��Ŀ����
����Se������������һ����Ԫ�أ��䵥�ʿ������ƹ������衢��ӡ�������ĵȵȣ���ҵ����ȡ���ķ���֮һ��������������ƴ��������Ĺ�ҵ���ϣ��õ������ᣨH2SeO3�����������ᣨH2SeO4�����������ٽ����������Ṳ�ȣ���H2SeO4ת��ΪH2SeO3����Ҫ��ӦΪ2HCl+H2SeO4�TH2SeO3+H2O+Cl2����Ȼ������Һ��ͨ��SO2����Ԫ�ػ�ԭΪ�������������ݴ���ȷ���ж�Ϊ��������
| A��H2SeO4�������Ա�Cl2�� |
| B��SeO2�������Ա�SO2�� |
| C��H2SeO4�������Ա�H2SeO3ǿ |
| D������1mol�����������ᡢSO2��ˮ��1mol |
���㣺�����ԡ���ԭ��ǿ���ıȽ�
ר�⣺������ԭ��Ӧר��
������������ԭ��Ӧ�������������Դ�����������������ԣ���ԭ���Ļ�ԭ�Դ��ڻ�ԭ����Ļ�ԭ�ԣ����Ԫ�صĻ��ϼ۵ı仯�������ת�Ƶ���Ŀ��
���
�⣺A����Ӧ��SeԪ�ػ��ϼ۽��ͣ�H2SeO4Ϊ��������ClԪ�ػ��ϼ����ߣ�Cl2Ϊ�������������ԭ��Ӧ�������������Դ�����������������ԣ���H2SeO4�������Ա�����ǿ����A����
B������������ͨ����������Һ�г��ֵ������ij�������˵�������Զ����������������ᣩ���������ԣ�����������л�ԭ�ԣ�����������������Դ��ڶ�������B����
C��2HCl+H2SeO4��H2SeO3+Cl2+H2O��Ӧ�У�H2SeO4Ϊ��������H2SeO3Ϊ��ԭ�����H2SeO3�������Ա�H2SeO4������C��ȷ��
D������������ͨ����������Һ�г��ֵ������ij�������Ӧ��SeԪ�ػ��ϼ���+4�۽��͵�0�ۣ�SԪ�ػ��ϼ���+4�����ߵ�+6�ۣ�������1mol��Ҫ��H2SeO31mol��SO22mol����D����
��ѡC��
B������������ͨ����������Һ�г��ֵ������ij�������˵�������Զ����������������ᣩ���������ԣ�����������л�ԭ�ԣ�����������������Դ��ڶ�������B����
C��2HCl+H2SeO4��H2SeO3+Cl2+H2O��Ӧ�У�H2SeO4Ϊ��������H2SeO3Ϊ��ԭ�����H2SeO3�������Ա�H2SeO4������C��ȷ��
D������������ͨ����������Һ�г��ֵ������ij�������Ӧ��SeԪ�ػ��ϼ���+4�۽��͵�0�ۣ�SԪ�ػ��ϼ���+4�����ߵ�+6�ۣ�������1mol��Ҫ��H2SeO31mol��SO22mol����D����
��ѡC��
�����������ۺϿ���������ԭ��Ӧ��������ѧ���ķ�����������ѧ�����Ŀ��飬Ϊ�߿��������ͣ��Ѷ��еȣ�ע���Ԫ�ػ��ϼ۵ĽǶȷ�����ظ�������ʵ����ʣ�

��ϰ��ϵ�д�
�����Ŀ
��OH-������ͬ�����������������ǣ�������
| A��NH3 |
| B��Na+ |
| C��F- |
| D��NH4+ |
�����йػ�ѧ�������������ȷ���ǣ�������
| A����ϩ�Ľṹ��ʽ��CH2CH2 |
B��CH4���ӵı���ģ�ͣ� |
C����ȩ���ӵĵ���ʽ��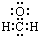 |
D��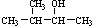 2-��-3-���� 2-��-3-���� |
���и���������ˮ��Һ���ܹ�����������ǣ�������
| A��Na+��H+��K+��CO32- |
| B��Mg2+��NH4+��SO42-��Cl- |
| C��Cu2+��K+��OH-��SO42- |
| D��K+��H+��OH-��NO3- |
�����й�ԭ��ص������У�������ǣ�������
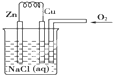
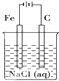
| A��ԭ����ǽ���ѧ��ת��Ϊ���ܵ�װ�� |
| B���õ������ӵ����ֲ�ͬ����ͬʱ����Һ���У����γ�ԭ��� |
| C����ԭ����У�����������һ���Ǹ���������������Ӧ |
| D����ԭ��طŵ�ʱ���������Һ�е��������������ƶ� |
����������ԭ��Ӧ��ˮ��Һ�в��ܴ���������������ǣ�������
| A��Ca2+��Br-��Fe3+��CO32- |
| B��Fe2+��Cl-��NO3-��H+ |
| C��Fe3+��Ca2+��H+��NO3- |
| D��NO3-��OH-��Cl-��Ba2+ |
������Һ���������ʵ���Ũ�ȹ�ϵ��ȷ���ǣ�������
| A�������£���0.01mol?L-1NH4HSO4��Һ�еμ�NaOH��Һ�����ԣ�c��SO42-����c��Na+����c��NH4+����c��OH-��=c��H+�� |
| B����NaHY��ˮ��Һ�Լ��ԣ�����Һ������Ũ�ȹ�ϵΪ��c��Na+����c��OH-����c��H+����c��HY-�� |
| C��0.1 mol/L Na2CO3��Һ��c��OH-��=c��HCO3-��+c��H+��+2c��H2CO3�� |
| D�����ʵ�������0.1mol��HCN��NaCN��Ϻ����1L��Һ��c��HCN��+c��CN-��=0.1 mol?L-1 |
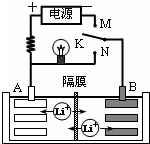 ��ͼ��һ�ֿɳ�������ӵ�س�ŵ�Ĺ���ʾ��ͼ���ŵ�ʱ�õ�صĵ缫��ӦʽΪ��������LixC6-xe-=C6+xLi+��LixC6��ʾ�ԭ��Ƕ��ʯī�γɵĸ��ϲ��ϣ�������Li1-xMnO2+xLi++xe-=LiMnO2��LiMnO2��ʾ���ԭ�ӵĶ������̣��������й�˵����ȷ���ǣ�������
��ͼ��һ�ֿɳ�������ӵ�س�ŵ�Ĺ���ʾ��ͼ���ŵ�ʱ�õ�صĵ缫��ӦʽΪ��������LixC6-xe-=C6+xLi+��LixC6��ʾ�ԭ��Ƕ��ʯī�γɵĸ��ϲ��ϣ�������Li1-xMnO2+xLi++xe-=LiMnO2��LiMnO2��ʾ���ԭ�ӵĶ������̣��������й�˵����ȷ���ǣ�������| A���������䡢�ŵ���������ٴ���3����ʽ������ת�� | |||
| B��K��M���ʱ��A��������������ԭ��Ӧ | |||
| C��K��N���ʱ��Li+��A����Ǩ�Ƶ�B���� | |||
D���õ�ص��ܷ�ӦʽΪLi1-xMnO2+LixC6
|