��Ŀ����
����ʽ�о���ѧϰ������ѧ������˼ά�����÷�����ij�о���ѧϰС�齫����װ����ͼ���ӣ�D��F��X��Y ���Dz��缫��C��E�����缫������Դ��ͨ�������е����̪��Һ����F�������Ժ�ɫ���Իش��������⣺

��1����ԴB���������� ��
��2����װ���е�ⷴӦ���ܻ�ѧ����ʽ�ǣ� ��
��3����������Һ��������ͬһʱ����C��D�缫�ϲμӷ�Ӧ�ĵ��ʻ����ɵĵ��ʵ����ʵ���֮�� ��
��4�����ñ�װ�ý���ͭ������������п�����ʣ�������G������Ӧ���� �����ͭ����ͭ���������Һ��ԭ����ʵ����ʵ���Ũ�Ƚ� ����������С�����䡱����
��5����׳�����Һ������ڵ��ǰ����500mL�����ҳ���������������Ϊ4.48L����״����ʱ���׳������������ʵ����ʵ���Ũ��Ϊ mol/L��

��1����ԴB����������
��2����װ���е�ⷴӦ���ܻ�ѧ����ʽ�ǣ�
��3����������Һ��������ͬһʱ����C��D�缫�ϲμӷ�Ӧ�ĵ��ʻ����ɵĵ��ʵ����ʵ���֮��
��4�����ñ�װ�ý���ͭ������������п�����ʣ�������G������Ӧ����
��5����׳�����Һ������ڵ��ǰ����500mL�����ҳ���������������Ϊ4.48L����״����ʱ���׳������������ʵ����ʵ���Ũ��Ϊ
���㣺ԭ��غ͵��صĹ���ԭ��
ר�⣺�绯ѧר��
��������1����ⱥ��ʳ��ˮʱ����̪���ļ���������������·�У������������������������������͵�Դ���������������͵�Դ����������
��2�����ݵ��ԭ������д��ط�Ӧ���׳���CΪ����������DΪ���缫���������Һ������ͭ��Һ�����ݵ��ԭ��������
��3�����ݵ缫��Ӧ�͵����غ�����ܽ���������������ʵ�����ϵ��
��4����⾫��ͭʱ����ͭ����������ͭ������������ͭ���ӵ�����Һ���������Һ��
��5���ҳ���EΪ������FΪ���������ҳ���������������Ϊ4.48L����״����ʱ���׳���������������Ũ�ȣ����ݵ����غ���㷴Ӧ�������ʵ����õ���������Ũ�ȣ�
��2�����ݵ��ԭ������д��ط�Ӧ���׳���CΪ����������DΪ���缫���������Һ������ͭ��Һ�����ݵ��ԭ��������
��3�����ݵ缫��Ӧ�͵����غ�����ܽ���������������ʵ�����ϵ��
��4����⾫��ͭʱ����ͭ����������ͭ������������ͭ���ӵ�����Һ���������Һ��
��5���ҳ���EΪ������FΪ���������ҳ���������������Ϊ4.48L����״����ʱ���׳���������������Ũ�ȣ����ݵ����غ���㷴Ӧ�������ʵ����õ���������Ũ�ȣ�
���
�⣺��1�������е����̪��Һ����F�������Ժ�ɫ��˵���ü��������ӷŵ磬���Ըõ缫������������E�缫��������D�缫��������C�缫��������G�缫��������H�缫��������X�缫��������Y��������A�ǵ�Դ��������B��ԭ��صĸ�����
�ʴ�Ϊ��������
��2��D��F��X��Y ���Dz��缫��C��E�����缫���׳���CΪ����������DΪ���缫���������Һ������ͭ��Һ���������ͭ��Һʱ����������ʧ���������������ӣ�������ͭ���ӷŵ磬���Ե�ⷴӦΪ��Fe+CuSO4
Cu+FeSO4��
�ʴ�Ϊ��Fe+CuSO4
Cu+FeSO4��
��3���׳���CΪ����������DΪ���缫���������Һ������ͭ��Һ���������ͭ��Һʱ����������ʧ���������������ӣ�Fe-2e-=Fe2+��������ͭ���ӷŵ磬�缫��ӦΪCu2++2e-=Cu�����ݵ����غ��֪��ͬһʱ��C��D�缫�ϲμӷ�Ӧ�ĵ��ʻ����ɵĵ��ʵ����ʵ���֮����1��1��
�ʴ�Ϊ��1��1��
��4����⾫��ͭʱ����ͭ����������ͭ������������ͭ���ӵ���������ʼ��ɣ������жϱ�װ����GΪ���ص�������H�缫��������G�缫Ϊ��ͭ��H�缫Ϊ��ͭ�����ݵ����غ�����������ܽ������ͭ������п���������ʽ���Ҳʧ���ӣ����Ե������Һ��ͭ����Ũ�ȼ�С�����Һ��ԭ����ʵ����ʵ���Ũ�ȼ�С��
�ʴ�Ϊ����ͭ����С��
��5���ҳ���EΪ������Fe-2e-=Fe2+��FΪ�������缫��Ӧ2H++2e-=H2�������ҳ���������������Ϊ4.48L����״�������������������ʵ���=0.2mol���������ҳص���ת��Ϊ0.4mol���׳�����������ʧ���������������ӣ�Fe-2e-=Fe2+��������������Ũ��=
=0.4mol/L��
�ʴ�Ϊ��0.4��
�ʴ�Ϊ��������
��2��D��F��X��Y ���Dz��缫��C��E�����缫���׳���CΪ����������DΪ���缫���������Һ������ͭ��Һ���������ͭ��Һʱ����������ʧ���������������ӣ�������ͭ���ӷŵ磬���Ե�ⷴӦΪ��Fe+CuSO4
| ||
�ʴ�Ϊ��Fe+CuSO4
| ||
��3���׳���CΪ����������DΪ���缫���������Һ������ͭ��Һ���������ͭ��Һʱ����������ʧ���������������ӣ�Fe-2e-=Fe2+��������ͭ���ӷŵ磬�缫��ӦΪCu2++2e-=Cu�����ݵ����غ��֪��ͬһʱ��C��D�缫�ϲμӷ�Ӧ�ĵ��ʻ����ɵĵ��ʵ����ʵ���֮����1��1��
�ʴ�Ϊ��1��1��
��4����⾫��ͭʱ����ͭ����������ͭ������������ͭ���ӵ���������ʼ��ɣ������жϱ�װ����GΪ���ص�������H�缫��������G�缫Ϊ��ͭ��H�缫Ϊ��ͭ�����ݵ����غ�����������ܽ������ͭ������п���������ʽ���Ҳʧ���ӣ����Ե������Һ��ͭ����Ũ�ȼ�С�����Һ��ԭ����ʵ����ʵ���Ũ�ȼ�С��
�ʴ�Ϊ����ͭ����С��
��5���ҳ���EΪ������Fe-2e-=Fe2+��FΪ�������缫��Ӧ2H++2e-=H2�������ҳ���������������Ϊ4.48L����״�������������������ʵ���=0.2mol���������ҳص���ת��Ϊ0.4mol���׳�����������ʧ���������������ӣ�Fe-2e-=Fe2+��������������Ũ��=
| 0.2mol |
| 0.5L |
�ʴ�Ϊ��0.4��
������������һ���绯ѧ�ۺ�֪ʶ��Ŀ����Ҫ��ԭ���ԭ���͵���ԭ���ķ���Ӧ�ã��缫�жϺ͵缫��Ӧ�ǽ���ؼ���ע������л��Ե缫������������ʧ���ӷ���������Ӧ�����ջ����ǹؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ���ɶ��ȫ���ƿؾ�ϵ�д�
���ɶ��ȫ���ƿؾ�ϵ�д�
�����Ŀ
�����������ܴ���������ǣ�������
| A��Na+ Al3+ AlO2- Cl- |
| B��Mg2+ Al3+ SO42- NO3- |
| C��Ba2+ K+ SO42- NO3- |
| D��Na+ K+ ClO- S2- |
������ʵ��������������ԭ�����͵��ǣ�������
| A����ˮ��������ƽ�⣺Br2+H2O?HBr+HBrO����������������Һ����Һ��ɫ��dz |
| B����Ӧ CO+NO2?CO2+NO ����H��0������ƽ�������¶���ϵ��ɫ���� |
| C������ 2 HI?H2+I2 ��ƽ�����С�����ʹ��ϵ��ɫ���� |
| D���ϳɰ���Ӧ��Ϊ��߰��IJ��ʣ�������Ӧ��ȡ�����¶ȵĴ�ʩ |
��һ�������£���2L�ܱ������г���3mol X��g����1mol Y��g�����з�Ӧ��2X��g��+Y��g��?3Z��g��+2W��g����ijʱ�̲ⶨ�����Ũ�ȣ������ܵ��ǣ�������
| A��Z=0.75mol/L |
| B��Z=1mol/L |
| C��W=1mol/L |
| D��Z=0.8mol/L |
���е缫��Ӧʽ��ȷ���ǣ�������
| A���ȼҵ��ⱥ��ʳ��ˮʱ�������缫��ӦʽΪ 2Cl--2e-=Cl2�� |
| B������ͭʱ�����Դ�����������Ǵ�ͭ���缫��ӦʽΪ Cu-2e-=Cu2+ |
| C������ȼ�ϵ�صĸ�����Ӧʽ O2+2H2O+4e-=4OH- |
| D�����������绯��ʴ��������Ӧʽ Fe-2e-=Fe2+ |
ͭ-пԭ�����ͼ��ʾ������������ȷ���ǣ�������
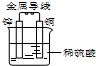

| A���������ڸ����õ��� |
| B��пΪ����������������Ӧ |
| C��ͭƬ�������ݲ��� |
| D�����Ӵ�ͭƬ��������������пƬ |
����˵������ȷ���ǣ�������
| A��һ����Ӧ�ܷ��Է�����ȡ���ڸ÷�Ӧ���Ȼ������� |
| B��ͬһ�����²�ͬ�����в�ͬ����ֵ������ϵ�Ļ��ҳ̶�Խ����ֵԽ�� |
| C���ʱ���һ���뷴Ӧ�ܷ��Է������йص����أ��������Է����еķ�Ӧ���Ƿ��ȷ�Ӧ |
| D��һ����Ӧ�ܷ��Է����У����ʱ���ر�Ĺ�ͬӰ���й� |
���и��������к�����ͬ���������ǣ�������
| A��1g H2��11.2L O2 |
| B��22.4L SO3��64g SO2 |
| C��1mol HCl��22.4L HCl |
| D��28g CO��44g CO2 |
��NAΪ�����ӵ���������ֵ������˵����ȷ���ǣ�������
| A�������£�16gCH4����10NA������ |
| B����״���£�22.4L������NA�������� |
| C����״���£�6.72LNO2��ˮ��ַ�Ӧת�Ƶĵ�����ĿΪ0.1NA |
| D��0.1mol��L-1CH3COOH��Һ�к���0.1NA��CH3COO- |