��Ŀ����
4��һ���¶��£���2L���ܱ������У�X��Y��Z�������������ʱ��仯��������ͼ1��ʾ��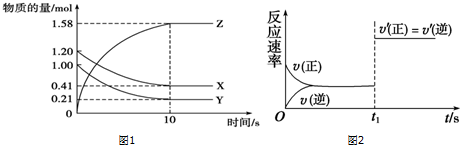
��1���ӷ�Ӧ��ʼ��10s����Z��ʾ�ķ�Ӧ����Ϊ0.079mol/��L•s����
��2��Y��ת����Ϊ79%��
��3���÷�Ӧ�Ļ�ѧ����ʽΪX��g��+Y��g��?2Z��g����
��4�����¶��� ƽ�ⳣ��K�ı���ʽΪ$\frac{{c}^{2}��Z��}{c��X��•c��Y��}$
��5��10s���ijһʱ�̣�t1���ı��������������������ʱ��ı仯ͼ����ͼ2��ʾ��������˵�����ϸ�ͼ�����C��D��
A��t1ʱ�̣�������X��Ũ��
B��t1ʱ�̣���������ϵ�¶�
C��t1ʱ�̣���С���������
D��t1ʱ�̣�ʹ���˴�����
���� ��1������v=$\frac{��c}{��t}$���㷴Ӧ���ʣ�
��2��Y��ת����Ϊ$\frac{�仯��}{��ʼ��}��100%$��
��3���������ʵ����ı仯�뻯ѧ��������������д��ѧ����ʽ��
��4��ƽ�ⳣ��K����������Ũ����֮�����Ϸ�Ӧ��Ũ����֮����
��5����ͼ���֪���淴Ӧ��������ƽ��û�ƶ���
��� �⣺��1���ͣ�Z��=$\frac{\frac{1.58mol}{2L}}{10s}$=0.079mol/��L•s�����ʴ�Ϊ��0.079mol/��L•s����
��2��Y��ת����Ϊ$\frac{�仯��}{��ʼ��}��100%$=$\frac{1-0.21}{1}$��100%=79%���ʴ�Ϊ��79%��
��3����ͼ����Կ�������Ӧ��10sʱ��X�����ʵ�����С���仯ֵΪ1.20mol-0.41mol=0.79mol��
Y�����ʵ�����С���仯ֵΪ1.0mol-0.21mol=0.79mol����X��YΪ��Ӧ�
Z�����ʵ������࣬�仯ֵΪ1.58mol��Ϊ�����
�������ʵ����ı仯�뻯ѧ�����������ȣ�
����n��X����n��Y����n��Z��=0.79mol��0.79mol��1.58mol=1��1��2��
���Է�Ӧ�ķ���ʽΪX��g��+Y��g��?2Z��g�����ʴ�Ϊ��X��g��+Y��g��?2Z��g����
��4����Ӧ�ķ���ʽΪX��g��+Y��g��?2Z��g����ƽ�ⳣ��K����������Ũ����֮�����Ϸ�Ӧ��Ũ����֮��������K=$\frac{{c}^{2}��Z��}{c��X��•c��Y��}$���ʴ�Ϊ��$\frac{{c}^{2}��Z��}{c��X��•c��Y��}$��
��5����ͼ���֪���淴Ӧ��������ƽ��û�ƶ���
A��t1ʱ�̣�������X��Ũ�ȣ�ƽ�����ƣ����������⣬��A����
B��t1ʱ�̣���������ϵ�¶ȣ�ƽ���ƶ������������⣬��B����
C��t1ʱ�̣���С����������������������ڷ�Ӧǰ���������ȣ�ƽ�ⲻ�ƶ����������⣬��C��ȷ��
D��t1ʱ�̣�ʹ���˴�������������ƽ�ⲻ�ƶ����������⣬��D��ȷ��
�ʴ�Ϊ��C��D��
���� ���⿼�鷴Ӧ���ʵļ���ƽ��ͼ�����⣬��Ŀ�ѶȲ���ע��������ʵ����ı仯�뻯ѧ�����������ȣ��Դ���д��ѧ����ʽ��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| A�� | Fe2��SO4��3������ˮ����������ˮ�� | |
| B�� | Al2O3�۵�ߣ����������²��� | |
| C�� | ����ͭ������ǿ������FeCl3��ʴCu����ӡˢ��·�� | |
| D�� | NaHCO3����ˮ�ʼ��ԣ���������ҩ������ʱ��Щ�������ҩЧ |
| A�� | v��A��=0.4mol/��L•s�� | B�� | v��B��=0.5mol/��L•s�� | C�� | v��C��=0.6mol/��L•s�� | D�� | v��D��=0.7mol/��L•s�� |
| A�� | �����£��������Һ�м���������ˮ����Һ��c��H+�������� | |
| B�� | �����£�2NO��g��+2CO��g��?N2��g��+2CO2��g�����Է����У���÷�Ӧ�ġ�H��0 | |
| C�� | ����ͭ������Һ�����¶�ʱ�о����������������о������� | |
| D�� | ��CH3COOH��Һ������ʵ�飬���ݺܰ���һ����֤��CH3COOH��������� |
| A�� | ��Ũ��NaHSO4��Ba��OH��2����Һ��������H++SO42-+Ba2++OH-�TBaSO4��+H2O | |
| B�� | �ں���Mn2+����Һ�м���ϡ�����ữ���ټ���PbO2��Ӧ��ϵ���Ϻ�ɫ5PbO2+2Mn2++4H+�T5Pb2++2MnO4-+2H2O | |
| C�� | ����������Һ�м������������Һ��2Fe2++H2O2+2H+�T2Fe3++2H2O | |
| D�� | Na2O2Ͷ��H218O�У�2H218O+2Na2O2�T4Na++4OH-+18O2�� |
| A�� | ��X��Yԭ���������l��YΪIIA�壬��Xһ����IIIA�� | |
| B�� | ��Y��OH��m������ˮ����X��OH��nһ��������ˮ | |
| C�� | ��HnXOmΪǿ�ᣬ��X���⻯����Һ��ˮһ�������� | |
| D�� | ��YԪ���γɵĵ��������壬��XԪ���γɵĵ���һ��Ҳ������ |
| A�� | 14 g��ϩ�ͱ�ϩ��������е���ԭ����Ϊ2NA | |
| B�� | 1 molN2��4 mol H2��Ӧ���ɵ�NH3������Ϊ2NA | |
| C�� | 1 molFe���ڹ������ᣬ����ת����Ϊ2NA | |
| D�� | ��״���£�2.24 LCCl4���еĹ��ۼ���Ϊ0.4NA |