��Ŀ����
10�������й����ʵ����ʻ���;��˵���У���ȷ���ǣ���������Cl2��Ư���ԣ�������ʹ�������ɫ������ɫ��
���ڡ��������ʵ���У�Ũ����������ˮ�Ժ�ǿ�����ԣ�
��ʵ���ҴӺ�����ȡ���ʵ�ķ�����ȡ��һ����һ�ܽ⡫����һ��ȡ��
����ʵ�����У�����Ũ��������������
�ݽ�Ũ�������ʢˮ����Ͳ�н���ϡ�ͣ�
������ˮ��Һ����Ϊľ�ķ������
�����ײ�������ֱ����10-9m��10һ7m֮�䣬������ײ������ڽ��壮
| A�� | �ۢޢ� | B�� | �٢ݢ� | C�� | �ڢۢ� | D�� | �ڢܢ� |
���� ������������Ư���ԣ�����Ư���Ե���������ˮ��Ӧ���ɵĴ����
���ڡ��������ʵ���У�Ũ����ʹ����̼����������������ԭ��Ӧ���ɶ����������壻
�۵���������ʽ���ڣ���Ҫ��������������
�ܶ���������Ũ�����Ӧ��
����Ͳ��������ϡ����Һ��
�����ƾ�����ȼ�����ʣ�
�߽������ڷ�ɢϵ��
��� �⣺������������Ư���ԣ�����Ư���Ե���������ˮ��Ӧ���ɵĴ����ᣬ�������������ʹ��ɫ������ɫ���ʴ���
���ڡ��������ʵ���У�Ũ����ʹ����̼����������������ԭ��Ӧ���ɶ����������壬Ũ����������ˮ�Ժ�ǿ�����ԣ�����ȷ��
�۵���������ʽ���ڣ���Ҫ����������������Ӻ�����ȡ���ʵ�ķ����ǣ�ȡ�������ա��ܽ�����ˡ���������ȡ���ʴ���
�ܶ���������Ũ�����Ӧ������Ũ�����������������壬����ȷ��
����Ͳ��������ϡ����Һ��ֻ��������ȡһ�������Һ�壬�ʴ���
�����Ʋ�ȼ�գ�������ȼ�����ʣ�����Ϊľ�ķ����������ȷ��
�߽������ڷ�ɢϵ�����ײ����ڽ��壬�ʴ���
��ѡD��
���� ���⿼���Ϊ�ۺϣ�Ϊ��Ƶ���㣬��Ƕȿ���Ԫ�ػ�����֪ʶ��������ѧ����˫���Ŀ��飬����������ѧ�����õĿ�ѧ���������ѧ����ѧϰ�Ļ����ԣ��ѶȲ���

| A�� | 127I��131I��Ϊͬ�������� | B�� | 137Cs�ĺ˵����Ϊ137 | ||
| C�� | ��235����������143 | D�� | ���ˮ�ķ�Ӧ������ˮ�ķ�Ӧ���� |
| ѡ�� | ������ | ���� |
| A | ������Al3+����Һ�У�K+��Na+��NO3-��CO32- | ���ܴ������棬����Al2��CO3��3�������� |
| B | ������Fe3+����Һ�У�K+��Mg2+��I-��NO3- | ���ܴ������棬��2Fe3++2I-=2Fe2++I2 |
| C | ��ˮ�����c��H+��=1��10-14mol/L����Һ�У� Ca2+��NO3-��HCO3-��Cl- | ���ܴ������棬����Һ�����ԣ���HCO3-��Ӧ����CO2���� |
| D | ʹ��̪������Һ�У� Na+��K+��SO32-��S2- | ���ܴ������棬��SO32-��S2-��Ӧ��������ɫ��S���� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ��������̼����ƺ��Ȼ��Ƶ�ˮ������Ӳˮ | |
| B�� | �����о�ˮ���ã���������������Ӳˮ������ʹӲˮ���� | |
| C�� | �����ӽ���������Ӳˮ��ֻ��������ʱӲˮ��������������Ӳˮ | |
| D�� | ֻ������ʱӲ�ȵ�ˮ��������кɱ����ˮ |
| A�� | NH4+��NO3-��Al3+��K+ | B�� | Na+��NO3-��S2-��K+ | ||
| C�� | MnO4-��SO32-��Na+��K+ | D�� | HCO3-��SO42-��Na+��K+ |
| A�� | ��֪������0.1mol•L-1NaClO��ҺpHΪ9.7��ˮ�ⷽ��ʽClO��+H2O?HClO+OH����ƽ�ⳣ��K��10��7.6 | |
| B�� | ��0.2 mol•L-1��������0.1 mol•L-1��KAlO2��Һ�������ϣ�����Һ������Ũ����С�����˳��Ϊ��c��OH-����c��Al3+����c��H+����c��K+����c��Cl-�� | |
| C�� | pH=5��HCOOH��Һ��pH=5��NH4NO3��Һ�У�c��H+������� | |
| D�� | 25��ʱ��pH=4��Ũ�Ⱦ�Ϊ0.1mol•L-1��CH3COOH��CH3COONa�����Һ��c��CH3COO-��+c��OH-����c��CH3COOH��+c��H+�� |
| X | ||||
| Z | W |
| A�� | X������ɵĻ���������п��ܺ��м��Թ��ۼ��ͷǼ��Թ��ۼ� | |
| B�� | ��ҵ���õ��Y��W��ɵĻ��������Ʊ�Y | |
| C�� | Z��W��Ԫ�ص���̬�⻯���У�W����̬�⻯����ȶ� | |
| D�� | X��Y��Z��WԪ������������Ӧ��ˮ������������ǿ����HWO4 |
| A�� | ���Ӱ뾶��Y2-��Z+ | |
| B�� | ��̬�⻯���ȶ��ԣ�HW��H2R | |
| C�� | XW4�����и�ԭ�Ӿ�����8�����ȶ��ṹ | |
| D�� | Y��Z��R����Ԫ����ɵĻ�����ֻ�������Ӽ� |
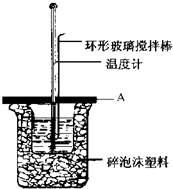 ʵ������50mL 0.50mol•L-1���ᡢ50mL 0.55mol•L-1NaOH��Һ������ͼװ�ã����вⶨ�к��ȵ�ʵ�飮�������������������Һ���ܶȶ���1g/cm3����֪�кͺ�������Һ�ı�����c=4.18J/��g•�棩��Ϊ�˼����к��ȣ�ʵ��ʱ��������������У�����ţ�ACE��
ʵ������50mL 0.50mol•L-1���ᡢ50mL 0.55mol•L-1NaOH��Һ������ͼװ�ã����вⶨ�к��ȵ�ʵ�飮�������������������Һ���ܶȶ���1g/cm3����֪�кͺ�������Һ�ı�����c=4.18J/��g•�棩��Ϊ�˼����к��ȣ�ʵ��ʱ��������������У�����ţ�ACE��