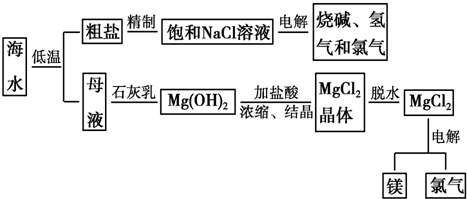
��Ŀ����
16���١����Ǽ����л�������ơ�����ʽ��ṹ��ʽ����C2H2���������顢�۱�����ClCH=CHCl����C5H4����CH3CH��C2H5��CH2CH��C2H5��CH3����C5H10
��
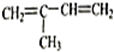 ����
����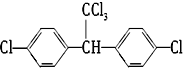 ����
����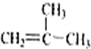
�ݴ˻ش��������⣺
��1�������л����У���Ϊͬϵ����Ǣڢޣ�������˳���칹���Ǣ٢ڢۢݢޢߢ��⣮������ţ�
��2����������ʵ�����H2����l��4 �ӳ����ò�������ƣ�2-��-2-��ϩ��
��3��ʵ������ȡ�ٵĻ�ѧ��Ӧ����ʽ��CaC2+2H2O��Ca��OH��2+C2H2����
��4���۵�ͬϵ��A��A�����й���66�����ӣ�������һ�����ֻ��һ�֣���д��A �Ľṹ��ʽ��
 ��
��
���� ��1��ͬϵ���ǽṹ���ƣ���������CH2ԭ���ŵ����ʻ���ͬϵ���ϩ����̼̼˫�����˵�ÿ��̼ԭ�����������������Ų�ͬʱ����ϩ������˳���칹��
��2����͵����ʵ��������������ӳɷ�Ӧ����2�ź�3��̼ԭ��֮����γ�һ��̼̼˫����
��3��ʵ�����õ�ʯ��ˮ��Ӧ��ȡ��Ȳ��
��4������ͬϵ��ڽṹ��ֻ��һ������������Ϊ��������������ͨʽCnH2n-6�����ݵ�����Ϊ66�������ʽ��
��� �⣺��1��ͬϵ���ǽṹ���ƣ���������CH2ԭ���ŵ����ʻ���ͬϵ����ڢľ�Ϊ�������ʽṹ���ƣ��ڷ�����������4��CH2ԭ���ţ��ʻ�Ϊͬϵ����л����д���̼̼˫������̼̼˫�����˵�ÿ��̼ԭ�����������������Ų�ͬʱ�����л������˳���칹����ֻ�Т�ClCH=CHCl����˳���칹������ľ�������˳���칹��
�ʴ�Ϊ���ڢޣ��٢ڢۢݢޢߢ��⣻
��2����͵����ʵ��������������ӳɷ�Ӧ������̼̼˫������Ȼ����2�ź�3��̼ԭ��֮����γ�һ��̼̼˫������2��̼ԭ������һ���������ò��������Ϊ��2-��-2-��ϩ���ʴ�Ϊ��2-��-2-��ϩ��
��3��ʵ�����õ�ʯ��ˮ��Ӧ��ȡ��Ȳ������ʽΪCaC2+2H2O��Ca��OH��2+C2H2�����ʴ�ΪCaC2+2H2O��Ca��OH��2+C2H2����
��4������A�DZ���ͬϵ����ڽṹ��ֻ��һ���������Ҳ���Ϊ��������������ͨʽCnH2n-6�����ڵ�����Ϊ66�����У�6n+2n-6=66�����n=9����A�ķ���ʽΪC9H20������A�ı�����һ�����ֻ��һ�֣���A�Ľṹ�ܶԳƣ��ṹΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
���� �����ۺϿ�����ͬϵ�ͬ���칹����жϺ��л�����Ʊ��Լ�ͬ���칹�����д���ۺ��Խ�ǿ���ѶȲ���

| A�� | dԪ�صķǽ�������ǿ | |
| B�� | ���Ǿ��������ֻ��������ϵ������� | |
| C�� | ֻ��a������Ԫ�����ɵĻ����ﶼ�����ӻ����� | |
| D�� | a��b��c��d�����γɵĻ������л�ѧ����Ϊ���Թ��ۼ� |
| A�� | 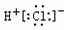 | B�� | 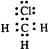 | C�� |  | D�� | 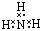 |
| A�� | c��H+��/c��OH-��=1012��ˮ��Һ�У�NH4+��Al3+��NO3-��Cl- | |
| B�� | pH=1����Һ�У�Fe2+��NO3-��AlO2-��Na+ | |
| C�� | ˮ�������c��H+��=10-12 mol•L-1����Һ�У�Ca2+��K+��Cl-��HCO3- | |
| D�� | c��Fe3+��=0.1 mol•L-1����Һ�У�K+��ClO-��SO42-��SCN- |
| A�� | ���ʵ���ɫ��˵���������Ӷ����� | |
| B�� | ���ʵ��ۡ��е���˵���������Ӷ����� | |
| C�� | ���ʵ������Լ��� | |
| D�� | ���ǵ��⻯����ȶ�����˵���������Ӷ���ǿ |
| A�� | �����ĵ���ʽ  | B�� | S2-�Ľṹʾ��ͼ��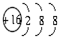 | ||
| C�� | ������̼���ӵĽṹʽΪ O-C-O | D�� | NaCl�ĵ���ʽ��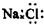 |
| A�� | ��ԭ�ӵĽṹʾ��ͼ 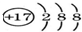 | |
| B�� | �廯�Ƶĵ���ʽ��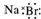 | |
| C�� | �Ȼ�þ�ĵ���ʽ��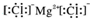 | |
| D�� | �õ���ʽ��ʾ�Ȼ�����ӵ��γɹ��̣� |
| A�� | ���������ںϽ��һ�֣����Ź㷺����; | |
| B�� | �й���IJ˵����й��߲˵IJ˵�����ʴ | |
| C�� | ���ǵؿ��к�����ߵĽ�����Ҳ���������������ߵĽ��� | |
| D�� | �������صĻ�������ʱӦŬ��ʵ���ô�������� |