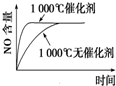
��Ŀ����
��1��Һ�����е���Ҫ�ɷ�֮һ�Ƕ��飮��25�桢101kPaʱ��10kg������ȫȼ������CO2��H2O��l���ų�����5��105 kJ������ȼ�յ��Ȼ�ѧ����ʽΪ �����ȼ����Ϊ ��
��2��1mol��̬�����Ӻ�1mol��̬�����ӽ������1mol�Ȼ��ƾ����ͷų�������Ϊ�Ȼ��ƾ���ľ����ܣ�
�������Ȼ�ѧ����ʽ�У���ֱ�ӱ�ʾ���Ȼ��ƾ��徧���ܵ��� ��
A��Na+��g��+Cl-��g����NaCl��s������Q
B��Na��s��+
Cl2��g����NaCl��s������Q1
C��Na��s����Na��g������Q2
D��Na��g��-e-��Na+��g������Q3
E.
Cl2��g����Cl��g������Q4
F��Cl��g��+e-��Cl-��g������Q5
��д����Q1���Q����Q2����Q3����Q4����Q5֮��Ĺ�ϵʽ ��
��2��1mol��̬�����Ӻ�1mol��̬�����ӽ������1mol�Ȼ��ƾ����ͷų�������Ϊ�Ȼ��ƾ���ľ����ܣ�
�������Ȼ�ѧ����ʽ�У���ֱ�ӱ�ʾ���Ȼ��ƾ��徧���ܵ���
A��Na+��g��+Cl-��g����NaCl��s������Q
B��Na��s��+
| 1 |
| 2 |
C��Na��s����Na��g������Q2
D��Na��g��-e-��Na+��g������Q3
E.
| 1 |
| 2 |
F��Cl��g��+e-��Cl-��g������Q5
��д����Q1���Q����Q2����Q3����Q4����Q5֮��Ĺ�ϵʽ
���㣺�Ȼ�ѧ����ʽ,�ø�˹���ɽ����йط�Ӧ�ȵļ���
ר�⣺��ѧ��Ӧ�е������仯
��������1�����������������ʵ�������ϻ�ѧ����ʽ��Ӧ�����ʵ������㷴Ӧ�ų��������������Ȼ�ѧ����ʽ��д��������ע���ʾۼ�״̬�ͷ�Ӧ�ʱ�д����
��2������1mol��̬�����Ӻ�1mol��̬�����ӽ������1mol�Ȼ��ƾ����ͷų�������Ϊ�Ȼ��ƾ���ľ����ܷ�����
��2������1mol��̬�����Ӻ�1mol��̬�����ӽ������1mol�Ȼ��ƾ����ͷų�������Ϊ�Ȼ��ƾ���ľ����ܷ�����
���
�⣺��1����10kg������ȫ��Ӧ���ʵ���
=
mol����ȫȼ�ղ����ɶ�����̼��Һ̬ˮʱ���ų�����Ϊ5��105kJ��2mol������ȫ��Ӧ����=
=5800KJ�����ݷ�Ӧ��Ͳ���״̬��ע�ۼ�״̬�Ͷ�Ӧ���µķ�Ӧ�ȣ���д���Ȼ�ѧ����ʽΪ��2C4H10��g��+13O2��g��=8CO2��g��+10H2O��l����H=-5800kJ/mol��ȼ��������ȫȼ��1mol���ʷų�����������Ϊ2900 kJ/mol��
�ʴ�Ϊ��2C4H10��g��+13O2��g��=8CO2��g��+10H2O��l����H=-5800kJ/mol��2900 kJ/mol��
��2����1mol��̬�����Ӻ�1mol��̬�����ӽ������1mol�Ȼ��ƾ����ͷų�������Ϊ�Ȼ��ƾ���ľ����ܣ����ʾ���Ȼ��ƾ��徧���ܵ��Ȼ�ѧ����ʽΪ��Na-��g��+Cl-��g���TNaCl��s������Q������A��ȷ��
�ʴ�Ϊ��A��
��A��Na-��g��+Cl-��g���TNaCl��s������Q
B��Na-��s��+
Cl2��g���TNaCl��s������Q1
C��Na��s��=Na��g������Q2
D��Na��g��-e-=Na-��g������Q3
E.
Cl2��g��=Cl��g������Q4
F��Cl��g��+e-=Cl��g������Q5
���ݸ�˹����A+C+D+E+F�õ���Na��s��+
Cl2��g����NaCl��s����Q1=��Q+��Q2+��Q3+��Q4+��Q5��
�ʴ�Ϊ����Q1=��Q+��Q2+��Q3+��Q4+��Q5��
| 10000g |
| 58g/mol |
| 10000 |
| 58 |
| ||
| 58 |
�ʴ�Ϊ��2C4H10��g��+13O2��g��=8CO2��g��+10H2O��l����H=-5800kJ/mol��2900 kJ/mol��
��2����1mol��̬�����Ӻ�1mol��̬�����ӽ������1mol�Ȼ��ƾ����ͷų�������Ϊ�Ȼ��ƾ���ľ����ܣ����ʾ���Ȼ��ƾ��徧���ܵ��Ȼ�ѧ����ʽΪ��Na-��g��+Cl-��g���TNaCl��s������Q������A��ȷ��
�ʴ�Ϊ��A��
��A��Na-��g��+Cl-��g���TNaCl��s������Q
B��Na-��s��+
| 1 |
| 2 |
C��Na��s��=Na��g������Q2
D��Na��g��-e-=Na-��g������Q3
E.
| 1 |
| 2 |
F��Cl��g��+e-=Cl��g������Q5
���ݸ�˹����A+C+D+E+F�õ���Na��s��+
| 1 |
| 2 |
�ʴ�Ϊ����Q1=��Q+��Q2+��Q3+��Q4+��Q5��
���������⿼���˻�ѧ��Ӧ�����仯���Ȼ�ѧ����ʽ��д��˹���ɼ���Ӧ�ã�ע�ⷴӦ�ʱ�������ݼ��ܼ��㣬��Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ���Ŀ��ּ�����ҵ�����ҵ����������ϵ�д�
���Ŀ��ּ�����ҵ�����ҵ����������ϵ�д� ����ѵ��ϵ�д�
����ѵ��ϵ�д� ��ĩ�����ϵ�д�
��ĩ�����ϵ�д�
�����Ŀ
��һ�������£��ɷ�����Ӧ��RO3n-+F2+2OH-�TRO4-+2F-+H2O����RO3n-��RԪ�صĻ��ϼ��ǣ�������
| A��+4 | B��+5 | C��+6 | D��+7 |
0.5mol CH4��ȫȼ������CO2��Һ̬H2Oʱ���ų�445kJ���������������Ȼ�ѧ����ʽ����ȷ���ǣ�������
| A��CH4��g��+2O2��g��=CO2��g��+4H2O��g����H=-890 kJ/mol | ||||
| B��CH4��g��+2O2��g��=CO2��g��+2H2O��l����H=+890 kJ/mol | ||||
| C��CH4��g��+2O2��g��=CO2��g��+2H2O��l����H=-890 kJ/mol | ||||
D��
|
��ͼ�׳غ��ҳ��е��ĸ��缫���Ƕ��Բ��ϣ������ͼʾ�ж�����˵����ȷ���ǣ�������
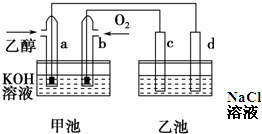

| A���׳��У�b�缫�Ǹ��� |
| B��a�缫�ĵ缫��ӦʽC2H5OH+16OH--12e-=2CO32-+11H2O |
| C��һ��ʱ�����ʪ�����KI��ֽ�����ҳ�d�缫�����ŵ� |
| D���ҳ��ڷ�Ӧǰ����Һ��pH���� |
Ӳ������Ҫ�ɷ��ǣ�������
| A������ | B���ֻ����� |
| C��ɳ�� | D��þ���Ͻ� |
����˵���У���ȷ���ǣ�������
| A��CO��Ħ��������28 g |
| B�����³�ѹ�£�1 mol N2�������22.4 L |
| C��18gH2O���е���ԭ������Ϊ2��6.02��1023 |
| D��0.1 mol/L Na2SO4��Һ�У�Na+�����ʵ���Ũ��Ϊ 0.1 mol/L |