��Ŀ����
��֪��ͨ��״���¼ס��ҡ�������Ϊ���嵥�ʣ�A��B��C��D��E��F��G��H��Ϊ���������A��B��E��G��Ϊ���壬CΪ����Һ�壮��Ӧ�١��ڡ����ǹ�ҵ��H����Ҫ������Ӧ����Ӧ������Ҫ��ʵ������ȡ����ķ�Ӧ���йص�ת����ϵ��ͼ��ʾ����Ӧ����������ȥ������ش��������⣺

��1����Ӧ�ܵĻ�ѧ����ʽΪ ��
��2��B��E��һ�������¿ɷ�����Ӧ���÷�Ӧ����ʵ�����壬������E�Ի�������Ⱦ���÷�Ӧ�����������뻹ԭ��������ʵ���֮��Ϊ ��
��3���ں��º�ѹ�����м���һ����B���������·�����ӦB?��+����δ��ƽ������ƽ����ټ�����B����ƽ�� �ƶ������������������������ƽ�����ԭƽ����ȣ�B��ת���� �����������С�����䡱����
��4�������£����ʵ���Ũ����ͬ��A��B��D���ߵ�ˮ��Һ����ˮ�������c��OH-����С˳���ǣ���A��B��D��ʾ�� ��
��5�����Ե缫���A��D�Ļ����Һ�������ɶ����ʺ�һ�ֶ�Ԫ������M����ס�������Ԫ����ͬ����MΪ�����η��ӣ��÷�Ӧ�ķ���ʽΪ ���ڼ�����Һ��M��NaClO2���������ƣ������ʵ���֮��1��6ǡ�÷�Ӧ�����ɻ�����B��������ClO2���壬�÷�Ӧ�����ӷ���ʽΪ ��
��6����Fe��FeO��Fe3O4�Ļ������ȷ֣�����һ�ݼ���1mol/L��A��Һ100mL��ǡ��ʹ�����ȫ���ܽ⣬�ҷų�336mL������£������壬��������Һ�м���KSCN��Һ����Һ����죻��һ�ݻ�������1mol/L ��H��Һ��Ҳǡ��ʹ�����ȫ���ܽ⣬����������Һ�м���KSCN��Һ����ҺҲ����죬���������H��Һ������� mL��

��1����Ӧ�ܵĻ�ѧ����ʽΪ
��2��B��E��һ�������¿ɷ�����Ӧ���÷�Ӧ����ʵ�����壬������E�Ի�������Ⱦ���÷�Ӧ�����������뻹ԭ��������ʵ���֮��Ϊ
��3���ں��º�ѹ�����м���һ����B���������·�����ӦB?��+����δ��ƽ������ƽ����ټ�����B����ƽ��
��4�������£����ʵ���Ũ����ͬ��A��B��D���ߵ�ˮ��Һ����ˮ�������c��OH-����С˳���ǣ���A��B��D��ʾ��
��5�����Ե缫���A��D�Ļ����Һ�������ɶ����ʺ�һ�ֶ�Ԫ������M����ס�������Ԫ����ͬ����MΪ�����η��ӣ��÷�Ӧ�ķ���ʽΪ
��6����Fe��FeO��Fe3O4�Ļ������ȷ֣�����һ�ݼ���1mol/L��A��Һ100mL��ǡ��ʹ�����ȫ���ܽ⣬�ҷų�336mL������£������壬��������Һ�м���KSCN��Һ����Һ����죻��һ�ݻ�������1mol/L ��H��Һ��Ҳǡ��ʹ�����ȫ���ܽ⣬����������Һ�м���KSCN��Һ����ҺҲ����죬���������H��Һ�������
���㣺������ƶ�
ר�⣺�ƶ���
������CΪ����Һ�壬ӦΪH2O���������ӦΪH2��O2�е����ʣ���B�ܺͱ���Ӧ����C��˵��BӦΪ�⻯���ΪH2����ΪO2���ɷ�Ӧ�٢ڢۿ�֪������������Ԫ�ش��ڶ��ֻ��ϼۣ���E��G��Ϊ���������G����ˮ��Ӧ����E��˵������������ԭ��Ӧ�����֪��ΪN2��BΪNH3��EΪNO��GΪNO2��HΪHNO3����Ӧ������Ҫ��ʵ������ȡ����ķ�Ӧ��������NH3��ӦΪNH4Cl��Ca��OH��2�ķ�Ӧ����DΪNH4Cl��FΪCa��OH��2��IΪCaCl2�����ΪCl��AΪHCl����϶�Ӧ���ʵ������Լ���ĿҪ������⣮
���
�⣺CΪ����Һ�壬ӦΪH2O���������ӦΪH2��O2�е����ʣ���B�ܺͱ���Ӧ����C��˵��BӦΪ�⻯���ΪH2����ΪO2���ɷ�Ӧ�٢ڢۿ�֪������������Ԫ�ش��ڶ��ֻ��ϼۣ���E��G��Ϊ���������G����ˮ��Ӧ����E��˵������������ԭ��Ӧ�����֪��ΪN2��BΪNH3��EΪNO��GΪNO2��HΪHNO3����Ӧ������Ҫ��ʵ������ȡ����ķ�Ӧ��������NH3��ӦΪNH4Cl��Ca��OH��2�ķ�Ӧ����DΪNH4Cl��FΪCa��OH��2��IΪCaCl2�����ΪCl��AΪHCl��
��1����Ӧ��Ϊʵ�����Ʊ������ķ�Ӧ������ʽΪ2NH4Cl+Ca��OH��2
CaCl2+2H2O+2NH3�����ʴ�Ϊ��2NH4Cl+Ca��OH��2
CaCl2+2H2O+2NH3����
��2��BΪNH3��EΪNO���������Ϣ��֪��Ӧ�ķ���ʽΪ4NH3+6NO=4N2+6H2O���ɷ���ʽ��֪���������뻹ԭ��������ʵ���֮��Ϊ2��3���ʴ�Ϊ��2��3��
��3��?��+���ķ�ӦΪ2NH3?N2+3H2����ƽ����ټ�����NH3����Ӧ��Ũ��������ƽ���������ƶ�������ƽ�����ԭƽ����ȣ�����ѹǿ���䣬��Ũ�Ȳ��䣬ƽ��״̬��ͬ��ת���ʲ��䣬
�ʴ�Ϊ�������䣻
��4�����ʵ���Ũ����ͬ��A��HCl����B��NH3����D��NH4Cl�����ߵ�ˮ��Һ��D�ٽ�ˮ�ĵ��룬��AΪǿ�ᣬ��Ũ��ʱ�������������Ũ�Ƚϴ���B��Ƚϣ�ˮ�ĵ���̶Ƚ�С������ˮ�������c��OH-����С˳����D��B��A��
�ʴ�Ϊ��D��B��A��
��5�����Ե缫���NH4Cl��HCl�Ļ����Һ��������H2���ʺ�һ�ֶ�Ԫ������M����ס�������Ԫ����ͬ����MΪ�����η��ӣ��������غ��֪ӦΪNCl3�����ⷽ��ʽΪNH4Cl+2HCl
NCl3+3H2���ڼ�����Һ��NCl3��NaClO2���������ƣ������ʵ���֮��1��6ǡ�÷�Ӧ�����ɻ�����NH3��������ClO2���壬��Ӧ�����ӷ���ʽΪNCl3+6ClO2-+3H2O�T6ClO2+3Cl-+3OH-+NH3��
�ʴ�Ϊ��NH4Cl+2HCl
NCl3+3H2��NCl3+6ClO2-+3H2O�T6ClO2+3Cl-+3OH-+NH3��
��6����һ������Fe��FeO��Fe3O4�Ļ�����У�����1mol?L-1HCl����Һ100mL��ǡ��ʹ�����ȫ���ܽ⣬�ҷų�336mL����״���£������壬��������Һ�м���KSCN��Һ����Һ��ɫ���֣�˵����Һ����ΪFeCl2����ClԪ���غ��֪n��FeCl2��=0.05mol��ͬʱת�Ƶ���Ϊ2��
=0.03mol��
��ȡͬ������Fe��FeO��Fe3O4��������1mol?L-1HNO3��Һ��Ҳǡ��ʹ�����ȫ���ܽ⣬����������Һ�м���KSCN��Һ����ҺҲ��ɫ���֣�������0.05molFe��NO3��2��ͬʱ����NO
=0.01mol����NԪ���غ��֪��Ҫ��HNO3���ʵ���Ϊ0.05mol��2+0.01mol=0.11mol������������Ϊ
=0.11L=110mL��
�ʴ�Ϊ��110��
��1����Ӧ��Ϊʵ�����Ʊ������ķ�Ӧ������ʽΪ2NH4Cl+Ca��OH��2
| ||
| ||
��2��BΪNH3��EΪNO���������Ϣ��֪��Ӧ�ķ���ʽΪ4NH3+6NO=4N2+6H2O���ɷ���ʽ��֪���������뻹ԭ��������ʵ���֮��Ϊ2��3���ʴ�Ϊ��2��3��
��3��?��+���ķ�ӦΪ2NH3?N2+3H2����ƽ����ټ�����NH3����Ӧ��Ũ��������ƽ���������ƶ�������ƽ�����ԭƽ����ȣ�����ѹǿ���䣬��Ũ�Ȳ��䣬ƽ��״̬��ͬ��ת���ʲ��䣬
�ʴ�Ϊ�������䣻
��4�����ʵ���Ũ����ͬ��A��HCl����B��NH3����D��NH4Cl�����ߵ�ˮ��Һ��D�ٽ�ˮ�ĵ��룬��AΪǿ�ᣬ��Ũ��ʱ�������������Ũ�Ƚϴ���B��Ƚϣ�ˮ�ĵ���̶Ƚ�С������ˮ�������c��OH-����С˳����D��B��A��
�ʴ�Ϊ��D��B��A��
��5�����Ե缫���NH4Cl��HCl�Ļ����Һ��������H2���ʺ�һ�ֶ�Ԫ������M����ס�������Ԫ����ͬ����MΪ�����η��ӣ��������غ��֪ӦΪNCl3�����ⷽ��ʽΪNH4Cl+2HCl
| ||
�ʴ�Ϊ��NH4Cl+2HCl
| ||
��6����һ������Fe��FeO��Fe3O4�Ļ�����У�����1mol?L-1HCl����Һ100mL��ǡ��ʹ�����ȫ���ܽ⣬�ҷų�336mL����״���£������壬��������Һ�м���KSCN��Һ����Һ��ɫ���֣�˵����Һ����ΪFeCl2����ClԪ���غ��֪n��FeCl2��=0.05mol��ͬʱת�Ƶ���Ϊ2��
| 0.336L |
| 22.4L/mol |
��ȡͬ������Fe��FeO��Fe3O4��������1mol?L-1HNO3��Һ��Ҳǡ��ʹ�����ȫ���ܽ⣬����������Һ�м���KSCN��Һ����ҺҲ��ɫ���֣�������0.05molFe��NO3��2��ͬʱ����NO
| 0.03mol |
| 5-2 |
| 0.11mol |
| 1L/mol |
�ʴ�Ϊ��110��
���������⿼��������ƶϣ�������ѧ���ķ��������ͼ��������Ŀ��飬Ϊ�߿��������ͣ�ע�������Ŀ�ƶϵ�ͻ�ƿڣ���C�ͷ�Ӧ��ת����ϵ�������Ƶķ��������ƶϣ���Ŀ�ѶȽϴ�

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
�Ȼ���BCl3�����۵�Ϊ-107�棬�е�Ϊ12.5�棬��������м����֮��ļн�Ϊ120�㣬����ˮ�⣬�й�������ȷ���ǣ�������
| A���Ȼ���Һ̬ʱ�ܵ������̬ʱ������ |
| B���Ȼ���ӵ�ˮ��ʹ��Һ��pH���� |
| C���Ȼ�����ӳ�ƽ���������Σ����Ǽ��Է��� |
| D���Ȼ���B-Cl֮����sp3�γɵĦҼ� |
����Ԫ�������ɷ��������ƶϣ����в���ȷ���ǣ�������
| A����֪Ra�ǵ������ڡ���A���Ԫ�أ���Ra��OH��2�ļ��Ա�Mg��OH��2�ļ���ǿ |
| B����֪As�ǵ������ڡ���A���Ԫ�أ���AsH3���ȶ��Ա�NH3���ȶ���ǿ |
| C����֪Cs��ԭ�Ӱ뾶��Na��ԭ�Ӱ뾶��Cs��ˮ��Ӧ��Na��ˮ��Ӧ������ |
| D����֪Cl�ĺ˵������Al�ĺ˵������Cl��ԭ�Ӱ뾶��Al��ԭ�Ӱ뾶С |
��������е�4��Hȫ��������ȡ�����ɵ���ͼ��ʾ�ķ��ӣ��Ը÷��ӵ���������ȷ���ǣ�������

| A������ʽΪC25H20 |
| B������������ԭ���п��ܴ���ͬһƽ�� |
| C������������ԭ�Ӳ����ܴ���ͬһƽ�� |
| D���÷�����ȫ�ӳ�ʱ��Ҫ12mol��H2 |
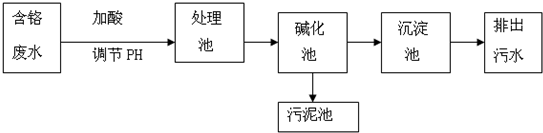
 ijͬѧ�����ϵ�֪��-AgI��һ�ֹ��嵼�壬�����ʺܸߣ���Ϊ���о���-AgI������Ag+���ӵ��绹��I-���ӵ��磬���һ����ͼ��ʾ����Ǧ��������Դ���ĵ绯ѧʵ�飮
ijͬѧ�����ϵ�֪��-AgI��һ�ֹ��嵼�壬�����ʺܸߣ���Ϊ���о���-AgI������Ag+���ӵ��绹��I-���ӵ��磬���һ����ͼ��ʾ����Ǧ��������Դ���ĵ绯ѧʵ�飮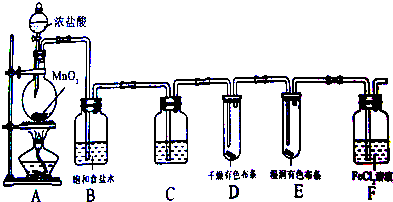
 ��ͼ��ʾ��ˮ�����Թ�����һö��������������۲죺
��ͼ��ʾ��ˮ�����Թ�����һö��������������۲죺