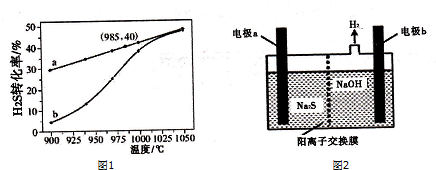
��Ŀ����
7��ij�����ܽ��õ�����Һ�У����ܺ���Fe2+��Fe3+��SO42-��NH4+��Ba2+��CO32-�����е�ij���֣���1����ͬѧ������ʵ�飺��ȡ������Һ���Թ��У���μ���Ũ����������Һ�����������ɰ�ɫ������Ѹ�ٱ�Ϊ����ɫ������Ϊ���ɫ�����д̼����������ɣ�����ȡ������Һ���Թ��У��������������ữ���Ȼ�����Һ���а�ɫ�������ɣ�����Һ��һ�����е�������Fe2+��SO42-��NH4+��д�����а�ɫ����ת��Ϊ���ɫ�����Ļ�ѧ����ʽ
��2����ͬѧ������ʵ�飺ȡ������Һ���Թ��У��μӼ������������Һ�������������ٵμ�H2O2��������Һ���ɫ�������μ�H2O2����ɫ����ȥ�������ݲ�����ΪŪ������Ե
�ɣ�����������֪��H2O2+SCN-��SO42-+CO2��+N2��+H2O+H+��SCN-��SΪ-2�ۣ�
�ٸ÷�Ӧ�У���������Ԫ��ΪN��S��ÿ����1molCO2ת�Ƶĵ�����Ϊ11����
�ڸ�����ͬѧ��ʵ���������жϻ�ԭ��ǿ��Ϊ��Fe2+��NCN-�����=����
�۸������ϣ���ͬѧ����IJ����ǣ�H2O2��SCN-����ʹ��ɫ����ȥ���������һ��ʵ�飬��֤��ͬѧ�IJ����Ƿ���ȷȡ������ɫ�����Һ���Թ��У��μ�KSCN��Һ������Һ�ָ���ɫ��֤����ͬѧ�ƶ���ȷ��
���� ��1����ȡ������Һ���Թ��У���μ���Ũ����������Һ�����ֿ�ʼ���ɰ�ɫ��������Fe3+����ɫ����Ѹ�ٱ�Ϊ����ɫ������Ϊ���ɫ��˵������Fe2+����һ������CO32-��ͬʱ���д̼�������ų���ӦΪ������˵������Fe2+��
����ȡ������Һ���Թ��У��������������ữ���ٵμ��Ȼ�����Һ���а�ɫ�������ɣ�˵������SO42-����Ba2+��
��2����ͬѧ��������ʵ�飺ȡ������Һ���Թ��У��μӼ������������Һ�������������ٵμ�H2O2��������Һ���ɫ��˵������Fe2+������Fe3+�������μ�H2O2����ɫ����ȥ�������ݲ���������H2O2+SCN-��SO42-+CO2��+N2��+H2O+H+����Ӧ��N��S���ữ�ϼ����ߣ�����������ϻ��ϼ۵ı仯�Լ����ʵ����ʽ����⣮
��� �⣺��1��ȡ������Һ���Թ��У���μ���Ũ����������Һ�����ֿ�ʼ���ɰ�ɫ��������Fe3+����ɫ����Ѹ�ٱ�Ϊ����ɫ������Ϊ���ɫ��˵������Fe2+��������ӦΪ4Fe��OH��2+O2+2H2O=4Fe��OH��3����һ������CO32-��ͬʱ���д̼�������ų���ӦΪ������˵������Fe2+��
����ȡ������Һ���Թ��У��������������ữ���ٵμ��Ȼ�����Һ���а�ɫ�������ɣ�˵������SO42-����Ba2+������һ�����е�������Fe2+��SO42-��NH4+��
�ʴ�Ϊ��Fe2+��SO42-��NH4+�� 4Fe��OH��2+O2+2H2O=4Fe��OH��3��
��2������ͬѧ��������ʵ�飺ȡ������Һ���Թ��У��μӼ������������Һ�������������ٵμ�H2O2��������Һ���ɫ��˵������Fe2+������Fe3+�������μ�H2O2����ɫ����ȥ�������ݲ���������H2O2+SCN-��SO42-+CO2��+N2��+H2O+H+����Ӧ��N��S���ữ�ϼ����ߣ�SԪ�ػ��ϼ���-2�����ߵ�+6�ۣ�NԪ�ػ��ϼ���-3�����ߵ�0�ۣ���������ÿ����lmol CO2���з���ʽ��֪����1molSO42-��0.5mol+N2��ת�Ƶĵ��ӵ����ʵ���Ϊ1mol��8+0.5mol��2��3=11mol��Ϊ11NA����
�ʴ�Ϊ��N��S��11��
���ɷ�Ӧ���Ⱥ�˳���֪��������������Fe2+����Fe2+��ԭ�Խ�ǿ��
�ʴ�Ϊ������
������ͬѧ�²���ȷ������ɫ�����Һ���еμ�KSCN��Һ����Һ��ָ���ɫ��������֤����Ϊ��ȡ������ɫ�����Һ���Թ��У��μ�KSCN��Һ������Һ�ָ���ɫ��֤����ͬѧ�ƶ���ȷ��
�ʴ�Ϊ��ȡ������ɫ�����Һ���Թ��У��μ�KSCN��Һ������Һ�ָ���ɫ��֤����ͬѧ�ƶ���ȷ��
���� ���⿼���˳������ӵ����ʼ����鷽����Ϊ��Ƶ���㣬��Ŀ�Ѷ��еȣ�ע���������ӷ�Ӧ������������ȷ�������ӵļ��鷽��������������ѧ���ķ���������������������������

| A�� | CO2�ĵ���ʽ | B�� | Cl-�Ľṹʾ��ͼ | ||
| C�� | MgCl2�ĵ���ʽ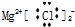 | D�� | ������Ϊ37����ԭ��${\;}_{37}^{17}$Cl |
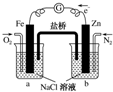
| A�� | �ձ�a�е���ҺpH���� | B�� | �ձ�b�з����ķ�ӦΪ2H++2e-�TH2�� | ||
| C�� | �ձ�a�з����ķ�ӦΪFe-2e-�TFe2+ | D�� | �ձ�b�з���������Ӧ |
| A�� | c��Z��=0.45mol/L | B�� | c��X2��=0.3mol/L c��Z��=0.1 mol/L | ||
| C�� | c��X2��=0.5mol/L | D�� | c��Y2��=0.9mol/L |
| A�� | �Ӵ����Ʊ�������Ҫ���豸�з���¯���ϳ����������� | |
| B�� | ��ҵ�Ʊ������У�β����������ʹ��NaOH��Һ���յ� | |
| C�� | ��ҵ�Ʊ������У�����������98.3%��Ũ�������գ�Ŀ���Ƿ�ֹ�γ��������Ա�ʹ��������������ȫ | |
| D�� | ���Ṥҵ�У��ڽӴ��Ұ�װ�Ƚ�������Ϊ������SO3ת��ΪH2SO4ʱ�ų����� |
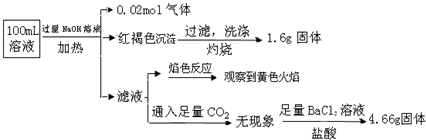
�ɴ˿�֪ԭ��Һ�У�������
| A�� | ԭ��Һ��c��Fe3+��=0.2 mol•L-1 | |
| B�� | SO42����NH4+��Na+һ�����ڣ�CO32����Al3+һ�������� | |
| C�� | ��Һ��������4�����Ӵ��ڣ�����Cl��һ�����ڣ���c��Cl������0.2 mol•L-1 | |
| D�� | ȡ����ԭ��Һ���Թ��У�����KSCN��Һ����Һ��Ѫ��ɫ��˵������Һһ��û��Fe2+��ֻ��Fe3+ |
| A�� | úȼ��ʱ����ѧ����Ҫת��Ϊ���� | |
| B�� | �׳�ƹ���ʱ������ת��Ϊ���� | |
| C�� | ���ˮ��������������ʱ����ѧ��ת��Ϊ���� | |
| D�� | ��ɫֲ����й������ʱ��̫����ת��Ϊ��ѧ�� |