��Ŀ����
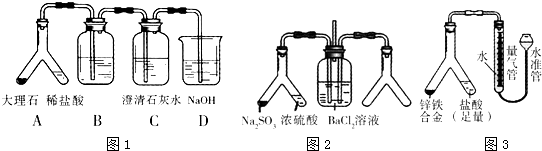
����Y�ι�������������Ͽ��Խ�������ʵ�飨�̶�װ���ԣ����������ش��������⣺
��1��ʵ��Ŀ�ģ�̽��CO2�����ʯ��ˮ�ķ�Ӧ��
��װ��B��ʢ�ŵ��Լ�Ϊ ��
����ͬѧ��Ϊ���Բ��װ��D�����Ƿ�ͬ��ù۵㣿 ����ǡ�����ԭ���� ��
��2��ʵ��Ŀ�ģ�̽��SO2��BaCl2��Ӧ����������������
����������ͨ���Ȼ�����Һ�в���������������ͨ����һ���������Բ�����ɫ��������ͼʾ�Ҳ�Y�ι���Ӧ���õ�ҩƷ�� �� ����Ҫʱ���Լ��ȣ����ó����Ļ�ѧʽΪ ��
��3��ʵ��Ŀ�ģ�п���Ͻ����������IJⶨ��
�ٶ�ȡ������������ʱ��������ˮ���е�Һ������������е�Һ�棬Ӧ��ȡ�Ĵ�ʩ�� ��
�����Ƶ�п���Ͻ������Ϊ0.117g���������г�����Ϊ1.00mL��ĩ����Ϊ45.80mL����Ͻ������ĺ���Ϊ %������2λС�����������������ɱ�״������

��1��ʵ��Ŀ�ģ�̽��CO2�����ʯ��ˮ�ķ�Ӧ��
��װ��B��ʢ�ŵ��Լ�Ϊ
����ͬѧ��Ϊ���Բ��װ��D�����Ƿ�ͬ��ù۵㣿
��2��ʵ��Ŀ�ģ�̽��SO2��BaCl2��Ӧ����������������
����������ͨ���Ȼ�����Һ�в���������������ͨ����һ���������Բ�����ɫ��������ͼʾ�Ҳ�Y�ι���Ӧ���õ�ҩƷ��
��3��ʵ��Ŀ�ģ�п���Ͻ����������IJⶨ��
�ٶ�ȡ������������ʱ��������ˮ���е�Һ������������е�Һ�棬Ӧ��ȡ�Ĵ�ʩ��
�����Ƶ�п���Ͻ������Ϊ0.117g���������г�����Ϊ1.00mL��ĩ����Ϊ45.80mL����Ͻ������ĺ���Ϊ
��������1��̽��CO2�����ʯ��ˮ�ķ�Ӧ����Ҫ�ų���������ĸ��ţ�������̼�����к����Ȼ�����Ŷ�����̼�ļ��飬������̼������Ⱦ���壬����Ҫβ�����գ�
��2��SO2��BaCl2��Ӧ������������Һ�б�����ڴ�����SO32-��
��3���ٶ�ȡ������������ʱ��Ϊ��С��Ӧʹˮ�ܡ���������Һ����ƽ��
�ڼ���Ͻ���FeΪxmol��ZnΪymol���з���ʽ��ɽ����⣮
��2��SO2��BaCl2��Ӧ������������Һ�б�����ڴ�����SO32-��
��3���ٶ�ȡ������������ʱ��Ϊ��С��Ӧʹˮ�ܡ���������Һ����ƽ��
�ڼ���Ͻ���FeΪxmol��ZnΪymol���з���ʽ��ɽ����⣮
����⣺��1����̽��CO2�����ʯ��ˮ�ķ�Ӧ��������̼�����к����Ȼ������壬����Ŷ�����̼�ļ��飬��Ҫͨ������̼��������Һ��ȥ�Ȼ��⣬
�ʴ�Ϊ������̼��������Һ��
�ڶ�����̼���岻����Ⱦ���壬���Բ�������װ�ã��ʴ�Ϊ���ǣ�������̼��һ�������壬����Ҫ���գ�
��2��SO2��BaCl2��Ӧ������������Һ�б�����ڴ�����SO32-���Ҳ�Y����Ӧ�����ɼ�����������������壬���Ǽ������壬��Һ�д��ڴ�����SO32-���������������壬��Һ�п�����SO42-�������ɵij�������ΪBaSO3��BaSO4��
�ʴ�Ϊ��Ũ��ˮ����ʯ�ң����� NaOH����ʯ�ң���BaSO3��BaSO4��
��3���ٶ�ȡ������������ʱ��Ϊ��С��Ӧʹˮ�ܡ���������Һ����ƽ���ʴ�Ϊ��̧�ߣ����ƶ���ˮ��λ�ã�ʹˮ�ܡ���������Һ����ƽ��
����Ͻ���FeΪxmol��ZnΪymol������
56x+65y=0.117
x+y=
=0.002mol
��֮�ã�x=0.001444��y=0.000556��
��Ͻ������ĺ���Ϊ
=69.14%��
�ʴ�Ϊ��69.14%��
�ʴ�Ϊ������̼��������Һ��
�ڶ�����̼���岻����Ⱦ���壬���Բ�������װ�ã��ʴ�Ϊ���ǣ�������̼��һ�������壬����Ҫ���գ�
��2��SO2��BaCl2��Ӧ������������Һ�б�����ڴ�����SO32-���Ҳ�Y����Ӧ�����ɼ�����������������壬���Ǽ������壬��Һ�д��ڴ�����SO32-���������������壬��Һ�п�����SO42-�������ɵij�������ΪBaSO3��BaSO4��
�ʴ�Ϊ��Ũ��ˮ����ʯ�ң����� NaOH����ʯ�ң���BaSO3��BaSO4��
��3���ٶ�ȡ������������ʱ��Ϊ��С��Ӧʹˮ�ܡ���������Һ����ƽ���ʴ�Ϊ��̧�ߣ����ƶ���ˮ��λ�ã�ʹˮ�ܡ���������Һ����ƽ��
����Ͻ���FeΪxmol��ZnΪymol������
56x+65y=0.117
x+y=
| 0.0448L |
| 22.4L/mol |
��֮�ã�x=0.001444��y=0.000556��
��Ͻ������ĺ���Ϊ
| 0.001444��56 |
| 0.117 |
�ʴ�Ϊ��69.14%��
���������⿼�����������ʵķ����жϣ�ʵ����֤�������ʵ�ʵ����ƺ�����װ�÷�������ѧ����ʽ���㣬��Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ѧ�ڸ�ϰһ��ͨѧϰ�ܶ�Ա��ĩ������ӱ����������ϵ�д�
ѧ�ڸ�ϰһ��ͨѧϰ�ܶ�Ա��ĩ������ӱ����������ϵ�д� â���̸����������������ϵ�д�
â���̸����������������ϵ�д�
�����Ŀ