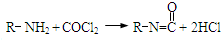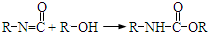��Ŀ����
һ���ѧ�о��ɹ�������ͭ�������CuMn2O4�����ڳ����´����������е�һ����̼�ͼ�ȩ��HCHO����
��1����һ�����ʵ���Ũ�ȵ�Cu��NO3��2 ��Mn��NO3��2 ��Һ�м���Na2CO3 ��Һ�����ó������������գ����Ƶ�CuMn2O4��
��Mn2+��̬�ĵ����Ų�ʽ�ɱ�ʾΪ ��
��NO3-�Ŀռ乹���� ����������������
��2����ͭ��������Ĵ��£�CO ������ΪCO2��HCHO ������ΪCO2 ��H2O��
�ٸ��ݵȵ�����ԭ����CO ���ӵĽṹʽΪ ��
��H2O ������O ԭ�ӹ�����ӻ�����Ϊ ��
��1mol CO2�к��еĦҼ���ĿΪ ��
��CO2�ĵ���ʽΪ ��
�ݱ�Cu��ԭ������С5��Ԫ��ԭ����Χ�����Ų�ʽΪ ��
��1����һ�����ʵ���Ũ�ȵ�Cu��NO3��2 ��Mn��NO3��2 ��Һ�м���Na2CO3 ��Һ�����ó������������գ����Ƶ�CuMn2O4��
��Mn2+��̬�ĵ����Ų�ʽ�ɱ�ʾΪ
��NO3-�Ŀռ乹����
��2����ͭ��������Ĵ��£�CO ������ΪCO2��HCHO ������ΪCO2 ��H2O��
�ٸ��ݵȵ�����ԭ����CO ���ӵĽṹʽΪ
��H2O ������O ԭ�ӹ�����ӻ�����Ϊ
��1mol CO2�к��еĦҼ���ĿΪ
��CO2�ĵ���ʽΪ
�ݱ�Cu��ԭ������С5��Ԫ��ԭ����Χ�����Ų�ʽΪ
���㣺ԭ�Ӻ�������Ų�,�жϼ��ӻ����ӵĹ���,ԭ�ӹ���ӻ���ʽ���ӻ������ж�
ר�⣺ԭ�������ṹר��,��ѧ���뾧��ṹ
��������1����Mn��ԭ������Ϊ25�������������ԭ����д��Mn�Ļ�̬ԭ�ӵĵ����Ų�ʽ��������ȷ��Mn2+��̬�ĵ����Ų�ʽ��
�����üӳɵ��ӶԻ���ģ���жϣ�
��2���ٸ���N2��COΪ�ȵ����壬��ϵȵ�����ṹ�����жϣ�
�ڸ�������ԭ���γɵĦļ��µ��Ӷ����ж��ӻ����ͣ�
�۾�CO2�ĽṹʽO=C=O��һ��˫����һ��Ϊ�ļ��жϣ�
�ܶ�����̼����C��O��ɵĹ��ۻ�����ṹʽΪO=C=O��
�ݱ�Cu��ԭ������С5��Ԫ����Cr������24�����ӣ�3d�ܼ��ϴﵽ�����ȶ�״̬��
�����üӳɵ��ӶԻ���ģ���жϣ�
��2���ٸ���N2��COΪ�ȵ����壬��ϵȵ�����ṹ�����жϣ�
�ڸ�������ԭ���γɵĦļ��µ��Ӷ����ж��ӻ����ͣ�
�۾�CO2�ĽṹʽO=C=O��һ��˫����һ��Ϊ�ļ��жϣ�
�ܶ�����̼����C��O��ɵĹ��ۻ�����ṹʽΪO=C=O��
�ݱ�Cu��ԭ������С5��Ԫ����Cr������24�����ӣ�3d�ܼ��ϴﵽ�����ȶ�״̬��
���
�⣺��1����Mn��ԭ������Ϊ25����̬ԭ�ӵĵ����Ų�ʽΪ��1s22s22p63s23p63d54s2����Mn2+��̬�ĵ����Ų�ʽ�ɱ�ʾΪ1s22s22p63s23p63d5����[Ar]3d5����
�ʴ�Ϊ��1s22s22p63s23p63d5����[Ar]3d5����
��NO3-��Nԭ���γ�3���ļ���û�йµ��Ӷԣ���ӦΪƽ�������Σ�
�ʴ�Ϊ��ƽ�������Σ�
��2����N2��COΪ�ȵ����壬���߽ṹ���ƣ�N2�ĽṹΪN��N����CO�ĽṹΪC��O��
�ʴ�Ϊ��C��O��
��H2O ������O�γ�2���ļ����µ��Ӷ���Ϊ
=2����Ϊ�ӻ�sp3��
�ʴ�Ϊ��sp3�ӻ���
��CO2�ĽṹʽΪO=C=O��������C�γ�2���ļ�����1molCO2�к��еĦҼ���ĿΪ��6.02��1023������2mol����
�ʴ�Ϊ��2��6.02��1023������2mol����
�ܶ�����̼����C��O��ɵĹ��ۻ�����ṹʽΪO=C=O������ʽΪ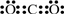 ��
��
�ʴ�Ϊ��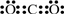 ��
��
�ݱ�Cu��ԭ������С5��Ԫ����Cr������24�����ӣ�3d�ܼ��ϴﵽ�����ȶ�״̬���۵����Ų�ʽΪ3d54s1��
�ʴ�Ϊ��3d54s1��
�ʴ�Ϊ��1s22s22p63s23p63d5����[Ar]3d5����
��NO3-��Nԭ���γ�3���ļ���û�йµ��Ӷԣ���ӦΪƽ�������Σ�
�ʴ�Ϊ��ƽ�������Σ�
��2����N2��COΪ�ȵ����壬���߽ṹ���ƣ�N2�ĽṹΪN��N����CO�ĽṹΪC��O��
�ʴ�Ϊ��C��O��
��H2O ������O�γ�2���ļ����µ��Ӷ���Ϊ
| 6-2��1 |
| 2 |
�ʴ�Ϊ��sp3�ӻ���
��CO2�ĽṹʽΪO=C=O��������C�γ�2���ļ�����1molCO2�к��еĦҼ���ĿΪ��6.02��1023������2mol����
�ʴ�Ϊ��2��6.02��1023������2mol����
�ܶ�����̼����C��O��ɵĹ��ۻ�����ṹʽΪO=C=O������ʽΪ
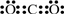 ��
���ʴ�Ϊ��
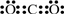 ��
���ݱ�Cu��ԭ������С5��Ԫ����Cr������24�����ӣ�3d�ܼ��ϴﵽ�����ȶ�״̬���۵����Ų�ʽΪ3d54s1��
�ʴ�Ϊ��3d54s1��
�����������ۺϿ������ʵĽṹ������֪ʶ�������ڵ����Ų�ʽ���ȵ����塢�ӻ���������λ����֪ʶ����Ŀ�Ѷ��еȣ�ע������ӻ����͵��жϷ�����

��ϰ��ϵ�д�
�����Ŀ
��NA��ʾ�����ӵ�������ֵ������������ȷ���ǣ�������
| A��1 mol�ǻ���-OH���к��е�������Ϊ10 NA |
| B����ͬ״���£�22.4 L NO��11.2 L O2��Ӧ�������ﺬ��NA������ |
| C����H2O2+Cl2=2HCl+O2��Ӧ�У�ÿ����32 g��������ת��2 NA������ |
| D��1 mol Na2O2����2 NA�������� |
����˵����ȷ���ǣ�������
| A��100mL1mol/L Al2��SO4��3��Һ�У�Al3+��Ϊ0.2��6.02��1023 |
| B��0.1molп��100mL1mol/L ��ϡ�����ַ�Ӧ�����������ķ�����Ϊ0.1��6.02��1023 |
| C��C60��C70�Ļ���ﹲ12g����������̼ԭ����Ϊ6.02��1023 |
| D��1mol����-CH3����1mol��������OH-��������������Ϊ10��6.02��1023 |
�����������ʵ��ó��Ľ�����ȷ���ǣ�������
| A����ij��Һ�м���ϡ���ᣬ����������ͨ�����ʯ��ˮ��ʯ��ˮ����ǣ�����Һһ������CO32- |
| B���ò�˿պȡ����ij��Һ������ɫ��Ӧ������ʻ�ɫ������Һһ������Na+ |
| C����ij��Һ�еμ�BaCl2��Һ������ɫ�������ٵμ�ϡHNO3�������ܽ⣬��ԭ��Һ��һ������SO42- |
| D����ij��Һ�еμ�KSCN ��Һ����Һ����ɫ���μ���ˮ����Һ�Ժ�ɫ������Һ��һ����Fe2+ |
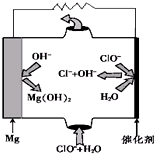 ��������Ҫ���Եز�����ʣ���������ơ�����þ����̬���ڣ�
��������Ҫ���Եز�����ʣ���������ơ�����þ����̬���ڣ�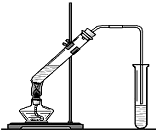 ��1��װ���е���Ҫ���ڱ���̼������Һ��Һ���ϣ����ܲ�����Һ�У�Ŀ���Ƿ�ֹ
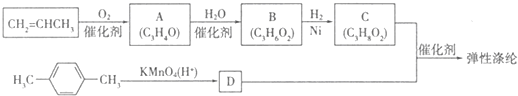
��1��װ���е���Ҫ���ڱ���̼������Һ��Һ���ϣ����ܲ�����Һ�У�Ŀ���Ƿ�ֹ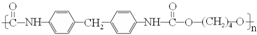 �����ϳ�·����ͼ��ʾ�����ַ�Ӧ�Լ������������ʡ�ԣ���
�����ϳ�·����ͼ��ʾ�����ַ�Ӧ�Լ������������ʡ�ԣ���