��Ŀ����
ʵ������һƿNa2SO3���壬���п��ܺ���NaCl��Na2SO4��KNO3��K2CO3��K2SO4�е�һ�ֻ������ʣ���ͨ������ʵ��ȷ������Ʒ�ijɷּ�Na2SO3����������
�ƴ���Ʒ5.0g������5.00mL6mol?L-1������Һ����������������ɫ����560mL���ѻ���Ϊ��״���������ݳ���������Һ�м��������BaCl2��Һ�������ˡ�����õ���ɫ����9.32g������ɫ�ܲ����۲죬��Һ����ɫ��Ӧ����ɫ��
�����ʵ���¼��д���пհף�
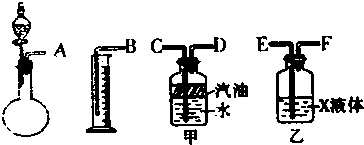
��1��������ͼ��ʾ������װ��������ʵ�飬���������ң����������ܿ����ӵ�˳����
������ĸ��ţ� �� �� �� �� �� ��
��2������Һ��X�� ���������͵������� ��
��3��ʵ���У�ȷ������ɵ�����������йز�����ע�������� ��
��4������ʵ���¼��������Ʒ��Na2SO3���������� ����Ʒ��һ�����е������� ��

�ƴ���Ʒ5.0g������5.00mL6mol?L-1������Һ����������������ɫ����560mL���ѻ���Ϊ��״���������ݳ���������Һ�м��������BaCl2��Һ�������ˡ�����õ���ɫ����9.32g������ɫ�ܲ����۲죬��Һ����ɫ��Ӧ����ɫ��
�����ʵ���¼��д���пհף�
��1��������ͼ��ʾ������װ��������ʵ�飬���������ң����������ܿ����ӵ�˳����
������ĸ��ţ�
��2������Һ��X��
��3��ʵ���У�ȷ������ɵ�����������йز�����ע��������
��4������ʵ���¼��������Ʒ��Na2SO3����������
���㣺̽�����ʵ���ɻ�������ʵĺ���
ר�⣺
��������������Ʒ6.30g������6.0mol?L-1��������������������ɫ����560mL����״����������ɫ�겣���۲죬��Һ����ɫ��Ӧ����ɫ��˵�������ӣ�֤���������һ����KNO3��K2CO2��K2SO4��������ɫ����560mL����״�����Ƕ����������ʵ���Ϊ0.025mol�����ݳ���������Һ�м����Թ�����BaCl2��Һ���õ���ɫ����9.32g���ж�Ϊ���ᱵ�������ʵ���=
=0.04mol��
��1����ʵ�������Ϊ�����巢��-�������-��Һ�����������ݷ���װ�á�����װ�á���ˮװ�ú��ռ�װ������
��2����ͼ��ϴ��ƿ����ȥ�������������е�ˮ��������ͼ������ˮ�����ⶨ��������������װ�ã����������Ƿ�ֹ���������ˮ�Ӵ���Ӧ������ʱҺ��X��Ũ���ᣬ����SO2������ˮ��Һ��ʱ��������ˮ���������ã�
��3������ʱ����Ҫ��ȷ�ز���ų���������������Ͳ��Һ��Ҫ��ͬ������Ҫȷ����ˮ����װ�ò�����������������õ�ԭ���������ų�Һ�������Ͳ�ж���Һ��������Ϊ��������������������ǣ���ȴ�����£���Ͳ���ƿҺ����ƽ�����������
��4���ݶ�������������������ʵ�����ΪNa2SO3�����ʵ�������������������������������ɫ��Ӧ֪��KNO3��K2CO3��K2SO4����ɫ����9.32 g�����ʵ���������������ʵ�����֪��Na2SO4������������������������Na2SO3������֪����NaCl��
| 9.32g |
| 233g/mol |
��1����ʵ�������Ϊ�����巢��-�������-��Һ�����������ݷ���װ�á�����װ�á���ˮװ�ú��ռ�װ������
��2����ͼ��ϴ��ƿ����ȥ�������������е�ˮ��������ͼ������ˮ�����ⶨ��������������װ�ã����������Ƿ�ֹ���������ˮ�Ӵ���Ӧ������ʱҺ��X��Ũ���ᣬ����SO2������ˮ��Һ��ʱ��������ˮ���������ã�
��3������ʱ����Ҫ��ȷ�ز���ų���������������Ͳ��Һ��Ҫ��ͬ������Ҫȷ����ˮ����װ�ò�����������������õ�ԭ���������ų�Һ�������Ͳ�ж���Һ��������Ϊ��������������������ǣ���ȴ�����£���Ͳ���ƿҺ����ƽ�����������
��4���ݶ�������������������ʵ�����ΪNa2SO3�����ʵ�������������������������������ɫ��Ӧ֪��KNO3��K2CO3��K2SO4����ɫ����9.32 g�����ʵ���������������ʵ�����֪��Na2SO4������������������������Na2SO3������֪����NaCl��
���
�⣺��1����ʵ�������Ϊ�����巢��-�������-��Һ�����������ݷ���װ�á�����װ�á���ˮװ�ú��ռ�װ������װ������˳��ӿ�Ϊ��A E F D C B��
�ʴ�Ϊ��A��E��F��D��C��B��
��2����ͼ��ϴ��ƿ����ȥ�������������е�ˮ��������ͼ������ˮ�����ⶨ��������������װ�ã����������Ƿ�ֹ���������ˮ�Ӵ���Ӧ��X��Һ��ŨH2SO4�������Ƿ�ֹSO2��ˮ�Ӵ���
�ʴ�Ϊ��ŨH2SO4����ֹSO2��ˮ�Ӵ���
��3����ˮ����װ�ò�����������������õ�ԭ���������ų�Һ�������Ͳ�ж���Һ��������Ϊ��������������������ǣ���ȴ�����£���Ͳ���ƿҺ����ƽ�����������
�ʴ�Ϊ����Ӧװ���������º��������ƶ���Ͳ��ʹ��Ͳ���ƿ��Һ�汣��ˮƽ��Ȼ���ȡ�����
��4������6.0mol?L-1����������������ʵ���м���������Һ�����Ϊ5.00mL��������ɫ����560mL����״���������������ʵ���Ϊ0.025mol�����������Ƶ����ʵ���Ϊ0.025mol����������=
��100%=63%���õ���ɫ����9.32g���ж�Ϊ���ᱵ�������ʵ���=
=0.04mol��������������ʵ���Ϊ0.03mol��˵��һ�����������ƣ�����Ϊ0.01mol��142g/mol=1.42g���������Ƶ����ʵ���Ϊ0.025mol������=0.025mol��126g/mol=3.15g����������Ϊ5.0g-1.42g-3.15g=0.43g��
����ɫ��Ӧ֪��KNO3��K2CO3��K2SO4������һ������NaCl��
�ʴ�Ϊ��63%��NaCl��Na2SO4��
�ʴ�Ϊ��A��E��F��D��C��B��
��2����ͼ��ϴ��ƿ����ȥ�������������е�ˮ��������ͼ������ˮ�����ⶨ��������������װ�ã����������Ƿ�ֹ���������ˮ�Ӵ���Ӧ��X��Һ��ŨH2SO4�������Ƿ�ֹSO2��ˮ�Ӵ���
�ʴ�Ϊ��ŨH2SO4����ֹSO2��ˮ�Ӵ���
��3����ˮ����װ�ò�����������������õ�ԭ���������ų�Һ�������Ͳ�ж���Һ��������Ϊ��������������������ǣ���ȴ�����£���Ͳ���ƿҺ����ƽ�����������
�ʴ�Ϊ����Ӧװ���������º��������ƶ���Ͳ��ʹ��Ͳ���ƿ��Һ�汣��ˮƽ��Ȼ���ȡ�����
��4������6.0mol?L-1����������������ʵ���м���������Һ�����Ϊ5.00mL��������ɫ����560mL����״���������������ʵ���Ϊ0.025mol�����������Ƶ����ʵ���Ϊ0.025mol����������=
| 0.025mol��126g/mol |
| 5.0g |
| 9.32g |
| 233g/mol |
����ɫ��Ӧ֪��KNO3��K2CO3��K2SO4������һ������NaCl��
�ʴ�Ϊ��63%��NaCl��Na2SO4��
���������⿼�����������ʵ�̽����ʵ������жϣ�ʵ����Ƶ����ݺͲ��裬�������ʵ����ʺ�ʵ����������ǽ���ؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ��ѧ�̸̳����¿α�ϵ�д�
��ѧ�̸̳����¿α�ϵ�д�
�����Ŀ
��ڶ�����ϡ������Ԫ�ص�ԭ�Ӿ�����ͬ�����������ǣ�������
| A��S2-? |
| B��Cl- |
| C��Al3+? |
| D��K+ |
���л�ѧʵ�����ע�ⰲȫ������˵������ȷ���ǣ�������
| A���������ὦ�����У�Ӧ������ˮ��ϴ����ϴ��գ�۾� |
| B��������Ũ����Һմ��Ƥ���ϣ�Ҫ�����ô���ˮ��ϴ��Ȼ��Ϳ��������Һ |
| C����ú����ȡ�ý����Ʊ��������ӣ��������ִ��� |
| D������������Һʱ����������Ͳ�м���һ�������ˮ�����ڽ�������������Ũ���� |

 ij��A���л���ѧ��ҵ�Ļ���ԭ�ϣ������������������һ�����ҵ�ʯ�ͻ�����չˮƽ��A����һ��ֲ���������ڼ���A�ɷ�����ͼ��ʾ��һϵ�л�ѧ��Ӧ����ͼ�ش��������⣺
ij��A���л���ѧ��ҵ�Ļ���ԭ�ϣ������������������һ�����ҵ�ʯ�ͻ�����չˮƽ��A����һ��ֲ���������ڼ���A�ɷ�����ͼ��ʾ��һϵ�л�ѧ��Ӧ����ͼ�ش��������⣺
 ���칹�����Ŀ��
���칹�����Ŀ��
 �����ҹ�����ϲ������Ʒ�����к��ж������������彡���ijɷ֣��ݲⶨ��Ҷ�к���450�����ϵ��л��ɷ���15�����ϵ�Ԫ�أ�ij��ѧ�о�С����̽����Ҷ�и�Ԫ�صĺ����������̽��ʵ�鷽�����£�����֪��Ҷ�е�������Ԫ�ضԸ����ӵIJⶨ��Ӱ�죩
�����ҹ�����ϲ������Ʒ�����к��ж������������彡���ijɷ֣��ݲⶨ��Ҷ�к���450�����ϵ��л��ɷ���15�����ϵ�Ԫ�أ�ij��ѧ�о�С����̽����Ҷ�и�Ԫ�صĺ����������̽��ʵ�鷽�����£�����֪��Ҷ�е�������Ԫ�ضԸ����ӵIJⶨ��Ӱ�죩