��Ŀ����
ij����ij�ӵ������м��������������ŷŵĹ�ҵ��ˮ�й�����K+��Ag+��Fe3+��Cl-��OH-��NO3-�������ӣ�
��1���׳��ķ�ˮ���Գʼ��ԣ��ʼ׳���ˮ�к��е����������� ��
��2���ҳ��ķ�ˮ�к���������������ӣ��������һ������ �������̿�����������������������ۡ��������Ի������еĽ����� ����Ϊ �������ӷ���ʽ��ʾ��
��3���������һ�������ǽ��׳����ҳ��ķ�ˮ���ʵ��ı�����ϣ�����ʹ��ˮ�е� �������ӷ��ţ�ת��Ϊ�����������˺�ķ�ˮ����Ҫ���� ���������ӷ��ţ����Կ���������ũ�
��1���׳��ķ�ˮ���Գʼ��ԣ��ʼ׳���ˮ�к��е�����������
��2���ҳ��ķ�ˮ�к���������������ӣ��������һ������
��3���������һ�������ǽ��׳����ҳ��ķ�ˮ���ʵ��ı�����ϣ�����ʹ��ˮ�е�
���㣺���ӹ�������
ר�⣺���ӷ�Ӧר��
�������׳��ķ�ˮ���Գʼ��ԣ�����Һ�к��д�����OH-����OH-���ӷ�Ӧ��Ag+��Fe3+���ܴ������棬������Һ�����Կ�֪�׳���ˮ��Ӧ����K+���ҳ��к���Ag+��Fe3+������Ag+��Fe3+��Ӧ��Cl--��OH-���ܹ��棬������Һ�����Կ�֪�ҳ�����NO3-��
�ɴ˿�֪���׳�����K+��Cl-��OH-���ҳ�����Ag+��Fe3+��NO3-�����������к��е�������ɼ����ʽ����⣮
�ɴ˿�֪���׳�����K+��Cl-��OH-���ҳ�����Ag+��Fe3+��NO3-�����������к��е�������ɼ����ʽ����⣮
���
�⣺��1���׳���ˮ�ʼ��ԣ�����Һ�к��д�����OH-����OH-���ӷ�Ӧ��Ag+��Fe3+���ܴ������棬������Һ�����Կ�֪�׳���ˮ��Ӧ����K+���ҳ��к���Ag+��Fe3+������Ag+��Fe3+��Ӧ��Cl--��OH-���ܹ��棬������Һ�����Կ�֪�ҳ�����NO3-��
�ɴ˿�֪���׳�����K+��Cl--��OH-���ҳ�����Ag+��Fe3+��NO3-��
�ʴ�Ϊ��K+��Cl-��OH-��
��2���Һ��еĽ���������K+��Ag+��Fe3+�����뵥��Fe�����û���Ag���������ӷ�ӦΪFe+2Ag+=2Ag+Fe2+���ʴ�Ϊ�����ۣ�Ag��Fe+2Ag+=2Ag+Fe2+��
��3�����׳����ҳ��ķ�ˮ���ʵ��ı�����ϣ�������AgCl��Fe��OH��3���������ɳ�����������Ag+��Fe3+��Cl--��OH-�ȣ������˺�ķ�ˮ��ҪKNO3��
�ʴ�Ϊ��Ag+��Fe3+��Cl-��OH-��KNO3��
�ɴ˿�֪���׳�����K+��Cl--��OH-���ҳ�����Ag+��Fe3+��NO3-��
�ʴ�Ϊ��K+��Cl-��OH-��
��2���Һ��еĽ���������K+��Ag+��Fe3+�����뵥��Fe�����û���Ag���������ӷ�ӦΪFe+2Ag+=2Ag+Fe2+���ʴ�Ϊ�����ۣ�Ag��Fe+2Ag+=2Ag+Fe2+��
��3�����׳����ҳ��ķ�ˮ���ʵ��ı�����ϣ�������AgCl��Fe��OH��3���������ɳ�����������Ag+��Fe3+��Cl--��OH-�ȣ������˺�ķ�ˮ��ҪKNO3��
�ʴ�Ϊ��Ag+��Fe3+��Cl-��OH-��KNO3��
���������⿼�����ӹ������⣬Ϊ��Ƶ���㣬ע���жϼ��ҳ���ˮ�ɷֵĽǶ��Լ����ӵ����ʣ����ظ��ֽⷴӦ�����ӹ��濼�飬��Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
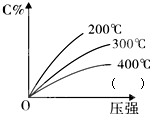 ���淴ӦmA���̣�+nB������?eC������+fD����������Ӧ�����У���������������ʱ��C�İٷֺ�����C%�����¶Ⱥ�ѹǿ�Ĺ�ϵ��ͼ������������ȷ���ǣ�������
���淴ӦmA���̣�+nB������?eC������+fD����������Ӧ�����У���������������ʱ��C�İٷֺ�����C%�����¶Ⱥ�ѹǿ�Ĺ�ϵ��ͼ������������ȷ���ǣ�������| A����ƽ����������C%���� |
| B����ƽ��������£�ƽ������ |
| C����ƽ�������A����������ƽ�������ƶ� |
| D����ѧ����ʽ��n��e+f |
��pH=3������a L�ֱ�������������Һ��Ϻ��Һ�������ԣ�����a��b��c��d�Ĺ�ϵ��ȷ���ǣ�������
��1��10-3 mol?L-1�İ�ˮb L
��c��OH-��=1��10-3 mol?L-1�İ�ˮc L
��c��OH-��=1��10-3 mol?L-1��Ba��OH��2��Һd L��
��1��10-3 mol?L-1�İ�ˮb L
��c��OH-��=1��10-3 mol?L-1�İ�ˮc L
��c��OH-��=1��10-3 mol?L-1��Ba��OH��2��Һd L��
| A��c��d=a��b |
| B��c��d��a=b |
| C��c��d��a��b |
| D��c=d��a=b |
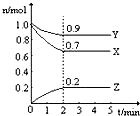 ij�¶�ʱ��2L�������������ʵ����ʵ�����ʱ��ı仯������ͼ��ʾ����ͼ�����ݷ������÷�Ӧ�Ļ�ѧ����ʽ�ͷ�Ӧ��ʼ��2minĩZ��ƽ�����ʷֱ�Ϊ��������
ij�¶�ʱ��2L�������������ʵ����ʵ�����ʱ��ı仯������ͼ��ʾ����ͼ�����ݷ������÷�Ӧ�Ļ�ѧ����ʽ�ͷ�Ӧ��ʼ��2minĩZ��ƽ�����ʷֱ�Ϊ��������| A��3X+Y?2Z�� 0.05 mol/��L?min�� |
| B��2X+Y?2Z�� 0.1 mol/��L?min�� |
| C��3X+Y?2Z�� 0.1 mol/��L?min�� |
| D��X+3Y?2Z�� 0.05 mol/��L?min�� |
��Һ����ˮ���������c��OH-��=1��10-12mol/L���������������Һ��һ���ܴ���������������ǣ�������
| A��Fe2+ Na+ NO3- Cl- |
| B��K+Na+ Cl-AlO2- |
| C��K+ Ba2+ Cl- NO3- |
| D��K+ NH4+SO42- CO32- |
������ʵ�У���������ɳ����ԭ�����͵��ǣ�������
| A������ơ�ƺ�ƿ�����Ϸ��������ĭ |
| B��ʵ�����г����ű���ʳ��ˮ�ķ����ռ����� |
| C����2HI��g��?I2��g��+H2��g����ƽ����ϵ����ѹǿ��ʹ��ɫ���� |
| D����ҵ����������Ĺ�����ʹ�ù����Ŀ�������߶�������������� |
��ȥþ���е��������ۣ���ѡ�ã�������
| A������ | B��ˮ |
| C������ | D������������Һ |
 ��
�� ��
�� ��
�� ��
��