��Ŀ����
Ԫ�����ڱ��е�VIIA��Ԫ�صĵ��ʼ��仯�������;�㷺�����������գ�
��1������Ԫ��ͬ��Ķ�����Ԫ�ص�ԭ�Ӻ����� �ֲ�ͬ�����ĵ��ӣ�
��2������Ϊ�塢��Ԫ�طǽ����ԣ�ԭ�ӵõ����������ݱ���ɵ��ж������� ������ţ���
a��IBr����Ϊ-1�� b��HBr��HI������c��HBr��HI�����ȶ��� d��Br2��I2���۵�
��3����ˮ��������У�����Ũ����ˮ��ͨ�� �������е�Br-���������á�����������������Br2�����ô������գ���֪�������������Ҫ��Ӧ�ǣ�Br2+Na2CO3+H2O��NaBr+NaBrO3+NaHCO3��δ��ƽ����������1mol Br2��ת�Ƶ��� mol����Ӧ����������Ϊ ��
��4����֪��AlF3���۵�Ϊ1040�棬AlCl3��178�������������ʽṹ�ĽǶȽ��������۵����ϴ��ԭ��
��5��HClO�кܺõ�ɱ������Ч����Ϊ��ǿ��ˮ��ɱ�������������ñ�����ˮ��С�մ�Ӧ���ﵽĿ�ģ�����ƽ���ƶ�ԭ��������ԭ�� ��
��1������Ԫ��ͬ��Ķ�����Ԫ�ص�ԭ�Ӻ�����
��2������Ϊ�塢��Ԫ�طǽ����ԣ�ԭ�ӵõ����������ݱ���ɵ��ж�������
a��IBr����Ϊ-1�� b��HBr��HI������c��HBr��HI�����ȶ��� d��Br2��I2���۵�
��3����ˮ��������У�����Ũ����ˮ��ͨ��
��4����֪��AlF3���۵�Ϊ1040�棬AlCl3��178�������������ʽṹ�ĽǶȽ��������۵����ϴ��ԭ��
��5��HClO�кܺõ�ɱ������Ч����Ϊ��ǿ��ˮ��ɱ�������������ñ�����ˮ��С�մ�Ӧ���ﵽĿ�ģ�����ƽ���ƶ�ԭ��������ԭ��
���㣺�ȡ��塢�⼰�仯������ۺ�Ӧ��,ͬһ������Ԫ�����ʵݱ������ԭ�ӽṹ�Ĺ�ϵ
ר�⣺
��������1������Ԫ��ͬ��Ķ�����Ԫ��ΪFԪ�أ�
��2��ͬһ����Ԫ�أ�Ԫ�صķǽ�����Խǿ�����⻯����ȶ���Խǿ��������������ˮ��������Խǿ���䵥�ʵ�������Խǿ���Դ��жϣ�
��3��һ���������������������Br-����ӦBr2+Na2CO3+H2O��NaBr+NaBrO3+NaHCO3�У�BrԪ�ػ��ϼ۷ֱ���0�۱仯Ϊ-1�ۡ�+5�ۣ���ϻ��ϼ۵ı仯���㣻
��4���������Ͳ�ͬ��
��5����ˮ�д���Cl2+H2O HCl+HClO��HCl����̼�����Ʒ�Ӧ����HClO����Ӧ��ƽ�������ƶ���
HCl+HClO��HCl����̼�����Ʒ�Ӧ����HClO����Ӧ��ƽ�������ƶ���
��2��ͬһ����Ԫ�أ�Ԫ�صķǽ�����Խǿ�����⻯����ȶ���Խǿ��������������ˮ��������Խǿ���䵥�ʵ�������Խǿ���Դ��жϣ�
��3��һ���������������������Br-����ӦBr2+Na2CO3+H2O��NaBr+NaBrO3+NaHCO3�У�BrԪ�ػ��ϼ۷ֱ���0�۱仯Ϊ-1�ۡ�+5�ۣ���ϻ��ϼ۵ı仯���㣻
��4���������Ͳ�ͬ��
��5����ˮ�д���Cl2+H2O
 HCl+HClO��HCl����̼�����Ʒ�Ӧ����HClO����Ӧ��ƽ�������ƶ���
HCl+HClO��HCl����̼�����Ʒ�Ӧ����HClO����Ӧ��ƽ�������ƶ������
�⣺��1������Ԫ��ͬ��Ķ�����Ԫ��ΪFԪ�أ�ԭ�Ӻ�����7�����ӣ��ֱ�λ��1s��2s��2p�������3�ֲ�ͬ���������ʴ�Ϊ��3�֣�
��2��a��IBr����Ϊ-1�ۣ�˵��Br�ķǽ����Խ�ǿ����a��ȷ��
b��HBr��HI�����Բ�����Ϊ�жϵ����ݣ���b����
c��HBr��HI�����ȶ���Խǿ����Ӧ�ķǽ�����Խǿ����c��ȷ��
d��Br2��I2���۵������������ʣ�������Ϊ�жϵ����ݣ���d����
�ʴ�Ϊ��a c��
��3��һ���������������������Br-����ӦBr2+Na2CO3+H2O��NaBr+NaBrO3+NaHCO3�У�BrԪ�ػ��ϼ۷ֱ���0�۱仯Ϊ-1�ۡ�+5�ۣ���Ӧ��Br2������������ԭ�����ã����ݵ���ת���غ��֪��2��n��������Br2��=2��5��n��ԭ����Br2������n��������Br2����n��ԭ����Br2��=5��1��������1mol Br2ʱ��ת�Ƶĵ�����Ϊ1mol��2��
��5=
mol=1.67mol��NaBrO3�������
�ʴ�Ϊ��Cl2��H2O2�� 1.67����
����NaBrO3��
��4��AlF3���۵�Ϊ1040�棬AlCl3��178��������ԭ����AlF3�����Ӿ��壬AlCl3�Ƿ��Ӿ��壬���Ӿ����۵�ߣ�
�ʴ�Ϊ��AlF3�����Ӿ��壬AlCl3�Ƿ��Ӿ��壻
��5����ˮ�д���Cl2+H2O HCl+HClO��HCl����̼�����Ʒ�Ӧ����HClO����Ӧ������С�մ���������֮��Ӧ��������Ũ���½���ƽ���������ƶ�����HClO������֮��Ӧ������HClOŨ������
HCl+HClO��HCl����̼�����Ʒ�Ӧ����HClO����Ӧ������С�մ���������֮��Ӧ��������Ũ���½���ƽ���������ƶ�����HClO������֮��Ӧ������HClOŨ������
�ʴ�Ϊ������ˮ�д���Cl2+H2O HCl+HClO����������ǿ�ᣬHClO��̼�ᶼ�����ᣬ��������Ki1��H2CO3����Ki��HClO����Ki2��H2CO3�����ۼ���С�մ���������֮��Ӧ��������Ũ���½���ƽ���������ƶ�����HClO������֮��Ӧ������HClOŨ������
HCl+HClO����������ǿ�ᣬHClO��̼�ᶼ�����ᣬ��������Ki1��H2CO3����Ki��HClO����Ki2��H2CO3�����ۼ���С�մ���������֮��Ӧ��������Ũ���½���ƽ���������ƶ�����HClO������֮��Ӧ������HClOŨ������
��2��a��IBr����Ϊ-1�ۣ�˵��Br�ķǽ����Խ�ǿ����a��ȷ��
b��HBr��HI�����Բ�����Ϊ�жϵ����ݣ���b����
c��HBr��HI�����ȶ���Խǿ����Ӧ�ķǽ�����Խǿ����c��ȷ��
d��Br2��I2���۵������������ʣ�������Ϊ�жϵ����ݣ���d����
�ʴ�Ϊ��a c��
��3��һ���������������������Br-����ӦBr2+Na2CO3+H2O��NaBr+NaBrO3+NaHCO3�У�BrԪ�ػ��ϼ۷ֱ���0�۱仯Ϊ-1�ۡ�+5�ۣ���Ӧ��Br2������������ԭ�����ã����ݵ���ת���غ��֪��2��n��������Br2��=2��5��n��ԭ����Br2������n��������Br2����n��ԭ����Br2��=5��1��������1mol Br2ʱ��ת�Ƶĵ�����Ϊ1mol��2��
| 1 |
| 1+5 |
| 5 |
| 2 |
�ʴ�Ϊ��Cl2��H2O2�� 1.67����
| 5 |
| 3 |
��4��AlF3���۵�Ϊ1040�棬AlCl3��178��������ԭ����AlF3�����Ӿ��壬AlCl3�Ƿ��Ӿ��壬���Ӿ����۵�ߣ�
�ʴ�Ϊ��AlF3�����Ӿ��壬AlCl3�Ƿ��Ӿ��壻
��5����ˮ�д���Cl2+H2O
 HCl+HClO��HCl����̼�����Ʒ�Ӧ����HClO����Ӧ������С�մ���������֮��Ӧ��������Ũ���½���ƽ���������ƶ�����HClO������֮��Ӧ������HClOŨ������
HCl+HClO��HCl����̼�����Ʒ�Ӧ����HClO����Ӧ������С�մ���������֮��Ӧ��������Ũ���½���ƽ���������ƶ�����HClO������֮��Ӧ������HClOŨ�������ʴ�Ϊ������ˮ�д���Cl2+H2O
 HCl+HClO����������ǿ�ᣬHClO��̼�ᶼ�����ᣬ��������Ki1��H2CO3����Ki��HClO����Ki2��H2CO3�����ۼ���С�մ���������֮��Ӧ��������Ũ���½���ƽ���������ƶ�����HClO������֮��Ӧ������HClOŨ������
HCl+HClO����������ǿ�ᣬHClO��̼�ᶼ�����ᣬ��������Ki1��H2CO3����Ki��HClO����Ki2��H2CO3�����ۼ���С�մ���������֮��Ӧ��������Ũ���½���ƽ���������ƶ�����HClO������֮��Ӧ������HClOŨ������
�����������ۺϿ���±��֪ʶ��������ѧ���ķ���������ʵ�������Ŀ��飬Ϊ�߿��������ͺ�Ƶ���㣬ע��������ʵ������Լ������ɵĵݱ���ɣ��ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
�����й���Һ��ɵ������������ǣ�������
| A����ɫ��Һ�п��ܴ�������Al3+��NH4+��Cl?��S2? |
| B������ʱ��c��H+��=1��10-13mol/L����Һ�п��ܴ�������Mg2+��Cu2+��SO42-��NO3- |
| C��0.1mol/LNH4HCO3��Һ�п��ܴ�������K+��Na+��NO3-��Cl- |
| D��������Һ�п��ܴ�������Na+��Ca2+��Cl-��SO42- |
�����£�Ũ�Ⱦ�Ϊ0.1mol/L��4��������ҺpH���£�����˵������ȷ���ǣ�������
| ���� | Na2CO3 | NaHCO3 | NaClO | NaHSO3 |
| pH | 11.6 | 9.7 | 10.3 | 5.2 |
| A��������Һ�У�Na2CO3��Һ��ˮ�ĵ���̶���� |
| B��NaHSO3��Һ�����Ե�ԭ���ǣ�NaHSO3=Na++H++SO32- |
| C������ˮ�м�������NaHCO3��s��������������ˮ�д������Ũ�� |
| D�������£���ͬ���ʵ���Ũ�ȵ�H2SO3��H2CO3��HClO��pH��С����H2SO3 |
����ȼ�ϵ�ص�һ��ͨ���������һ��ͨ�����ͣ���C2H6����������ͣ���������Dz��������ƣ�Y2O3��������ﯣ�ZrO2�����壬�ڽϸ��¶����ܴ���O2-�����жԸ�ȼ�ϵ�ص�˵������ȷ���ǣ�������

| A����缫a��ȼ�ϵ�صĸ��� |
| B�����ɵ�ˮ��B�����������ʹ�C������ |
| C���ڹ��������У�O2-���������� |
| D��ͨ�����͵�һ���Ǹ������缫��ӦΪ��C2H6+7O2--14e-�T2CO2+3H2O |
����װ�ö�����ȡ����������ҩƷѡ�ô�����ǣ�������
A�� |
B�� |
C��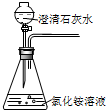 |
D�� |
ij��ɫ��Һ�к��У���K+����Ba2+����Cl-����Br-����SO32-����CO32-����SO42-�е�һ�ֻ��֣����ν�������ʵ�飬��ÿ�������Լ����������۲쵽���������£����н�����ȷ���ǣ�������
| ���� | ���� | ���� |
| �� | ��pH��ֽ���� | ��Һ��pH����7 |
| �� | ����Һ�еμ���ˮ���ټ���CCl4������ | CCl4��ʳ�ɫ |
| �� | ���������Һ�м���Ba��NO3��2��Һ��ϡHNO3 | �а�ɫ�������� |
| �� | ���ˣ�����Һ�м���AgNO3��Һ��ϡHNO3 | �а�ɫ�������� |
| A�����ܺ��е������Ǣ٢ڢ� |
| B���϶����е������Ǣ٢� |
| C������ȷ���������Ǣ٢ۢ� |
| D���϶�û�е������Ǣڢݢ� |
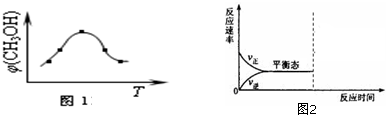
�������ʼ����ۻ�ʱ�ƻ����Թ��ۼ����ǣ�������
| A���ɱ� | B������� |
| C���������� | D���������� |
ij�л�����ҩ���������м��壬��ṹ��ʽ��ͼ�������й�������ȷ���ǣ�������
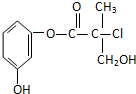

| A�����л�������ˮ�ɷ��ӳɷ�Ӧ |
| B�����л�����Ũ�����Ϲ��ȿɷ�����ȥ��Ӧ |
| C��1 mol���л���������NaOH��Һ��Ӧ�������3 mol NaOH |
| D�����л��ᆳ��������������������ͭ����Һ��������ש��ɫ���� |