A��ȷ��
B�����ɶ�����̼�����ʵ���Ϊ$\frac{0.528g}{44g/mol}$=0.012mol��0.352g ά����C�к���̼Ԫ�ص�����Ϊ��0.012mol��12g/mol=0.144g��������Ԫ�ص�����Ϊ��0.016g������C��HԪ�ص�����Ϊ��0.144g+0.016g=0.160g��0.352g����ά����C��һ��������Ԫ�أ�������Ԫ�ص�����Ϊ��0.352g-0.144g-0.016g=0.192g����B��ȷ��
C����Ʒ����ԭ�����ʵ���Ϊ$\frac{0.192g}{16g/mol}$=0.012mol����Ʒ��C��H��Oԭ����Ŀ֮��Ϊ0.012mol��0.016mol��0.012mol=3��4��3����ά����C�����ʽΪC3H4O3��ȷ��ά����C�ķ���ʽ����Ҫ֪��ά����C����Է�����������C����
D��ά����C�׳ƿ���Ѫ�ᣬ�������Ի������Һ���ױ���������һ�ֿ�����������D��ȷ��
��ѡC��
���� ���⿼���л������ʽȷ�����л�������ʣ��ѶȲ���ע������ȼ�շ�����ԭ���غ�ȷ���л������ʽ��

 �������¿��ÿ�ʱ��ҵϵ�д�
�������¿��ÿ�ʱ��ҵϵ�д� Ӣ�żƻ�ͬ����ʱ��Чѵ��ϵ�д�
Ӣ�żƻ�ͬ����ʱ��Чѵ��ϵ�д�| A�� | ��״���£�2.24LSO3����������Ϊ0.1NA | |
| B�� | 0.1 mol Fe���������ᷴӦ��ת�Ƶĵ�����Ϊ0.3 NA | |
| C�� | 1molNa2O2������ˮ��Ӧת�Ƶĵ�����ΪNA | |
| D�� | ���³�ѹ�£�92g��NO2��N2O4������庬�е�ԭ����Ϊ6NA |
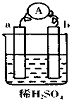 ����ͼ��ʾ��װ���У�a�Ľ����Ա���Ҫǿ��bΪ̼�������ڴ�װ�õĸ���������ȷ���ǣ�������
����ͼ��ʾ��װ���У�a�Ľ����Ա���Ҫǿ��bΪ̼�������ڴ�װ�õĸ���������ȷ���ǣ�������| A�� | ̼����������ų�����ҺpH��С | B�� | a��������b�Ǹ��� | ||
| C�� | ��Һ������������a�� | D�� | a���Ϸ�����������Ӧ |
| A�� | ���������ĵ缫��ӦΪH2O2+2H++2e-�T2H2O | |
| B�� | ����ܷ�ӦΪMg+H2O2�TMg��OH��2 | |
| C�� | ����ʱ��������Χ��ˮ��pH��С | |
| D�� | ��ع���ʱ����Һ�е�H+���ƶ� |
| A�� | �����嵥�ʵ������Կ��ѵ�����ת��Ϊ�ⵥ�� | |
| B�� | ʵ���ҳ���NaOH��Һ���ն����Cl2�Ա�����Ⱦ | |
| C�� | Cl2����ʹ��ɫ������ɫ����Cl2���������� | |
| D�� | �����ĺ��ȵ���˿��Cl2�о���ȼ�գ���Ӧ����FeCl2 |
| Ԫ�ش��� | L | M | Q | R | T |
| ԭ�Ӱ뾶/nm | 0.160 | 0.143 | 0.102 | 0.089 | 0.074 |
| ��Ҫ���ϼ� | +2 | +3 | +6��-2 | +2 | -2 |
| A�� | L��R�ĵ�����ϡ���ᷴӦ����L��R | |
| B�� | M��T�γɵĻ����������� | |
| C�� | Q��T��Ԫ�ص��⻯����Ӽ䶼������� | |
| D�� | L��Q�γɵļ����Ӻ����������� |
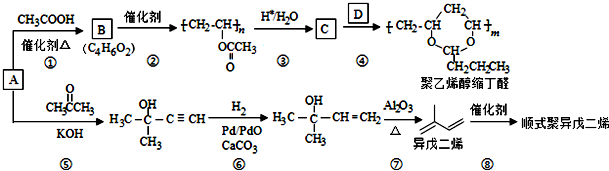
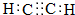 ��B���еĺ�����������������д���ƣ���
��B���еĺ�����������������д���ƣ���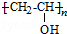 ��DΪCH3CH2CH2CHO��
��DΪCH3CH2CH2CHO��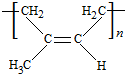 ��
��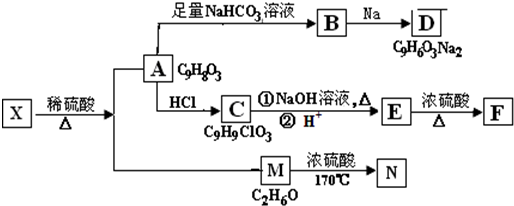
 +NaHCO3��
+NaHCO3�� +H2O+CO2����
+H2O+CO2����