��Ŀ����
X��Y��Z��W�����ڱ�ǰ�����ڵ�Ԫ�أ��й����ǵ���Ϣ���±���ʾ����д���пհף�����ʾ����������ĸX��Y��Z��W����
��1��X��Y��Z����Ԫ�ص縺���ɴ�С˳��Ϊ ��
��2��ZԪ�ػ�̬ԭ�ӵĵ����Ų�ʽΪ ��
��3��Wλ��Ԫ�����ڱ��� ���ڵ� �壮
��4����Z��һ���������dz��õľ�ˮ��������ʱ����Һ��pH 7���=�����������������������ǣ� ���������ӷ���ʽ��ʾ��
��5��25�桢101kPaʱ��32gY����ͼ���̬�⻯����ȫȼ�������ȶ���������ʱ�ų�1780.6kJ��������д���÷�Ӧ���Ȼ�ѧ����ʽ ��
| Ԫ�� | ���ֽṹ֪ʶ | �������� |
| X | Xԭ�Ӻ������ռ��9��ԭ�ӹ�� | X��һ�����������γ��������Ҫ���� |
| Y | Yԭ�ӵĴ�������������������������һ�� | Y���γɶ�����̬�⻯�� |
| Z | Zԭ�ӵ���������������2n-3 ��nΪԭ�Ӻ�����Ӳ����� | ��ѧ��Ӧ��Zԭ����ʧȥ���������γ�Zn+ |
| W | W��̬ԭ�ӵ���Χ�����Ų�ʽΪ3d64s2 | �����£�W��ˮ������Ӧ���������ͺ�ɫ������ |
��2��ZԪ�ػ�̬ԭ�ӵĵ����Ų�ʽΪ
��3��Wλ��Ԫ�����ڱ���
��4����Z��һ���������dz��õľ�ˮ��������ʱ����Һ��pH
��5��25�桢101kPaʱ��32gY����ͼ���̬�⻯����ȫȼ�������ȶ���������ʱ�ų�1780.6kJ��������д���÷�Ӧ���Ȼ�ѧ����ʽ
���㣺λ�ýṹ���ʵ����ϵӦ��
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
������X��Y��Z��W�Ƕ����ڵ�����Ԫ�أ�Xԭ�Ӻ������ռ��9��ԭ�ӹ��������������1s��2s��2p��3s��3p�������X��һ�����������γ��������Ҫ���ʣ���XΪ��Ԫ�أ�Yԭ�ӵĴ�������������������������һ�룬��Y��2�����Ӳ㣬����������Ϊ4����YΪ̼Ԫ�أ�Zԭ�ӵ���������������2n-3��nΪԭ�Ӻ�����Ӳ���������ѧ��Ӧ��Zԭ����ʧȥ���������γ�Zn+��ZΪ����Ԫ�أ�����������Ϊn����2n-3=n����n=3����ZΪAlԪ�أ�W��̬ԭ�ӵ���Χ�����Ų�ʽΪ3d64s2������WΪFeԪ�أ��ݴ˴��⣮
���
�⣺X��Y��Z��W�Ƕ����ڵ�����Ԫ�أ�Xԭ�Ӻ������ռ��9��ԭ�ӹ��������������1s��2s��2p��3s��3p�������X��һ�����������γ��������Ҫ���ʣ���XΪ��Ԫ�أ�Yԭ�ӵĴ�������������������������һ�룬��Y��2�����Ӳ㣬����������Ϊ4����YΪ̼Ԫ�أ�Zԭ�ӵ���������������2n-3��nΪԭ�Ӻ�����Ӳ���������ѧ��Ӧ��Zԭ����ʧȥ���������γ�Zn+��ZΪ����Ԫ�أ�����������Ϊn����2n-3=n����n=3����ZΪAlԪ�أ�W��̬ԭ�ӵ���Χ�����Ų�ʽΪ3d64s2������WΪFeԪ�أ�
��1��X��Y��Z�ֱ�ΪS��C��Al������Ԫ�������ɿ�֪�����ǵĵ縺���ɴ�С˳��ΪS��C��Al���ʴ�Ϊ��S��C��Al��
��2��ZΪAlԪ�أ����������Ϊ13����̬ԭ�ӵĵ����Ų�ʽΪ1s22s22p63s23p1���ʴ�Ϊ��1s22s22p63s23p1��
��3��WΪFeԪ�أ�λ��Ԫ�����ڱ��е������ڣ��ڢ����壬�ʴ�Ϊ���ģ�����
��4����W��һ���������dz��õľ�ˮ����ΪAl2��SO4��3����������Һ��������ˮ��Al3++3H2O?Al��OH��3+3H+����ҺpH��7��
�ʴ�Ϊ������Al3++3H2O?Al��OH��3+3H+��
��5��Y����ͼ���̬�⻯��ΪCH4��25�桢101kPaʱ��32gCH4�����ʵ���Ϊ2mol����ȫȼ�������ȶ���������ʱ�ų�1780.6kJ��������1mol������ȫȼ�շų�������Ϊ1780.6kJ��
=890.3kJ���ʸ÷�Ӧ���Ȼ�ѧ��Ӧ����ʽΪ��CH4��g��+2O2��g��=CO2��g��+2H2O��l����H=-890.3KJ/mol��
�ʴ�Ϊ��CH4��g��+2O2��g��=CO2��g��+2H2O��l����H=-890.3KJ/mol��
��1��X��Y��Z�ֱ�ΪS��C��Al������Ԫ�������ɿ�֪�����ǵĵ縺���ɴ�С˳��ΪS��C��Al���ʴ�Ϊ��S��C��Al��
��2��ZΪAlԪ�أ����������Ϊ13����̬ԭ�ӵĵ����Ų�ʽΪ1s22s22p63s23p1���ʴ�Ϊ��1s22s22p63s23p1��
��3��WΪFeԪ�أ�λ��Ԫ�����ڱ��е������ڣ��ڢ����壬�ʴ�Ϊ���ģ�����
��4����W��һ���������dz��õľ�ˮ����ΪAl2��SO4��3����������Һ��������ˮ��Al3++3H2O?Al��OH��3+3H+����ҺpH��7��
�ʴ�Ϊ������Al3++3H2O?Al��OH��3+3H+��
��5��Y����ͼ���̬�⻯��ΪCH4��25�桢101kPaʱ��32gCH4�����ʵ���Ϊ2mol����ȫȼ�������ȶ���������ʱ�ų�1780.6kJ��������1mol������ȫȼ�շų�������Ϊ1780.6kJ��
| 1 |
| 2 |
�ʴ�Ϊ��CH4��g��+2O2��g��=CO2��g��+2H2O��l����H=-890.3KJ/mol��
������������Ҫ������ԭ�Ӻ�������Ų�ʽ��Ԫ�����ڱ��������ˮ�⡢�Ȼ�ѧ����ʽ����д���е��Ѷȣ�����Ĺؼ����ڸ��ݱ����ṩ��ԭ�ӽṹ�ص㼰���������ƶ�Ԫ�أ�

��ϰ��ϵ�д�
 ������ʱ����ҵ����ϵ�д�
������ʱ����ҵ����ϵ�д� ��ĩ���ƾ�ϵ�д�
��ĩ���ƾ�ϵ�д� ���ɿ��ñ���ϵ�д�
���ɿ��ñ���ϵ�д�
�����Ŀ
 ���淴ӦA+aB?C+2D��aΪ��ѧ������������֪B��C��DΪ��̬���ʣ���Ӧ�����У���������������ʱ��C�İٷֺ�����C%�����¶ȣ�T����ѹǿ��p���Ĺ�ϵ��ͼ��ʾ������˵������ȷ���ǣ�������
���淴ӦA+aB?C+2D��aΪ��ѧ������������֪B��C��DΪ��̬���ʣ���Ӧ�����У���������������ʱ��C�İٷֺ�����C%�����¶ȣ�T����ѹǿ��p���Ĺ�ϵ��ͼ��ʾ������˵������ȷ���ǣ�������| A��T2��T1��p2��p1 |
| B���÷�ӦΪ���ȷ�Ӧ |
| C����a=2����AΪҺ̬���̬���� |
| D�����º��ݣ�����B�����ʵ�����B��ת�������� |
�����£���Na2CO3��NaHCO3�����ΰ����ʵ�����1��1��Ϻ�����ˮ���ϡ��Һ�������йظû����Һ˵����ȷ���ǣ�������
| A����Һ��c��CO32-����c��HCO3-����1��1 |
| B����Һ��c��OH-��+��CO32-��=c��H+��+c��HCO3-��+c��H2CO3�� |
| C������Һ�еμ�����ϡ�����NaOH��Һ��HCO3-�����ʵ���������� |
| D�������Һ���ɣ�ˮ��Ӿ磬���յ�NaOH���� |
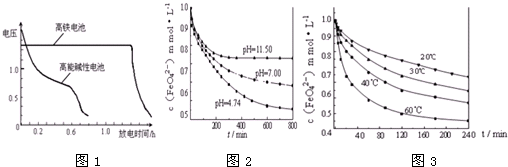
 NaHSO3��Һ�ڲ�ͬ�¶��¾��ɱ�����KIO3��������NaHSO3��ȫ���ļ���I2����������I2��������ʱ��������NaHSO3�ķ�Ӧ���ʣ���Ũ�Ⱦ�Ϊ0.020mol?L-1NaHSO3�����������ۣ�10.0ml��KIO3��������������Һ40.0ml��ϣ���¼10��55�����Һ����ʱ�䣬55��ʱδ�۲쵽��Һ������ʵ������ͼ����ͼ�����������жϲ���ȷ���ǣ�������
NaHSO3��Һ�ڲ�ͬ�¶��¾��ɱ�����KIO3��������NaHSO3��ȫ���ļ���I2����������I2��������ʱ��������NaHSO3�ķ�Ӧ���ʣ���Ũ�Ⱦ�Ϊ0.020mol?L-1NaHSO3�����������ۣ�10.0ml��KIO3��������������Һ40.0ml��ϣ���¼10��55�����Һ����ʱ�䣬55��ʱδ�۲쵽��Һ������ʵ������ͼ����ͼ�����������жϲ���ȷ���ǣ�������| A��40��֮ǰ��40��֮����Һ������ʱ�����¶ȵı仯�����෴ |
| B���¶ȸ���40��ʱ�����۲���������ʵ���ָʾ�� |
| C��ͼ��a���Ӧ��NaHSO3ƽ����Ӧ����Ϊ2.5��10-4mol?L-1?s-1 |
| D��ͼ��b��c�����Ӧ��NaHSO3��Ӧ���ʣ�b��c |
 һ���¶��£�����̼����MCO3��M��Mg2+��Ca2+��Mn2+���ij����ܽ�ƽ��������ͼ��ʾ����֪��pM=-lg c��M����pCO32-=-lg c��CO32-��������˵������ȷ���ǣ�������
һ���¶��£�����̼����MCO3��M��Mg2+��Ca2+��Mn2+���ij����ܽ�ƽ��������ͼ��ʾ����֪��pM=-lg c��M����pCO32-=-lg c��CO32-��������˵������ȷ���ǣ�������| A��MgCO3��CaCO3��MnCO3 ��Ksp���μ�С |
| B��a ��ɱ�ʾMnCO3 �ı�����Һ����c��Mn2+��=c��CO32-�� |
| C��b ��ɱ�ʾ��CaCO3��������Һ����c��Ca2+����c��CO32-�� |
| D��c ��ɱ�ʾMgCO3 �IJ�������Һ����c��Mg2+����c��CO32-�� |