��Ŀ����
16����֪��ijһԪ����״����ɵ�����ȡ0.68g������40mL0.2mol/L��NaOH��Һ��ϼ��ȣ�ʹ����ȫˮ�������0.2mol/L�������к�ʣ��ļ��ȥ15mL���ᣮ��ȡ27.2g��������ȫȼ�պ�õ�70.4gCO2��14.4gˮ����1����������Է���������
��2�������ķ���ʽ��
��3���������ĺ˴Ź��������г���4����ҷ����֮��Ϊ3��2��2��1��д�������Ľṹ��ʽ��
���� ȡ27.2g��������ȫȼ�պ�õ�70.4gCO2��14.4gˮ����֪n��C��=n��CO2��=$\frac{70.4g}{44g/mol}$=1.6mol��m��C��=19.2g��
n��H��=$\frac{14.4g��2}{18g/mol}$=1.6mol��m��H��=1.6g��
n��O��=$\frac{27.2g-19.2g-1.6g}{16g/mol}$=0.4mol��
�ɵ�n��C����n��H����n��O��=4��4��1�����ʽΪC4H4O��
����RCOOCH3+NaOH��RCOONa+CH3OH�����������������ʵ����ɼ���������Է������������ݽṹ�ص���ж��л���Ľṹ��ʽ���Դ˽����⣮
��� �⣺��1��ȡ27.2g��������ȫȼ�պ�õ�70.4gCO2��14.4gˮ����֪n��C��=n��CO2��=$\frac{70.4g}{44g/mol}$=1.6mol��m��C��=19.2g��
n��H��=$\frac{14.4g��2}{18g/mol}$=1.6mol��m��H��=1.6g��
n��O��=$\frac{27.2g-19.2g-1.6g}{16g/mol}$=0.4mol��
�ɵ�n��C����n��H����n��O��=4��4��1�����ʽΪC4H4O��ʽ��Ϊ68��
����NaOH��Һ��Ӧ���ĵ�n��NaOH��=0.2mol/L����0.04L-0.015L��=0.005mol��
RCOOCH3+NaOH��RCOONa+CH3OH��
1 1
n 0.005mol
n��RCOOCH3��=0.005mol��
M��RCOOCH3��=$\frac{0.68g}{0.005mol}$=136g/mol����Է�������Ϊ136��
�𣺸�������Է�������Ϊ136��
��2�������ʽΪ��C4H4O��x��
x=$\frac{136}{68}$=2��
�����ʽΪC8H8O2��
�𣺸����ķ���ʽΪC8H8O2��
��3���������ĺ˴Ź��������г���4����ҷ����֮��Ϊ3��2��2��1������л�����һԪ������״����ɵ������ɷ���ʽ��֪������Ӧ���б������ṹ��ʽΪ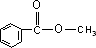 ��
��
�𣺸����Ľṹ��ʽΪ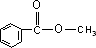 ��
��
���� ���⿼���л�����ƶϣ������ڿ���ѧ���ķ��������������������Ѷ��еȣ�ע��������غ�ĽǶȺ���ˮ����ص����ֽ����⣬����ʱע���������˼·��

��NH3��H2O��
��NH4+��H3O+��
��NH3��H3O+��
��O3��SO2��
��CO2��BeCl2��
| A�� | ȫ�� | B�� | �ۢܢ� | C�� | �������� | D�� | �ڢ� |
| A�� | Զ���ֵ��������п��ɱ����ִ����ܸ�ʴ | |
| B�� | ���������ڳ�ʪ���������� | |
| C�� | ��п��Ƭ�ȶ�����Ƭ����ʴ | |
| D�� | �������ڿ����в��ױ���ʴ |
| A�� | V��NaOH��=0ʱ��c��H+��=1��10-2mol•L-1 | |
| B�� | V��NaOH��=10mLʱ��c��H+��=1��10-7mol•L-1 | |
| C�� | V��NaOH����10mLʱ��c��Na+����c��C2O42-����c��HC2O4-�� | |
| D�� | V��NaOH����10mLʱ�������ܴ���c��Na+��=2c��C2O42-��+c��HC2O4-�� |
| A�� | ˮ�����ӻ�KW���¶ȡ�����ᣨ�Ũ�ȵĸı���ı� | |
| B�� | ���Է����еĻ�ѧ��Ӧ��һ���ǡ�H��0����S��0 | |
| C�� | ���Ѵﻯѧƽ��ķ�Ӧ�����ı�Ũ�ȣ���ƽ���ƶ�����ƽ�ⳣ����K��һ���ı� | |
| D�� | Kspֻ�����ܵ���ʵ����ʺ��¶��йأ�������Һ�е�����Ũ���� |
| A�� | ��̿�ڸ�������ˮ�����ķ�Ӧ�����ȷ�Ӧ | |
| B�� | ȼ�����㹻�Ŀ������ܳ��ȼ�� | |
| C�� | ��������������ķ�Ӧ�Ƿ��ȷ�Ӧ | |
| D�� | ��ѧ��Ӧ�Ĺ��̣��ض��������ı仯 |
| A�� | ���������Ҫ���øߴ��ȵ��ʹ��ƳɵĹ�̫���ܵ�� | |
| B�� | �赥�������������Ʒ�Ӧ����������ᷴӦ�����Թ�������� | |
| C�� | �����Ľṹ�ͽ��ʯ���ƣ��Ǿ��н�������ĻҺ�ɫ���� | |
| D�� | ��̫���ܵ�ؿɽ�̫����ֱ��ת��Ϊ���ܣ����ٻ�ʯȼ�ϵ�ʹ�ã��������� |
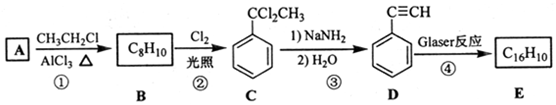
 ��D �Ļ�ѧ����Ϊ����Ȳ��
��D �Ļ�ѧ����Ϊ����Ȳ�� ����1mol E�ϳ�1��4-���������飬��������Ҫ��������4mol��
����1mol E�ϳ�1��4-���������飬��������Ҫ��������4mol��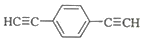 ��Ҳ�ɷ���Glaserż����Ӧ���ɾۺ���þۺϷ�Ӧ�Ļ�ѧ����ʽΪn
��Ҳ�ɷ���Glaserż����Ӧ���ɾۺ���þۺϷ�Ӧ�Ļ�ѧ����ʽΪn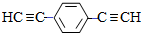 $\stackrel{����}{��}$
$\stackrel{����}{��}$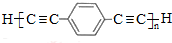 +��n-1��H2��
+��n-1��H2��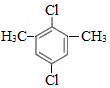 ��
�� ��
��