��Ŀ����
��֪��CH3CH2CH2CH2CH2CH2Br
| NaOH�Ҵ� | �� |

��֪DΪ
 ��F1��F2��Ϊͬ���칹�壬�ӳ�HBrʱF1�����ּӳɲ��F2ֻ��һ�ּӳɲ��G1��G2��Ϊͬ���칹�壮����գ�
��F1��F2��Ϊͬ���칹�壬�ӳ�HBrʱF1�����ּӳɲ��F2ֻ��һ�ּӳɲ��G1��G2��Ϊͬ���칹�壮����գ���1��A�Ľṹ��ʽ�ǣ�
��2����ͼ������ȡ����Ӧ���ǣ������ִ��ţ���
��3����ͼ�����ڼӳɷ�Ӧ����
��4��G1�Ľṹ��ʽ�ǣ�


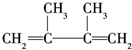 ��E������Br2��Ӧʱ�����ּӳɷ�ʽ����1��2�ӳ�����
��E������Br2��Ӧʱ�����ּӳɷ�ʽ����1��2�ӳ�����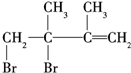 ��1��4�ӳ�����
��1��4�ӳ�����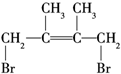 ���ټӳ�HBrʱF1�����ּӳɲ��F2ֻ��һ�ּӳɲ����F2Ϊ
���ټӳ�HBrʱF1�����ּӳɲ��F2ֻ��һ�ּӳɲ����F2Ϊ ��G1Ϊ
��G1Ϊ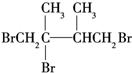 ������л���Ľṹ�����ʽ����⣮
������л���Ľṹ�����ʽ����⣮ ��E������Br2��Ӧʱ�����ּӳɷ�ʽ����1��2�ӳ�����
��E������Br2��Ӧʱ�����ּӳɷ�ʽ����1��2�ӳ����� ��1��4�ӳ�����
��1��4�ӳ����� ���ټӳ�HBrʱF1�����ּӳɲ��F2ֻ��һ�ּӳɲ����F2Ϊ
���ټӳ�HBrʱF1�����ּӳɲ��F2ֻ��һ�ּӳɲ����F2Ϊ ��G1Ϊ
��G1Ϊ ��
����1�������Ϸ�����֪AΪ��CH3��2C�TC��CH3��2���ʴ�Ϊ����CH3��2C�TC��CH3��2��
��2����ͼ�еķ�Ӧֻ���ڹ��������µķ�ӦΪȡ����Ӧ�����ڣ��ʴ�Ϊ���ڣ�
��3�����Ϸ�Ӧ�����ڼӳɷ�Ӧ���Т٢ۢܢޢߢ࣬������ȥ��Ӧ���Тݣ�����ȡ����Ӧ��Ϊ�ڣ�
�ʴ�Ϊ���٢ۢܢޢߢࣻ
��4�������Ϸ�����֪G1Ϊ
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��9�֣���1���±�Ϊϩ��������巢���ӳɷ�Ӧ��������ʣ�����ϩΪ������
| ϩ����� | ������� |
| (CH3)2C=CHCH3 | 10.4 |
| CH3CH=CH2 | 2.03 |
| CH2=CH2 | 1.00 |
| CH2=CHBr | 0.04 |
A��(CH3)2C=C(CH3)2 B��CH3CH=CHCH2CH3 C��CH2="CH" CH3 D��CH2=CHBr
��2��0.5molijȲ���������1molHCl�����ӳɷ�Ӧ�õ��ȴ��������ɵ��ȴ����������3mol Cl2����ȡ����Ӧ������ֻ��C��Cl����Ԫ�صĻ����������Ľṹ��ʽ�� ��
��3��ij������A������Է�������Ϊ104��̼����������Ϊ92.3����
��A�����п��ܹ�ƽ���̼ԭ������� ����
�ڷ�����A��һ�������¿����ɼӾ۸߷��ӣ��ø߷��ӽṹ�е�����Ϊ ��
��һ�������£�A��������Ӧ���õ��Ļ�������̼����������Ϊ85.7����д���γɸû�������л���Ӧ����ʽ ��
����֪
 ����д��A��ϡ�����KMnO4��Һ�ڼ��������·�Ӧ������Ľṹ��ʽ ��
����д��A��ϡ�����KMnO4��Һ�ڼ��������·�Ӧ������Ľṹ��ʽ �� ��9�֣���1���±�Ϊϩ��������巢���ӳɷ�Ӧ��������ʣ�����ϩΪ������
|
ϩ����� |
������� |
|
(CH3)2C=CHCH3 |
10.4 |
|
CH3CH=CH2 |
2.03 |
|
CH2=CH2 |
1.00 |
|
CH2=CHBr |
0.04 |
���л���������ӳ�ʱ��ȡ���������ʵ�Ӱ������й������ƣ����з�Ӧ����������_______________������ţ���
A��(CH3)2C=C(CH3)2 B��CH3CH=CHCH2CH3 C��CH2=CH CH3 D��CH2=CHBr
��2��0.5molijȲ���������1molHCl�����ӳɷ�Ӧ�õ��ȴ��������ɵ��ȴ����������3mol Cl2����ȡ����Ӧ������ֻ��C��Cl����Ԫ�صĻ����������Ľṹ��ʽ�� ��
��3��ij������A������Է�������Ϊ104��̼����������Ϊ92.3����
��A�����п��ܹ�ƽ���̼ԭ������� ����
�ڷ�����A��һ�������¿����ɼӾ۸߷��ӣ��ø߷��ӽṹ�е�����Ϊ ��
��һ�������£�A��������Ӧ���õ��Ļ�������̼����������Ϊ85.7����д���γɸû�������л���Ӧ����ʽ ��
����֪ ����д��A��ϡ�����KMnO4��Һ�ڼ��������·�Ӧ������Ľṹ��ʽ ��
����д��A��ϡ�����KMnO4��Һ�ڼ��������·�Ӧ������Ľṹ��ʽ ��


 ����д��A��ϡ�����KMnO4��Һ�ڼ��������·�Ӧ������Ľṹ��ʽ
����д��A��ϡ�����KMnO4��Һ�ڼ��������·�Ӧ������Ľṹ��ʽ

 ��F������ֻ��������ԭ�ӣ����ҽ��ڲ����ڵ�̼ԭ���ϣ�
��F������ֻ��������ԭ�ӣ����ҽ��ڲ����ڵ�̼ԭ���ϣ�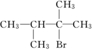


 ��F������ֻ��������ԭ�ӣ����ҽ��ڲ����ڵ�̼ԭ���ϡ�
��F������ֻ��������ԭ�ӣ����ҽ��ڲ����ڵ�̼ԭ���ϡ�