��Ŀ����
6��������һ�ּ��߷�չDZ���������Դ����̫����Ϊ��Դ���Ȼ�ѧ���ѭ���ֽ�ˮ��һ�ָ�Ч������Ⱦ�����ⷽ�����䷴Ӧ������ͼ1��ʾ��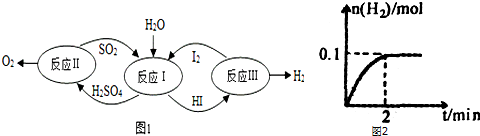
��1��д����ӦI��II�Ļ�ѧ����ʽ������д��Ӧ��������
I��SO2+2H2O+I2=H2SO4+2HI��II2H2SO4=2SO2+O2+2H2O��III��2HI?H2+I2
��2���������������˴����õ�������SO2��I2��д��ѧʽ����
��3��һ���¶��£���2L�ܱ������м���1molHI��g����������ӦIII�����ɵ�I2Ϊ���壬H2���ʵ�����ʱ��ı仯��ͼ2��ʾ����0-2min�ڵ�ƽ����Ӧ����v��HI��=0.05mol/��L•min������
����Ϊ�÷�Ӧ�ﵽƽ��״̬�ж����ݵ��Ǣܢ�
��n��H2��=n��I2��
��ѹǿ���ٱ仯
�������ܶȲ��ٱ仯
��2��H-I���ѵ�ͬʱ����1��H-H����
�ݸ����Ũ�ȱ��ֲ���
��c��HI����c��H2����c��I2��=2��1��1
��4����֪�йؼ������ݣ�I2-151kJ/mol��H2-436kJ/mol��HI-298kJ/mol������0.2molHI�ֽ�ʱ�������仯�����գ�����ա��ų�����0.9kJ��������
���� ��1����ͼ��֪����ӦIΪ����������ⷢ��������ԭ��Ӧ���������HI����ӦIIΪ����ֽ����ɶ�������������ˮ���ݴ���д����ʽ��
��2���������ص����ڻ�ѧ��Ӧǰ���������䣬����SO2��I2�ڷ�Ӧǰ�������������
��3����ͼ��֪��2min��H2���ʵ����ı仯����Ȼ����㻯ѧ��Ӧ���ʣ������û�ѧ��Ӧ����֮�ȵ��ڻ�ѧ������֮������𣬸��ݻ�ѧƽ��״̬������
��4�����ݷ�Ӧ�ȼ��㣬�÷�Ӧ����ӦΪ���ȷ�Ӧ����ѧ������ʱ�����յ�����-��ѧ������ʱ��ų�������=��Ӧ���յ�������
��� �⣺��1����ͼ��֪����ӦIΪ����������ⷢ��������ԭ��Ӧ���������HI���÷�ӦΪSO2+2H2O+I2=H2SO4+2HI����ӦIIΪ����ֽ⣬�÷�ӦΪ2H2SO4=2SO2+O2+2H2O��
�ʴ�Ϊ��SO2+2H2O+I2=H2SO4+2HI��2H2SO4=2SO2+O2+2H2O��
��2����SO2+2H2O+I2�TH2SO4+2HI��.2H2SO4�T2SO2+O2+2H2O��.2HI?H2+I2
SO2��I2�ڷ�Ӧǰ�����������ʲ��䣬���˴����ã�
�ʴ�Ϊ��SO2��I2��
��3����ͼ��֪2min�����������ʵ���������0.1mol����������Ũ��Ϊ0.05mol/L����������ʾ�Ļ�ѧ��Ӧ����Ϊ$\frac{0.05mol/L}{2min}$=0.025mol/��L��min�����ɷ�Ӧ�еĻ�ѧ��������֪v��HI��Ϊ0.025mol/��L��min����2=0.05mol/��L��min����
��n��H2��=n��I2����û����ȷ��Ӧ�������жϷ�Ӧ�Ƿ�ƽ�⣻
��ѹǿ��ʼ���ղ��ٱ仯�������ж�ƽ�⣻
��������ʼ�����ܶȲ��ٱ仯�������ж�ƽ�⣻
��2��H-I���ѵ�ͬʱ����1��H-H���ѣ����淴Ӧ������ȣ����жϷ�Ӧ�Ƿ�ƽ�⣻
�ݸ����Ũ�ȱ��ֲ������ж�ƽ��ı�־��
��c��HI����c��H2����c��I2��=2��1��1��û����ȷ��Ӧ�������жϷ�Ӧ�Ƿ�ƽ�⣻
�ʴ�Ϊ��0.05mol/L��min���ܢݣ�
��4��������Ŀ�е����ݣ���֪HI�ֽ�ķ�Ӧ�ȣ���H=+298kJ/mol��2-151kJ/mol-436kJ/mol=+9kJ/mol����ֽ�0.2molHI��Ҫ���գ�0.1��9kJ/mol=0.9kJ��
�ʴ�Ϊ�����գ�0.9��
���� ���⿼���Ϊ�ۺϣ����ؿ��黯ѧ��Ӧ���ʼ�ƽ��״̬���ص㡢��Ӧ�ȵļ��㣬ע���������������ѧƽ��״̬���жϷ�������Ŀ�Ѷ��еȣ�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
����������Ϣ�ش��������⣺
��1������������X�Ļ�ѧʽΪFe��
��2��д������CuCl�Ļ�ѧ����ʽ��CuCl2+CuSO4+SO2+2H2O=2CuCl��+2H2SO4��
��3����CuCl�����ɹ����������ϲ���Ҫ����SO2���壬��ϻ�ѧ����ʽ�ͱ�Ҫ������˵������Cu+2 H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CuSO4+SO2��+2H2O��Ӧ�����ɵ�CuSO4�� SO2Ϊ1��1�����������ϲ���Ҫ����SO2���壮ʵ��������SO2Ҫ�ʵ�������ԭ����������Cu2+�Ļ�ԭ���ʣ�ͬʱ���Է�ֹ���ɵ�Cu+��������������һ�㼴�ɣ���
��4��ʵ��̽��pH��CuCl���ʵ�Ӱ�����±���ʾ��
| pH | 1 | 2 | 3 | 4 | 5 | 6 | 7 |
| CuCl����/% | 70 | 90 | 82 | 78 | 75 | 72 | 70 |
��5���Ȼ���ͭ�Ķ���������
�ٳ�ȡ��Ʒ0.25g������FeCl3��Һ����ƿ�У�����ܽ⣮
����0.10mol•L-1���������Һ�ζ�����֪��CuCl+FeCl3�TCuCl2+FeCl2��Fe2++Ce4+�TFe3++Ce3+������ƽ��ʵ�������£�ƽ��ʵ�������ܳ���1%����
| ƽ��ʵ����� | 1 | 2 | 3 |
| 0.25g��Ʒ�������������Һ�������mL�� | 24.35 | 24.05 | 23.95 |
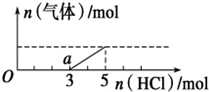 ��һ������CO2����ͨ��2Lδ֪Ũ�ȵ�NaOH��Һ�У���������Һ����μ���ϡ������������������Һ���ȣ�������������HCl�����ʵ����Ĺ�ϵ��ͼ��ʾ������������ܽ��HCl�Ļӷ���������˵������ȷ���ǣ�������
��һ������CO2����ͨ��2Lδ֪Ũ�ȵ�NaOH��Һ�У���������Һ����μ���ϡ������������������Һ���ȣ�������������HCl�����ʵ����Ĺ�ϵ��ͼ��ʾ������������ܽ��HCl�Ļӷ���������˵������ȷ���ǣ�������| A�� | O����Һ���������ʵĻ�ѧʽΪNaOH��Na2CO3 | |
| B�� | a����Һ��c ��HCO3-����c ��CO32-�� | |
| C�� | ��״���£�ͨ��CO2�����Ϊ44.8L | |
| D�� | ԭNaOH��Һ�����ʵ���Ũ��Ϊ5mol/L |
| A�� | 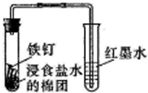 �۲�����������ʴ | B�� | 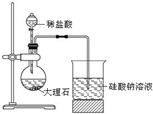 �Ƚ��ȡ�̼����ķǽ����� | ||
| C�� |  ʵ��������ʯ�� | D�� |  ������������ȡ����Ӧ |
| A�� | ϡ����������������Һ��Ӧ��H++OH-=H2O | |
| B�� | ����ϡ���ᷴӦ��Al+2H+=Al3++H2�� | |
| C�� | ���Ȼ�����Һ������������Һ��Ӧ��FeCl3+3OH-=Fe��OH��3��+3C1- | |
| D�� | ������̼�����ʯ��ˮ��Ӧ��CO2+2OH-=CO32-+H2O |
| A�� | CH3Cl | B�� | ��CH3��3CCH2Cl | C�� | 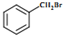 | D�� |  |
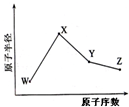 W��X��Y��Z�����ֳ����Ķ�����Ԫ�أ���ԭ�Ӱ뾶��ԭ�������仯��ͼ����֪W��һ�ֺ��ص�������Ϊ18��������Ϊ10��Xԭ�Ӻ�Neԭ�ӵĺ�����������1��Y�ĵ�����һ�ֳ����İ뵼����ϣ�Z�ķǽ�������ͬ����Ԫ������ǿ������˵��������ǣ�������
W��X��Y��Z�����ֳ����Ķ�����Ԫ�أ���ԭ�Ӱ뾶��ԭ�������仯��ͼ����֪W��һ�ֺ��ص�������Ϊ18��������Ϊ10��Xԭ�Ӻ�Neԭ�ӵĺ�����������1��Y�ĵ�����һ�ֳ����İ뵼����ϣ�Z�ķǽ�������ͬ����Ԫ������ǿ������˵��������ǣ�������| A�� | ��Ӧ�����Ӱ뾶X��W | |
| B�� | ��Ӧ��̬�⻯����ȶ���Y��Z | |
| C�� | ������XZW�Ⱥ����Ӽ��ֺ����ۼ� | |
| D�� | Z��X������������Ӧ��ˮ���������Y�������ﷴӦ |
ʵ��һ����ͬѧ�о���ʵ�鱨�������
| ʵ �� �� �� | �� �� | �� �� |
| �ٷֱ�ȡ�������2mol/L�������Թ��У� �ڷֱ�Ͷ���С����״��ͬ��Cu��Fe��Mg�� | ��Ӧ������ Mg��Fe��Cu | ��Ӧ�������Խ���ã���Ӧ����Խ�죮 |
ʵ�������֪2KMnO4+5H2C2O4+3H2SO4�TK2SO4+2MnSO4+8H2O+10CO2�����ڸ������������Һ�Ͳ�����Һ��Ӧʱ�����ֿ�ʼһ��ʱ�䷴Ӧ���ʽ�������Һ��ɫ�����ԣ�������ͻȻ��ɫ����Ӧ�������Լӿ죮
��1���������ʵ������ijͬѧ��ΪKMnO4��H2C2O4��Ӧ�Ƿ��ȷ�Ӧ��������Һ�¶����ߣ���Ӧ���ʼӿ죮��Ӱ�컯ѧ��Ӧ���ʵ����ؿ�������뷴Ӧ�������Լӿ��ԭ���������ɵ��������д����ã�
��2������ʵ��֤����IJ��룬�����Ը��������Һ��������Һ�⣬����Ҫѡ����Լ����������B��ѡ���ţ���
A������� B�������� C��ˮ D���Ȼ��̣�